Abstract
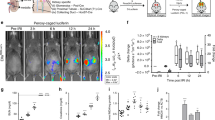
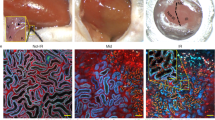
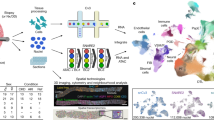
Many proteins have been proposed to act as surrogate markers of organ damage, yet for many candidates the essential biomarker characteristics that link the protein to the injured organ have not yet been described. We generated an Ngal reporter mouse by inserting a double-fusion reporter gene encoding luciferase-2 and mCherry (Luc2-mC) into the Ngal (Lcn2) locus. The Ngal-Luc2-mC reporter accurately recapitulated the endogenous message and illuminated injuries in vivo in real time. In the kidney, Ngal-Luc2-mC imaging showed a sensitive, rapid, dose-dependent, reversible, and organ- and cell-specific relationship with tubular stress, which correlated with the level of urinary Ngal (uNgal). Unexpectedly, specific cells of the distal nephron were the source of uNgal. Cells isolated from Ngal-Luc2-mC mice also revealed both the onset and the resolution of the injury, and the actions of NF-κB inhibitors and antibiotics during infection. Thus, imaging of Ngal-Luc2-mC mice and cells identified injurious and reparative agents that affect kidney damage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bosch, J.P. et al. Renal functional reserve in humans. Effect of protein intake on glomerular filtration rate. Am. J. Med. 75, 943–950 (1983).
Lassnigg, A. et al. Minimal changes of serum creatinine predict prognosis in patients after cardiothoracic surgery: a prospective cohort study. J. Am. Soc. Nephrol. 15, 1597–1605 (2004).
Christensen, E.I. & Verroust, P.J. Interstitial fibrosis: tubular hypothesis versus glomerular hypothesis. Kidney Int. 74, 1233–1236 (2008).
Supavekin, S. et al. Differential gene expression following early renal ischemia/reperfusion. Kidney Int. 63, 1714–1724 (2003).
Mori, K. et al. Endocytic delivery of lipocalin-siderophore-iron complex rescues the kidney from ischemia-reperfusion injury. J. Clin. Invest. 115, 610–621 (2005).
Nickolas, T.L. et al. Sensitivity and specificity of a single emergency department measurement of urinary neutrophil gelatinase-associated lipocalin for diagnosing acute kidney injury. Ann. Intern. Med. 148, 810–819 (2008).
Bennett, M. et al. Urine NGAL predicts severity of acute kidney injury after cardiac surgery: a prospective study. Clin. J. Am. Soc. Nephrol. 3, 665–673 (2008).
Mishra, J. et al. Identification of neutrophil gelatinase-associated lipocalin as a novel early urinary biomarker for ischemic renal injury. J. Am. Soc. Nephrol. 14, 2534–2543 (2003).
Barasch, J. & Mori, K. Cell biology: iron thievery. Nature 432, 811–813 (2004).
Waikar, S., Sabbisetti, V.S. & Bonventre, J. Normalization of urinary biomarkers to creatinine during changes in glomerular filtration rate. Kidney Int. 78, 486–494 (2010).
Mishra, J. et al. Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet 365, 1231–1238 (2005).
Ramphal, R. et al. Control of Pseudomonas aeruginosa in the lung requires the recognition of either lipopolysaccharide or flagellin. J. Immunol. 181, 586–592 (2008).
Li, X., Hassoun, H.T., Santora, R. & Rabb, H. Organ crosstalk: the role of the kidney. Curr. Opin. Crit. Care 15, 481–487 (2009).
Klausen, P., Niemann, C.U., Cowland, J.B., Krabbe, K. & Borregaard, N. On mouse and man: neutrophil gelatinase associated lipocalin is not involved in apoptosis or acute response. Eur. J. Haematol. 75, 332–340 (2005).
Chassin, C. et al. Renal collecting duct epithelial cells react to pyelonephritis-associated Escherichia coli by activating distinct TLR4-dependent and -independent inflammatory pathways. J. Immunol. 177, 4773–4784 (2006).
