Abstract
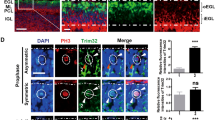
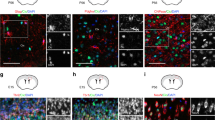
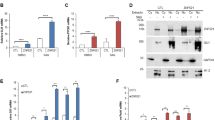
The developmental protein Numb is a major determinant of binary cell fates1,2,3. It is also required for the differentiation of cerebellar granule cell progenitors (GCPs)4 at a stage of development responsive to the morphogenic glycoprotein Hedehog5,6. Hedgehog signalling is crucial for the physiological maintenance and self-renewal of neural stem cells and its deregulation is responsible for their progression towards tumorigenesis5,7,8,9,10,11. The mechanisms that inhibit this pathway during the differentiation stage are poorly understood. Here, we identify Numb as a Hedgehog-pathway inhibitor that is downregulated in early GCPs and GCP-derived cancer cells. We demonstrate that the Hedgehog transcription factor Gli1 is targeted by Numb for Itch-dependent ubiquitination, which suppresses Hedgehog signals, thus arresting growth and promoting cell differentiation. This novel Numb-dependent regulatory loop may limit the extent and duration of Hedgehog signalling during neural-progenitor differentiation, and its subversion may be a relevant event in brain tumorigenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rhyu, M. S., Jan, L. Y. & Jan, Y. N. Asymmetric distribution of numb protein during division of the sensory organ precursor cell confers distinct fates to daughter cells. Cell 76, 477–491 (1994).
Cayouette, M. & Raff, M. Asymmetric segregation of Numb: a mechanism for neural specification from Drosophila to mammals. Nature Neurosci. 5, 1265–1269 (2002).
Shen, Q., Zhong, W., Jan, Y. N. & Temple, S. Asymmetric Numb distribution is critical for asymmetric cell division of mouse cerebral cortical stem cells and neuroblasts. Development 129, 4843–4853 (2002).
Klein, A. L., Zilian, O., Suter, U. & Taylor, V. Murine numb regulates granule cell maturation in the cerebellum. Dev. Biol. 266, 161–177 (2004).
Ruiz i Altaba, A., Sanchez, P. & Dahmane, N. Gli and hedgehog in cancer: tumours, embryos and stem cells. Nature Rev. Cancer 2, 361–372 (2002).
Wechsler-Reya, R. J. & Scott, M. P. Control of neuronal precursor proliferation in the cerebellum by Sonic Hedgehog. Neuron 22, 103–114 (1999).
Lai, K., Kaspar, B. K., Gage, F. H., & Schaffer, D. V. Sonic hedgehog regulates adult neural progenitor proliferation in vitro and in vivo. Nature Neurosci. 6, 21–27 (2003).
Machold, R. et al. Sonic hedgehog is required for progenitor cell maintenance in telencephalic stem cell niches. Neuron 39, 937–950 (2003).
Palma, V. & Ruiz i Altaba, A. Hedgehog-GLI signaling regulates the behavior of cells with stem cell properties in the developing neocortex. Development 131, 337–345 (2004).
Palma, V. et al. Sonic hedgehog controls stem cell behavior in the postnatal and adult brain. Development 132, 335–344 (2005).
Ahn, S. & Joyner, A. L. In vivo analysis of quiescent adult neural stem cells responding to Sonic hedgehog. Nature 437, 894–897 (2005).
Petersen, P. H., Zou, K., Hwang, J. K., Jan, Y. N. & Zhong, W. Progenitor cell maintenance requires numb and numblike during mouse neurogenesis. Nature 419, 929–934 (2002).
Li, H. S. et al. Inactivation of Numb and Numblike in embryonic dorsal forebrainimpairs neurogenesis and disrupts cortical morphogenesis. Neuron 40, 1105–1118 (2003).
