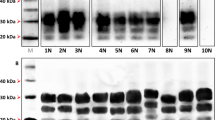
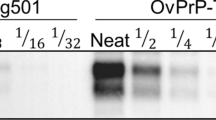
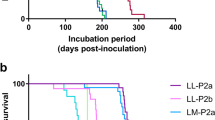
Abstract
Classical genetic analysis has identified Sinc/Prni as the major gene controlling mouse scrapie incubation time. Sinc/Prni is linked to Prnp, the gene encoding the prion protein (PrP). Prnp alleles express distinct PrP protein variants, PrP A and PrP B, which arise from codon 108L/F and 189 T/V dimorphisms. Prnp genotype segregates with incubation time length which suggests, but does not prove, that incubation time is controlled by PrP dimorphisms, and that the Sinc/Prni and Prnp loci are congruent. We have used gene targetting to construct mice in which the endogenous Prnp allele has been modified to express PrP B instead of PrP A. Challenge with a mouse-adapted BSE strain results in dramatically shortened incubation times and demonstrates that PrP dimorphisms at codon 108 and/or 189 control incubation time, and that Sinc/Prni and Prnp are congruent.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Wells, G.A.H. et al. A novel progressuve sponigform encephalopathy in cattle. Vet. Rec. 121, 419–420 (1987).
Creutzfeldt, H.G. Über eine eigenartige herdformige Erkrankung des Zentralnervensystems. Z. Gesamte Neurol. Psychiatrie 57, 247–250 (1920).
Jakob, A. Über eigenartige Erkrankungen des Zentralnervensystems mit bemerkenswertem anatomischen Befunden (spastische Pseudosklerose-Encephalomyopathie mit diseminierten Degenerationsherden). Z. Gesamte Neurol. Psychiatrie 64, 147–228 (1921).
Gerstmann, J., Straussler, E. & Scheinker, I. Über eine eigenartige herediatar-familare Erkankung des Zentralnervensystems zugleich ein Beitrag zur Frage des vorzeitigen lokalen Alterns. Z. Neurol. 154, 736–762 (1936).
Gadjusek, D.C. & Zigas, V. Degenerative disease of the central nervous system in New Guinea: epidemic occurrence of “ Kuru rdquo; in the native population. New Engl. J. Med. 257, 974–978 (1957).
Lugaresi, E. et al Fatal Familial Insomnia and dysautonomia with selective degeneration of thalamic nuclei. New Engl. J. Med. 315, 997–1003 (1986).
DeArmond, S.J. et al Identification of prion amyloid filaments in scrapie-infected brain. Cell 41, 221–235 (1985).
Hope, J. et al The major polypeptide of scrapie associated fibrils (SAF) has the same size, charge distribution and N-terminal protein sequence as predicted for the normal brain protein (PrP). EMBOJ. 10, 2591–2597 (1986).
Bell, J.E. & Ironside, J.W. Neuropathology of spongiform encephalopathies in humans. B. Med. Bull. 49, 738–777 (1993).
Hainfellner, J.A. et al Pathology and immunocytochemistry of a Kuru Brain. Brain Pathology 7, 547–553 (1997).
Basler, K. et al. Scrapie and cellular PrP isoforms are encoded by the same chromosomal gene. Cell 46, 417–428 (1986).
Chandler, R.L. Encephalopathy in mice produced with scrapie brain material. Lancet 1. 1378–1379 (1961).
Dickinson, A.G. & MacKay, J.M.K. Genetical control of the incubation period in mice of the neurological disease, scrapie. Heredity 19, 279–288 (1964).
Dickinson, A.G., Meikle, V. & Fraser, H. Identification of a gene which controls the incubation period of some strains of scrapie agent in mice. J. Comp. Path. 78, 293–299 (1968).
Carlson, G. et al. Linkage of prion protein and scrapie incubation time genes. Cell 46, 503–511 (1986).
Carlson, G. et al Genetics and polymorphism of the mouse prion gene complex: the control of scrapie incubation time. Mol. Cell. Biol. 8, 5528–5540 (1988).