El-Achkar, T.M. et al. Sepsis induces changes in the expression and distribution of Toll-like receptor 4 in the rat kidney. Am. J. Physiol. Renal Physiol. 290, F1034–F1043 (2006).
Flo, T.H. et al. Lipocalin 2 mediates an innate immune response to bacterial infection by sequestrating iron. Nature 432, 917–921 (2004).
Brzuszkiewicz, E. et al. How to become a uropathogen: comparative genomic analysis of extraintestinal pathogenic Escherichia coli strains. Proc. Natl. Acad. Sci. USA 103, 12879–12884 (2006).
Cowland, J.B., Sorensen, O.E., Sehested, M. & Borregaard, N. Neutrophil gelatinase-associated lipocalin is up-regulated in human epithelial cells by IL-1 beta, but not by TNF-alpha. J. Immunol. 171, 6630–6639 (2003).
Chen, Z. et al. Signal-induced site-specific phosphorylation targets I kappa B alpha to the ubiquitin-proteasome pathway. Genes Dev. 9, 1586–1597 (1995).
Gong, G. et al. Discovery of novel small molecule cell type-specific enhancers of NF-kappaB nuclear translocation. Bioorg. Med. Chem. Lett. 19, 1191–1194 (2009).
Xie, Y. et al. Identification of N-(quinolin-8-yl)benzenesulfonamides as agents capable of down-regulating NFκB activity within two separate high-throughput screens of NFκB activation. Bioorg. Med. Chem. Lett. 18, 329–335 (2008).
Mishra, J. et al. Neutrophil gelatinase-associated lipocalin: a novel early urinary biomarker for cisplatin nephrotoxicity. Am. J. Nephrol. 24, 307–315 (2004).
Haase-Fielitz, A. et al. The predictive performance of plasma neutrophil gelatinase-associated lipocalin (NGAL) increases with grade of acute kidney injury. Nephrol. Dial. Transplant. 24, 3349–3354 (2009).
Hall, I.E. et al. IL-18 and urinary NGAL predict dialysis and graft recovery after kidney transplantation. J. Am. Soc. Nephrol. 21, 189–197 (2010).
Kümpers, P. et al. Serum neutrophil gelatinase-associated lipocalin at inception of renal replacement therapy predicts survival in critically ill patients with acute kidney injury. Crit. Care 14, R9 (2010).
Goetz, D.H. et al. The neutrophil lipocalin NGAL is a bacteriostatic agent that interferes with siderophore-mediated iron acquisition. Mol. Cell 10, 1033–1043 (2002).
Haase, M. et al. Sodium bicarbonate to prevent increases in serum creatinine after cardiac surgery: a pilot double-blind, randomized controlled trial. Crit. Care Med. 37, 39–47 (2009).
Heyman, S., Rosenberger, C. & Rosen, S. Experimental ischemia-reperfusion: biases and myths—the proximal vs. distal hypoxic tubular injury debate revisited. Kidney Int. 77, 9–16 (2010).
Han, K.-H. et al. Effects of ischemia-reperfusion injury on renal ammonia metabolism and the collecting duct. Am. J. Physiol. Renal Physiol. 293, F1342–F1354 (2007).
Srichai, M.B. et al. Apoptosis of the thick ascending limb results in acute kidney injury. J. Am. Soc. Nephrol. 19, 1538–1546 (2008).
di Mari, J.F., Davis, R. & Safirstein, R.L. MAPK activation determines renal epithelial cell survival during oxidative injury. Am. J. Physiol. 277, F195–F203 (1999).
Rosenberger, C. et al. Adaptation to hypoxia in the diabetic rat kidney. Kidney Int. 73, 34–42 (2008).
Brezis, M. & Rosen, S. Hypoxia of the renal medulla–its implications for disease. N. Engl. J. Med. 332, 647–655 (1995).
Steen, H. et al. Cardiac troponin T at 96 hours after acute myocardial infarction correlates with infarct size and cardiac function. J. Am. Coll. Cardiol. 48, 2192–2194 (2006).
