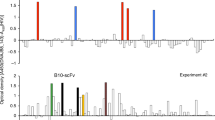
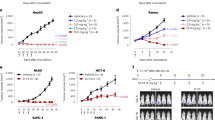
Abstract
We present a system for cancer targeting based on single–chain Fv (scFv) antibodies selected from combinatorial libraries, produced in bacteria and purified by using an engineered tag. Combinatorial libraries of scFv genes contain great diversity, and scFv antibodies with characteristics optimized for a particular task can be selected from them using filamentous bacteriophage. We illustrate the benefits of this system by imaging patients with carcinoembryonic antigen (CEA)–producing cancers using an iodine–123 labeled scFv anti–CEA selected for high affinity. All known tumor deposits were located, and advantages over current imaging technology are illustrated. ScFvs are produced in a cloned form and can be readily engineered to have localizing and therapeutic functions that will be applicable in cancer and other diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Chester, K.A. & Hawkins, R.E. Clinical issues in antibody design. Trends Biotechnol. 13, 294–300 (1995).
Yokota, T., Milenic, D.E., Whitlow, M. & Schlom, J. Rapid tumour penetration of a single-chain Fv and comparison with other immunoglobulin forms. Cancer Res. 52, 3402–3408 (1992).
Huston, J.S. et al. Protein engineering of antibody binding sites: Recovery of specific activity in an anti-digoxin single-chain Fv analogue produced in. Escherichia coll Proc Natl. Acad. Sci. USA 85, 5879–5883 (1988).
Colcher, D. et al. In vivo tumor targeting of a recombinant single-chain antigen-binding protein. J. Natl Cancer Inst 82, 1191–1197 (1990).
Milenic, D.E. et al Construction, binding properties, metabolism, and tumor targeting of a single chain Fv derived from the pancarcinoma monoclonal antibody CC49. Cancer Res. 51, 6363–6371 (1991).
Savage, P. et al Construction, characterisation and kinetics of a single chain antibody recognising the tumour associated antigen placental alkaline phos-phatase. Br. J. Cancer 68, 738–742 (1993).
Huse, W.E. et al Generation of a large combinatorial library of the immunolog-ical repertoire in phage lambda. Science 246, 1275–1281 (1989).
McCafferty, J., Griffiths, A.D., Winter, G. & DJ.Phage antibodies: Filamentous phage displaying antibody variable domains. Nature 348, 552–554 (1990).
Clackson, T., Hoogenboom, H.R., Griffiths, A.D. & Winter, G. Making antibody fragments using phage display libraries. Nature 352, 624–628 (1991).
Hawkins, R.E., Russell, S.J. & Winter, G. Selection of phage antibodies by binding affinity: Mimicking affinity maturation. J. Mol Biol. 226, 889–896 (1992).
Chester, K.A. et al Phage libraries for generation of clinically useful antibodies. Lancet 343, 455–456 (1994).
Griffiths, A.D. et al Isolation of high affinity human antibodies directly from large synthetic repertoires. EMBOJ. 13, 3245–3260 (1994).
Verhaar, M.J. et al A single chain Fv derived from a filamentous phage library has distinct tumour targeting advantages over one derived from a hybridoma. Int.J. Cancer 61, 497–501 (1995).
Casey, J.L. et al Purification of bacterially expressed single chain Fv antibodies for clinical applications using metal chelate chromatography. J. Immunol Methods 179, 105–116 (1995).
Campaign operation manual for control of production, preclinical toxicology and phase I trials of anti tumour antibodies and drug antibody conjugates. Br. J. Cancer 54, 557–568 (1986).
Houghten, R.A. et al Generation and use of synthetic peptide combinatorial libraries for basic research and drug discovery. Nature 354, 84–86 (1991).
Tuerk, C. & Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 249, 505–510 (1990).
Bunin, B.A., Plunket, M.J. & Ellman, J.A. The combinatorial synthesis and chemical and biological evaluation of a 1,4-benzodiazepine library. Proc. Natl. Acad. Sci. USA 91, 4708–4712 (1994).