Hunter, N., Hope, J., McConnell, I. & Dickinson, A.G. Linkage of the scrapie associated fibril protein (PrP) gene and Sine using congenic mice and restriction fragment length polymorphism analysis. J. Gen. Virol. 68, 2711–2716 (1987).
Westaway, D. et al Distinct prion proteins in short and long scrapie incubation period mice. Cell 51, 561–662 (1987).
Carlson, G., Ebeling, C., Torchia, M., Westaway, D. & Prusiner, S.B. Delimiting the location of the scrapie prion incubation time gene on chromosome 2 of the mouse. Genetics 133, 979–988 (1993).
Race, R.E., Graham, K., Ernst, D. & Chesebro, B. Analysis of linkage between scrapie incubation period and the prion protein gene in mice. J. Gen. Virol. 71, 493–497 (1990).
Bruce, M.E., McConnell, I., Fraser, H. & Dickinson, A.G. The disease characteristics of different strains of scrapie in Sine congenic mouse lines: implications for the nature of the agent and host control of pathogenesis. J. Gen. Virol. 72, 595–603 (1991).
Hunter, N. et al Are Sine and the PrP gene congruent -evidence from PrP gene analysis in Sine congenic mice. J. Gen. Virol 73, 2751–2755 (1992).
Westaway, D. et al Paradoxical shortening of scrapie incubation times by expression of prion protein transgenes derived from long incubation period mice. Neuron 7, 59–68 (1991).
Carlson, G. et al Prion isolate specified allotype interactions between the cellular and scrapie prion proteins in congenic and transgenic mice. Proc Natl. Acad. Sci. USA 91, 5690–5694 (1994).
Bruce, M.E. & Dickinson, A.G. Genetic control of amyloid plaque production and incubation period in scrapie infected mice. J. Neuropath. Exp. Neurol. 44, 285–294 (1985).
Moore, R.C. et al Double replacement gene targetting for the production of a series of mouse strains with different prion protein gene alterations Biotechnology 13, 999–1004 (1995).
Magin, T.M., McWhir, J. & Melton, D.W. A new mouse embryonic stem cell line with good germ line contribution and targetting frequency. Nucleic Acids Res. 20, 3795–3796 (1992).
Simpson, E.M. et al Genetic variation among 129 substrains and its importance fortargetted mutagenesis in mice. Nature Genet. 16, 19–27 (1997).
Manson, J.C., Clarke, A.R., McBride, P.A., McConnell, I. & Hope, J. PrP gene dosage determines the timing but not the final intensity or distribution of lesions in scrapie pathology. Neurodegeneration 13, 331–340 (1994).
Westaway, D. et al. Structure and polymorphism of the mouse prion protein gene. Proc. Natal. Acad. Sci. USA 91, 6418–6422 (1994).
Baybutt, H. & Manson, J. Characterisation of two promoters for prion protein (PrP) gene expression in neuronal cells. Gene 184, 125–131 (1997).
Prusiner, S.B. et al Transgenic studies implicate interactions between homologous PrP isoforms in scrapie prion replication. Cell 63, 673–686 (1990).
Telling, G.C. et al. Interactions between wild-type and mutant prion proteins modulate neurodegeneration in transgenic mice. Genes Devel. 10, 1736–1750 (1996).
Fischer, M. et al. Prion protein (PrP) with amino-proximal deletions restoring susceptibility of PrP knockout mice to scrapie. EMBO J. 15, 1255–1264 (1996).
Bueler, H. et al. Mice devoid of PrP are resistant to scrapie. Cell 73, 1339–1347 (1993).
Prusiner, S.B. et al Ablation of the prion protein (PrP) gene in mice prevents scrapie and facilitates production of anti-PrP antibodies. Proc. Natal. Acad. Sci. USA 90, 10608–10612 (1993).
Sakaguchi, S. et al Accumulation of proteinase K-resistant Prion protein (PrP) is restricted by the expression level of normal PrP in mice inoculated with a mouse-adapted strain of the Creutzfeldt-Jakob Disease agent. J. Virol. 69, 7586–7592 (1995).