Haase, M., Bellomo, R., Devarajan, P., Schlattmann, P. & Haase-Fielitz, A. Accuracy of neutrophil gelatinase-associated lipocalin (NGAL) in diagnosis and prognosis in acute kidney injury: a systematic review and meta-analysis. Am. J. Kidney Dis. 54, 1012–1024 (2009).
Newby, L.K. et al. Value of serial troponin T measures for early and late risk stratification in patients with acute coronary syndromes. The GUSTO-IIa Investigators. Circulation 98, 1853–1859 (1998).
Bolignano, D. et al. Neutrophil gelatinase-associated lipocalin in patients with autosomal-dominant polycystic kidney disease. Am. J. Nephrol. 27, 373–378 (2007).
Damman, K. et al. Both in- and out-hospital worsening of renal function predict outcome in patients with heart failure: results from the Coordinating Study Evaluating Outcome of Advising and Counseling in Heart Failure (COACH). Eur. J. Heart Fail. 11, 847–854 (2009).
Köllisch, G. et al. Various members of the Toll-like receptor family contribute to the innate immune response of human epidermal keratinocytes. Immunology 114, 531–541 (2005).
Rice, B.W., Cable, M.D. & Nelson, M.B. In vivo imaging of light-emitting probes. J. Biomed. Opt. 6, 432–440 (2001).
Lysenko, E.S., Ratner, A.J., Nelson, A.L. & Weiser, J.N. The role of innate immune responses in the outcome of interspecies competition for colonization of mucosal surfaces. PLoS Pathog. 1, e1 (2005).
Zhang, Z. et al. Improved techniques for kidney transplantation in mice. Microsurgery 16, 103–109 (1995).
Acknowledgements
We are grateful for the advice of Q. Al-Awqati and J.A. Oliver. This work was supported by a grant from the Office of Columbia University Technology Ventures. J.B., N.P. and A.Q. are supported by grants from the US National Institute of Diabetes and Digestive and Kidney Diseases (DK-55388 and DK-58872) and the March of Dimes for Birth Defects. Additional funding to J.B. was provided by the Glomerular Center of Columbia University.
Author information
Authors and Affiliations
Contributions
N.P., A.Q. and C.-S.L. created the Ngal reporter mouse; Q.Z. and B.S. performed surgeries; S.-X.D., G.G., Y.L., D.W.L. created NF-κB inhibitors; N.P., R.K. and A.J.R. studied bacteria-induced Ngal expression; V.D. evaluated the pattern of Ngal expression; K.M.S.-O., M.V., W.Y., C.S.F., K.M., A.K. and P.D. analyzed data; N.P., A.Q. and J.B. and wrote the paper; A.Q., N.P. and J.B. invented the luminescent mouse.
Corresponding author
Ethics declarations
Competing interests
Columbia University and Cincinnati Children's Hospital Medical Center have received licensing fees from Biosite (Inverness Medical) and Abbott Diagnostics.
Supplementary information
Supplementary Text and Figures
Supplementary Methods, Supplementary Table 1 and Supplementary Figures 1–12 (PDF 2054 kb)
Rights and permissions
About this article
Cite this article
Paragas, N., Qiu, A., Zhang, Q. et al. The Ngal reporter mouse detects the response of the kidney to injury in real time. Nat Med 17, 216–222 (2011). https://doi.org/10.1038/nm.2290
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2290
This article is cited by
-
Pasteurella multocida activates apoptosis via the FAK-AKT-FOXO1 axis to cause pulmonary integrity loss, bacteremia, and eventually a cytokine storm
Veterinary Research (2024)
-
Renal clearance of graphene oxide: glomerular filtration or tubular secretion and selective kidney injury association with its lateral dimension
Journal of Nanobiotechnology (2023)
-
Lipocalin 2—not only a biomarker: a study of current literature and systematic findings of ongoing clinical trials
Immunologic Research (2023)
-
Aktuelles zu diagnostischen und prognostischen Biomarkern einer diabetischen Nephropathie
Die Diabetologie (2023)
-
Single-cell RNA sequencing and kidney organoid differentiation
Clinical and Experimental Nephrology (2023)