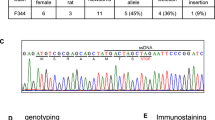
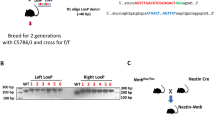
Abstract
Neuromedin U (NMU) is a hypothalamic neuropeptide that regulates body weight and composition. Here we show that mice lacking the gene encoding NMU (Nmu−/− mice) develop obesity. Nmu−/− mice showed increased body weight and adiposity, hyperphagia, and decreased locomotor activity and energy expenditure. Obese Nmu−/− mice developed hyperleptinemia, hyperinsulinemia, late-onset hyperglycemia and hyperlipidemia. Notably, however, treatment with exogenous leptin was effective in reducing body weight in obese Nmu−/− mice. In addition, central leptin administration did not affect NMU gene expression in the hypothalamus of rats. These results indicate that NMU plays an important role in the regulation of feeding behavior and energy metabolism independent of the leptin signaling pathway. These characteristic functions of NMU may provide new insight for understanding the pathophysiological basis of obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Cummings, D.E. & Schwartz, M.W. Genetics and pathophysiology of human obesity. Annu. Rev. Med. 54, 453–471 (2003).
Marx, J. Cellular warriors at the battle of the bulge. Science 299, 846–849 (2003).
Kalra, S.P. et al. Interacting appetite-regulating pathways in the hypothalamic regulation of body weight. Endocr. Rev. 20, 68–100 (1999).
Minamino, N., Kangawa, K. & Matsuo, H. Neuromedin U-8 and U-25: novel uterus stimulating and hypertensive peptides identified in porcine spinal cord. Biochem. Biophys. Res. Commun. 130, 1078–1085 (1985).
Ballesta, J. et al. Occurrence and developmental pattern of neuromedin U-immunoreactive nerves in the gastrointestinal tract and brain of the rat. Neuroscience 25, 797–816 (1988).
Howard, A.D. et al. Identification of receptors for neuromedin U and its role in feeding. Nature 406, 70–74 (2000).
Graham, E.S. et al. Neuromedin U and Neuromedin U receptor-2 expression in the mouse and rat hypothalamus: effects of nutritional status. J. Neurochem. 87, 1165–1173 (2003).
Fujii, R. et al. Identification of neuromedin U as the cognate ligand of the orphan G protein-coupled receptor FM-3. J. Biol. Chem. 275, 21068–21074 (2000).
Kojima, M. et al. Purification and identification of neuromedin U as an endogenous ligand for an orphan receptor GPR66 (FM3). Biochem. Biophys. Res. Commun. 276, 435–438 (2000).
Nakazato, M. et al. Central effects of neuromedin U in the regulation of energy homeostasis. Biochem. Biophys. Res. Commun. 277, 191–194 (2000).
Hanada, R. et al. A role for neuromedin U in stress response. Biochem. Biophys. Res. Commun. 289, 225–228 (2001).
Nakahara, K. et al. The gut-brain peptide neuromedin U is involved in the mammalian circadian oscillator system. Biochem. Biophys. Res. Commun. 318, 156–161 (2004).
Campfield, L.A., Smith, F.J., Guisez, Y., Devos, R. & Burn, P. Recombinant mouse OB protein: evidence for a peripheral signal linking adiposity and central neural networks. Science 269, 546–549 (1995).
Pelleymounter, M.A. et al. Effects of the obese gene product on body weight regulation in ob/ob mice. Science 269, 540–543 (1995).
Schwartz, M.W., Seeley, R.J., Campfield, L.A., Burn, P. & Baskin, D.G. Identification of targets of leptin action in rat hypothalamus. J. Clin. Invest. 98, 1101–1106 (1996).
Robinson, S.W., Dinulescu, D.M. & Cone, R.D. Genetic models of obesity and energy balance in the mouse. Annu. Rev. Genet. 34, 687–745 (2000).
Kurokawa, M., Akino, K. & Kanda, K. A new apparatus for studying feeding and drinking in the mouse. Physiol. Behav. 70, 105–112 (2000).
Nicholls, D.G. A history of UCP1. Biochem. Soc. Trans. 29, 751–755 (2001).
Masaki, T. et al. Corticotropin-releasing hormone-mediated pathway of leptin to regulate feeding, adiposity, and uncoupling protein expression in mice. Endocrinology 144, 3547–3554 (2003).
Schrauwen, P. & Hesselink, M. Uncoupling protein 3 and physical activity: the role of uncoupling protein 3 in energy metabolism revisited. Proc. Nutr. Soc. 62, 635–643 (2003).
Vidal-Puig, A.J. et al. Energy metabolism in uncoupling protein 3 gene knockout mice. J. Biol. Chem. 275, 16258–16266 (2000).
Fan, W., Boston, B.A., Kesterson, R.A., Hruby, V.J. & Cone, R.D. Role of melanocortinergic neurons in feeding and the agouti obesity syndrome. Nature 385, 165–168 (1997).
Abbott, C.R. et al. Investigation of the melanocyte stimulating hormones on food intake. Lack Of evidence to support a role for the melanocortin-3-receptor. Brain. Res. 869, 203–210 (2000).
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131–141 (1997).
Moussa, N.M. & Claycombe, K.J. The yellow mouse obesity syndrome and mechanisms of agouti-induced obesity. Obes. Res. 7, 506–514 (1999).
Egawa, M., Yoshimatsu, H. & Bray, G.A. Effect of corticotropin releasing hormone and neuropeptide Y on electrophysiological activity of sympathetic nerves to interscapular brown adipose tissue. Neuroscience 34, 771–775 (1990).
