Abstract
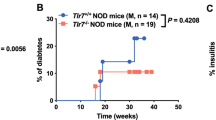
The mechanism of action of natural killer (NK) cells in type 1 diabetes is still unknown. Here we show that the activating receptor NKp46 recognizes mouse and human ligands on pancreatic beta cells. NK cells appeared in the pancreas when insulitis progressed to type 1 diabetes, and NKp46 engagement by beta cells led to degranulation of NK cells. NKp46-deficient mice had less development of type 1 diabetes induced by injection of a low dose of streptozotocin. Injection of soluble NKp46 proteins into nonobese diabetic mice during the early phase of insulitis and the prediabetic stage prevented the development of type 1 diabetes. Our findings demonstrate that NKp46 is essential for the development of type 1 diabetes and highlight potential new therapeutic modalities for this disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Delovitch, T.L. & Singh, B. The nonobese diabetic mouse as a model of autoimmune diabetes: immune dysregulation gets the NOD. Immunity 7, 727–738 (1997).
Kikutani, H. & Makino, S. The murine autoimmune diabetes model: NOD and related strains. Adv. Immunol. 51, 285–322 (1992).
Like, A.A. & Rossini, A.A. Streptozotocin-induced pancreatic insulitis: new model of diabetes mellitus. Science 193, 415–417 (1976).
O'Brien, B.A., Harmon, B.V., Cameron, D.P. & Allan, D.J. Beta-cell apoptosis is responsible for the development of IDDM in the multiple low-dose streptozotocin model. J. Pathol. 178, 176–181 (1996).
Paik, S.G., Fleischer, N. & Shin, S.I. Insulin-dependent diabetes mellitus induced by subdiabetogenic doses of streptozotocin: obligatory role of cell-mediated autoimmune processes. Proc. Natl. Acad. Sci. USA 77, 6129–6133 (1980).
Hutchings, P. et al. Transfer of diabetes in mice prevented by blockade of adhesion-promoting receptor on macrophages. Nature 348, 639–642 (1990).
Miyazaki, A. et al. Predominance of T lymphocytes in pancreatic islets and spleen of pre-diabetic non-obese diabetic (NOD) mice: a longitudinal study. Clin. Exp. Immunol. 60, 622–630 (1985).
Dotta, F. et al. Coxsackie B4 virus infection of beta cells and natural killer cell insulitis in recent-onset type 1 diabetic patients. Proc. Natl. Acad. Sci. USA 104, 5115–5120 (2007).
Rodacki, M. et al. Altered natural killer cells in type 1 diabetic patients. Diabetes 56, 177–185 (2007).
Poirot, L., Benoist, C. & Mathis, D. Natural killer cells distinguish innocuous and destructive forms of pancreatic islet autoimmunity. Proc. Natl. Acad. Sci. USA 101, 8102–8107 (2004).
Alba, A. et al. Natural killer cells are required for accelerated type 1 diabetes driven by interferon-β. Clin. Exp. Immunol. 151, 467–475 (2008).
Flodstrom, M., Shi, F.D., Sarvetnick, N. & Ljunggren, H.G. The natural killer cell–friend or foe in autoimmune disease? Scand. J. Immunol. 55, 432–441 (2002).
Hansson, M., Kiessling, R. & Andersson, B. Human fetal thymus and bone marrow contain target cells for natural killer cells. Eur. J. Immunol. 11, 8–12 (1981).
Nakamura, N. et al. Intrinsic cytotoxicity of natural killer cells to pancreatic islets in vitro. Diabetes 39, 836–843 (1990).
Morse, R.H., Seguin, R., McCrea, E.L. & Antel, J.P. NK cell-mediated lysis of autologous human oligodendrocytes. J. Neuroimmunol. 116, 107–115 (2001).
Karre, K. NK cells, MHC class I molecules and the missing self. Scand. J. Immunol. 55, 221–228 (2002).
Ogasawara, K. et al. Impairment of NK cell function by NKG2D modulation in NOD mice. Immunity 18, 41–51 (2003).
Gazit, R. et al. Lethal influenza infection in the absence of the natural killer cell receptor gene Ncr1. Nat. Immunol. 7, 517–523 (2006).
Moretta, L. Lymphocyte effector mechanisms in innate and adaptive immunity. Curr. Opin. Immunol. 17, 303–305 (2005).
Satoh-Takayama, N. et al. The natural cytotoxicity receptor NKp46 is dispensable for IL-22-mediated innate intestinal immune defense against Citrobacter rodentium. J. Immunol. 183, 6579–6587 (2009).
Ogasawara, K. et al. NKG2D blockade prevents autoimmune diabetes in NOD mice. Immunity 20, 757–767 (2004).
Maier, L.M. et al. NKG2D-RAE-1 receptor-ligand variation does not account for the NK cell defect in nonobese diabetic mice. J. Immunol. 181, 7073–7080 (2008).