Green, A.J., Dewhurst, S.E., Begent, R.H., Bagshawe, K.D. & Riggs, S.J. Accurate quantification of 131I distribution by gamma camera imaging. Eur. J. Nucl. Med. 16, 361–365 (1990).
Lane, D.M. et al Radioimmunotherapy of metastatic colorectal tumours with iodine-131-labeled antibody to carcinoembryonic antigen: Phase I/II study with comparative biodistribution of intact and F(ab')2 antibodies. Br. J. Cancer 70, 521–525 (1994).
Begent, R.H.J. J. etal. Antibody distribution and dosimetry in patients receiving radi-olabelled antibody therapy for colorectal cancer. Br. J. Cancer 60, 406–412 (1989).
Takakura, Y., Fujita, T., Hashido, M. & Sezoni, H. Disposition characteristics of macromolecules in tumour-bearing mice. Pharm. Res. 7, 339–346 (1990).
Michael, N.P. et al In vitro and in vivo characterisation of a recombinant car-boxypeptidase G2::anti-CEA scFv fusion protein. Immunotechnology 2, 47–57 (1996).
Bagshawe, K.D., Sharma, S.K., Springer, C.S. & Antoniw, P. Antibody directed enzyme prodrug therapy: Pilot scale clinical trial. Tumour Target. 1, 17–29 (1995).
Yang, W-P. et al. CDR walking mutagenesis for the affinity maturation of a potent human anti-HIV antibody into the picomolar range. J. Mol. Biol. 254, 392–403 (1995).
Barbas, C.F. III.Synthetic human antibodies. Nature Med. 1, 837–839 (1995).
Melton, R.G. et al. Optimisation of small-scale coupling of A5B7 monoclonal antibody to carboxypeptidase G2. J. Immunol. Methods 158, 49–56 (1993).
Moertel, C.G. Chemotherapy for colorectal cancer. N. Engl. J. Med. 330, 1136–1142(1994).
Glimelius, B. B for the Nordic Gastrointestinal Tumour Adjuvant Therapy Group. Expectancy or primary chemotherapy in patients with advanced asymptomatic colorectal cancer: A randomized trial. Eur. J. Cancer S82 (Suppl. 2), 462 (1991).
Verhaar, M.J. et al. 99mTc radiolabelling using a phage-derived single chain Fv with C-terminal cysteine for colorectal tumour imaging. J. Nucl. Med. 37, 868–872 (1996).
Begent, R.H.J. et al Cancer Research Campaign operation manual for control recommendations for products derived from recombinant DNA technology prepared for investigational administration to patients with cancer in phase I trials. Eur. J. Cancer 29A, 1907–1910 (1993).
Hunter, W.M. & Greenwood, F.C. Preparation of iodine-131 labelled human growth hormone of high specific activity. Nature 194, 495–496 (1962).
Ledermann, J.A. et al. A phase-I study of repeated therapy with radiolabelled antibody to carcinoembryonic antigen using intermittent or continuous administration of cyclosporin A to suppress the immune response. Int. J. Cancer 47, 659–664 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Begent, R., Verhaar, M., Chester, K. et al. Clinical evidence of efficient tumor targetting based on single–chain Fv antibody selected from a combinatorial library. Nat Med 2, 979–984 (1996). https://doi.org/10.1038/nm0996-979
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0996-979
This article is cited by
-
scFv biofunctionalized nanoparticles to effective and safe targeting of CEA-expressing colorectal cancer cells
Journal of Nanobiotechnology (2023)
-
A novel Carcinoembryonic Antigen (CEA)-Targeted Trimeric Immunotoxin shows significantly enhanced Antitumor Activity in Human Colorectal Cancer Xenografts
Scientific Reports (2019)
-
The clinical efficacy of first-generation carcinoembryonic antigen (CEACAM5)-specific CAR T cells is limited by poor persistence and transient pre-conditioning-dependent respiratory toxicity
Cancer Immunology, Immunotherapy (2017)
-
Definition and application of good manufacturing process-compliant production of CEA-specific chimeric antigen receptor expressing T-cells for phase I/II clinical trial
Cancer Immunology, Immunotherapy (2014)
-
Nanoparticles: A Promising Modality in the Treatment of Sarcomas
Pharmaceutical Research (2011)