Manson, J.C., McBride, P. & Hope, J. Expression of the PrP gene in the brain of Sine congenic mice and its relationship to the development of scrapie. Neurodegeneration 1, 45–52 (1992).
Meyer, R.K. et al Separation and properties of cellular and scrapie prion proteins. Proc Natal. Acad. Sci. USA 83, 2310–2314 (1986).
Farquhar, C.F. et al Protease resistant PrP deposition in brain and non-central nervous system tissues of a murine model of bovine spongiform encephalopathy. J. Gen. Virol 77, 1941–1946 (1996).
Fraser, H. & Dickinson, A.G. Distribution of experimentally induced scrapie lesions in the brain. Nature 216, 1310–1311 (1967).
Fraser, H. & Dickinson, A.G. The sequential development of the brain lesions of scrapie in three strains of mice. J. Comp. Path. 78, 301–311 (1968).
Kingsbury, D.T. et al. Genetic control of scrapie and Creutzfeldt-Jakob disease in mice. J. Immunology 131, 491–496 (1983).
Bruce, M., Chree, A., McConnell, I. & Wells, G.A.H. Transmission of bovine spongiform encephalopathy and scrapie to mice: strain variation and the species barrier. Phil. Trans. R. Soc. Lond. B 343, 405–411 (1994).
Fraser, H. The pathology of natural and experimental scrapie. In: Slow Virus Diseases of Animals and Man (ed. R.H. Kimberlin. Amsterdam) 267–305 (1976).
Bruce, M.E., McBride, P.A. & Farquhar, C.F. Precise targetting of the pathology of the sialoglycoprotein, PrP, and vacuolar degeneration in mouse scrapie. Neurosci. Lett. 102, 1–6 (1989).
Hecker, R. et al Replication of distinct scrapie prion isolates is region specific in brains of transgenic mice and hamsters. Genes Devel 6, 1213–1228 (1992).
Kunkel, T.A., Roberts, J.D. & Zakour, R.A. Rapid and efficient site-specific mutagenesis without phenotypic selection. Meth. Enzymol. 154, 367–382 (1987).
Thompson, S., Clarke, A.R., Pow, A.M., Hooper, M.L. & Melton, D.W. Germline transmission and expression of a corrected HPRT gene produced by gene targetting in embryonic stem cells. Cell 56, 313–321 (1989).
Fraser, H., Bruce, M.E., Chree, A., McConnell, I. & Wells, G.A.H. Transmission of bovine spongiform encephalopathy and scrapie to mice. J. Gen. Virol. 73, 1891–1897 (1992).
Sternberger, L.A., Hardy, P.H., Cuculis, J.J. & Meyer, H. The unlabelled antibody-enzyme method of immunohistochemistry. Preparation of soluble antigen-antibody complex (horse-radish peroxidase-antihorseradish peroxidase) and its use in identifying spirochetes. J. Histochem. Cytochem. 18, 315–333 (1970).
Farquhar, C.F., Somerville, R.A. & Ritchie, L.A. Postmortem immunodiagnosis of scrapie and bovine spongiform encephalopathy J. Virol. Meth. 24, 215–222 (1989).
Farquhar, C.F. et al A review of the detection of PrPSc. In: Proceedings of a Consultation on BSE with the Scientific Veterinary Committee of the Commission of the European Communities 301–313 (European Commission, Brussels, 1993).
Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685 (1970).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moore, R., Hope, J., McBride, P. et al. Mice with gene targetted prion protein alterations show that Prnp, Sine and Prni are congruent. Nat Genet 18, 118–125 (1998). https://doi.org/10.1038/ng0298-118
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0298-118
This article is cited by
-
Prion strains: shining new light on old concepts
Cell and Tissue Research (2023)
-
New developments in prion disease research using genetically modified mouse models
Cell and Tissue Research (2023)
-
Cellular toxicity of scrapie prions in prion diseases; a biochemical and molecular overview
Molecular Biology Reports (2023)
-
Environmental and host factors that contribute to prion strain evolution
Acta Neuropathologica (2021)
-
Chronic wasting disease (CWD) prion detection in blood from pre-symptomatic white-tailed deer harboring PRNP polymorphic variants
Scientific Reports (2020)