Ivanov, T.R., Lawrence, C.B., Stanley, P.J. & Luckman, S.M. Evaluation of neuromedin U actions in energy homeostasis and pituitary function. Endocrinology 143, 3813–3821 (2002).
Ozaki, Y. et al. Centrally administered neuromedin U activates neurosecretion and induction of c-fos messenger ribonucleic acid in the paraventricular and supraoptic nuclei of rat. Endocrinology 143, 4320–4329 (2002).
Hanada, T. et al. Central actions of neuromedin U via corticotropin-releasing hormone. Biochem. Biophys. Res. Commun. 311, 954–958 (2003).
Wren, A.M. et al. Hypothalamic actions of neuromedin U. Endocrinology 143, 4227–4234 (2002).
Schwartz, M.W. et al. Leptin increases hypothalamic pro-opiomelanocortin mRNA expression in the rostral arcuate nucleus. Diabetes 46, 2119–2123 (1997).
Mizuno, T.M. & Mobbs, C.V. Hypothalamic agouti-related protein messenger ribonucleic acid is inhibited by leptin and stimulated by fasting. Endocrinology 140, 814–817 (1999).
Elmquist, J.K. & Flier, J.S. Neuroscience. The fat-brain axis enters a new dimension. Science 304, 63–64 (2004).
Schwartz, M.W., Peskind, E., Raskind, M., Boyko, E.J. & Porte, D., Jr. Cerebrospinal fluid leptin levels: relationship to plasma levels and to adiposity in humans. Nat. Med. 2, 589–593 (1996).
Caro, J.F. et al. Decreased cerebrospinal-fluid/serum leptin ratio in obesity: a possible mechanism for leptin resistance. Lancet 348, 159–161 (1996).
Friedman, J.M. & Halaas, J.L. Leptin and the regulation of body weight in mammals. Nature 395, 763–770 (1998).
Masaki, T., Yoshimatsu, H., Chiba, S., Watanabe, T. & Sakata, T. Central infusion of histamine reduces fat accumulation and upregulates UCP family in leptin-resistant obese mice. Diabetes 50, 376–384 (2001).
Withers, P.C. Measurement of VO2, VCO2, and evaporative water loss with a flow-through mask. J. Appl. Physiol. 42, 120–123 (1977).
Yamamoto, Y., Ueta, Y., Yamashita, H., Asayama, K. & Shirahata, A. Expressions of the prepro-orexin and orexin type 2 receptor genes in obese rat. Peptides 23, 1689–1696 (2002).
Nomura, M. et al. Enhanced up-regulation of corticotropin-releasing hormone gene expression in response to restraint stress in the hypothalamic paraventricular nucleus of oxytocin gene-deficient male mice. J. Neuroendocrinol. 15, 1054–1061 (2003).
Yamamoto, Y. et al. Effects of food restriction on the hypothalamic prepro-orexin gene expression in genetically obese mice. Brain. Res. Bull. 51, 515–521 (2000).
Isse, T. et al. Effects of leptin on fasting-induced inhibition of neuronal nitric oxide synthase mRNA in the paraventricular and supraoptic nuclei of rats. Brain. Res. 846, 229–235 (1999).
Hanada, R. et al. Differential regulation of melanin-concentrating hormone and orexin genes in the agouti-related protein/melanocortin-4 receptor system. Biochem. Biophys. Res. Commun. 268, 88–91 (2000).
Acknowledgements
We would like to thank Y. Yamashita, Y. Maruyama, M. Minamino, M. Miyazato, K. Mori, Y. Date, M. Nakazato, H. Ohgusu, M. Naitou, A. Yoshimura and D. Marini for their assistance. This work was supported by the Program for Promotion of Basic Research Activities for Innovative Biosciences (PROBRAIN) (to M.K.), the Fujisawa Foundation, Takeda Science Foundation, Brain Science Foundation, the Naito Foundation and the Mitsubishi Foundation (to M.K.). This work was also supported in part by the Program for Promotion of Fundamental Studies in Health Sciences of Pharmaceuticals and Medical Devices Agency (PMDA) of Japan (to K.K.), and Grant-in-Aids for Scientific Research (S) (to K.K.) (B) (to M.K.), and Young Scientists (B) (to R.H.) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Generation of NMU−/− mice. (PDF 98 kb)
Supplementary Fig. 2
Plasma levels of glucose, insulin, leptin, lipids, corticosterone and assessment of thyroid function in NMU−/− and WT mice after a 16-hr starvation period (n = 16). (PDF 36 kb)
Supplementary Fig. 3
Circadian phenotypes of feeding behavior in NMU−/− mice and WT mouse. (PDF 55 kb)
Supplementary Fig. 4
Suppression of food intake by bolus ICV administration of NMU (3 nmol/mouse or 5nmol/rat) in animals without leptin signaling after fasting (n = 6 per group). (PDF 30 kb)
Rights and permissions
About this article
Cite this article
Hanada, R., Teranishi, H., Pearson, J. et al. Neuromedin U has a novel anorexigenic effect independent of the leptin signaling pathway. Nat Med 10, 1067–1073 (2004). https://doi.org/10.1038/nm1106
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1106
This article is cited by
-
Genomic adaptation to extreme climate conditions in beef cattle as a consequence of cross-breeding program
BMC Genomics (2023)
-
Genes and Signaling Pathways Involved in the Regulation of Selenium-Enriched Yeast on Liver Metabolism and Health of Broiler (Gallus gallus)
Biological Trace Element Research (2023)
-
Neuromedin U-deficient rats do not lose body weight or food intake
Scientific Reports (2022)
-
Genomics of body fat distribution
Journal of Genetics (2021)
-
Convergent and divergent genetic changes in the genome of Chinese and European pigs
Scientific Reports (2017)