Moretta, A., Bottino, C., Mingari, M.C., Biassoni, R. & Moretta, L. What is a natural killer cell? Nat. Immunol. 3, 6–8 (2002).
Mandelboim, O. et al. Recognition of haemagglutinins on virus-infected cells by NKp46 activates lysis by human NK cells. Nature 409, 1055–1060 (2001).
Arnon, T.I. et al. The mechanisms controlling the recognition of tumor- and virus-infected cells by NKp46. Blood 103, 664–672 (2004).
Aktas, E., Kucuksezer, U.C., Bilgic, S., Erten, G. & Deniz, G. Relationship between CD107a expression and cytotoxic activity. Cell. Immunol. 254, 149–154 (2009).
Alter, G., Malenfant, J.M. & Altfeld, M. CD107a as a functional marker for the identification of natural killer cell activity. J. Immunol. Methods 294, 15–22 (2004).
Flodstrom, M., Tyrberg, B., Eizirik, D.L. & Sandler, S. Reduced sensitivity of inducible nitric oxide synthase-deficient mice to multiple low-dose streptozotocin-induced diabetes. Diabetes 48, 706–713 (1999).
Lodoen, M. et al. NKG2D-mediated natural killer cell protection against cytomegalovirus is impaired by viral gp40 modulation of retinoic acid early inducible 1 gene molecules. J. Exp. Med. 197, 1245–1253 (2003).
Halfteck, G.G. et al. Enhanced in vivo growth of lymphoma tumors in the absence of the NK-activating receptor NKp46/NCR1. J. Immunol. 182, 2221–2230 (2009).
Kitagawa, Y. et al. Islet cells but not thyrocytes are susceptible to lysis by NK cells. J. Autoimmun. 4, 703–716 (1991).
MacKay, P., Jacobson, J. & Rabinovitch, A. Spontaneous diabetes mellitus in the Bio-Breeding/Worcester rat. Evidence in vitro for natural killer cell lysis of islet cells. J. Clin. Invest. 77, 916–924 (1986).
Foulis, A.K., McGill, M., Farquharson, M.A. & Hilton, D.A. A search for evidence of viral infection in pancreases of newly diagnosed patients with IDDM. Diabetologia 40, 53–61 (1997).
Horwitz, M.S. et al. Diabetes induced by Coxsackie virus: initiation by bystander damage and not molecular mimicry. Nat. Med. 4, 781–785 (1998).
Lodde, B.M. et al. NOD mouse model for Sjogren's syndrome: lack of longitudinal stability. Oral Dis. 12, 566–572 (2006).
Matsumoto, S. et al. Isolation of tissue progenitor cells from duct-ligated salivary glands of swine. Cloning Stem Cells 9, 176–190 (2007).
Baert, F. et al. Influence of immunogenicity on the long-term efficacy of infliximab in Crohn's disease. N. Engl. J. Med. 348, 601–608 (2003).
Acknowledgements
We thank E. Pikarsky for help and advice; E. Horowitz for assistance; members of the Mandelboim laboratory for discussions and the Physician Research Program of Hadassah Hospital for assistance. Supported by the Juvenile Diabetes Research Foundation (O.M.), the Israel Science Foundation (Morasha grant to C.G.) and the Leifermann Foundation (Y.N.).
Author information
Authors and Affiliations
Contributions
C.G. designed all experiments, did all experiments, analyzed the data and wrote the manuscript; A.P. made the initial observation that NKp46 recognizes beta cells and supervised the project; M.E., R.G., S.M., N.S.-G., H.A., H.G., T.N., O.H. and M.M. contributed reagents; Y.D. provided guidance and reagents; V.D. helped in the immunohistochemical experiments and in determining the insulitis scoring; Y.N. supervised the project and contributed reagents; and O.M. supervised the entire project and analyzed the data, and all experiments were done in the O.M. laboratory under the guidance of O.M.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 (PDF 194 kb)
Supplementary Movie 1
Early PBS treated group. (WMV 3016 kb)
Supplementary Movie 2
Early NKp46 treated group. (WMV 1316 kb)
Rights and permissions
About this article
Cite this article
Gur, C., Porgador, A., Elboim, M. et al. The activating receptor NKp46 is essential for the development of type 1 diabetes. Nat Immunol 11, 121–128 (2010). https://doi.org/10.1038/ni.1834
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1834
This article is cited by
-
Administration of Human Derived Upper gut Commensal Prevotella histicola delays the onset of type 1 diabetes in NOD mice
BMC Microbiology (2022)
-
Mesenchymal stem cells and natural killer cells interaction mechanisms and potential clinical applications
Stem Cell Research & Therapy (2022)
-
The effect of spermidine on autoimmunity and beta cell function in NOD mice
Scientific Reports (2022)
-
High serum IgA and activated Th17 and Treg predict the efficacy of abatacept in patients with early, seropositive rheumatoid arthritis
Clinical Rheumatology (2021)
-
High percentages and activity of synovial fluid NK cells present in patients with advanced stage active Rheumatoid Arthritis
Scientific Reports (2019)