McGill, M. A. & McGlade, C. J. Mammalian numb proteins promote Notch1 receptor ubiquitination and degradation of the Notch1 intracellular domain. J. Biol. Chem. 278, 23196–23203 (2003).
Berdnik, D. et al. The endocytic protein α-Adaptin is required for Numb-mediated asymmetric cell division in Drosophila. Dev. Cell 3, 221–231 (2002).
Verdi, J. M. et al. Distinct human NUMB isoforms regulate differentiation versus proliferation in the neuronal lineage. Proc. Natl Acad. Sci. USA 96, 10472–10476 (1999).
Wakamatsu, Y., Maynard, T. M., Jones, S. U. & Weston, J. A. NUMB localizes in the basal cortex of mitotic avian neuroepithelial cells and modulates neuronal differentiation by binding to NOTCH-1. Neuron 23, 71–81 (1999).
Jiang, J. & Struhl, G. Regulation of the Hedgehog and Wingless signalling pathways by the F-box/WD40-repeat protein Slimb. Nature 391, 493–496 (1998).
Huntzicker, E. G. et al. Dual degradation signals control Gli protein stability and tumor formation. Genes Dev. 20, 276–281 (2006).
Wang, B. & Li, Y. Evidence for the direct involvement of βTrCP in Gli3 protein processing. Proc. Natl Acad. Sci. USA 103, 33–38 (2006).
Pan, Y. et al. Sonic hedgehog signaling regulates Gli2 transcriptional activity by suppressing its processing and degradation. Mol. Cell Biol. 26, 3365–3377 (2006).
Zhong, W., Feder, J. N., Jiang, M. M., Jan, L. Y. & Jan, Y. N. Asymmetric localization of a mammalian numb homolog during mouse cortical neurogenesis. Neuron 17, 43–53 (1996).
Guo, M., Jan, L. Y. & Jan, Y. N. Control of daughter cell fates during asymmetric division: interaction of Numb and Notch. Neuron 17, 27–41 (1996).
Argenti, B. et al. Hedgehog antagonist REN(KCTD11) regulates proliferation and apoptosis of developing granule cell progenitors. J. Neurosci. 25, 8338–8346 (2005).
Angers, A., Ramjaun, A. R. & McPherson, P. S. The HECT domain ligase itch ubiquitinates endophilin and localizes to the trans-Golgi network and endosomal system. J. Biol. Chem. 279, 11471–11479 (2004).
Santolini, E. at al. Numb is an endocytic protein. J. Cell Biol. 151, 1345–1351 (2000).
Ferretti, E., De Smaele, E., Di Marcotullio, L., Screpanti, I. & Gulino, A. Hedgehog checkpoints in medulloblastoma: the chromosome 17p deletion paradigm. Trends Mol. Med. 11, 537–545 (2005).
Di Marcotullio, L. et al. REN(KCTD11) is a suppressor of Hedgehog signaling and is deleted in human medulloblastoma. Proc. Natl Acad. Sci. USA 101, 10833–10838 (2004).
Caussinus, E. & Gonzalez, C. Induction of tumor growth by altered stem-cell asymmetric division in Drosophila melanogaster. Nature Genet. 37, 1125–1129 (2005).
Hallahan, A. R. et al. The SmoA1 mouse model reveals that notch signaling is critical for the growth and survival of sonic hedgehog-induced medulloblastomas. Cancer Res. 64, 7794–7800 (2004).
Fan, X. et al. Notch1 and notch2 have opposite effects on embryonal brain tumor growth. Cancer Res. 64, 7787–7793 (2004).
Pece, S. et al. Loss of negative regulation by Numb over Notch is relevant to human breast carcinogenesis. J. Cell Biol. 167, 215–221 (2004).
Roncarati, R. et al. The γ-secretase-generated intracellular domain of β-amyloid precursor protein binds Numb and inhibits Notch signaling. Proc. Natl Acad. Sci. USA 99, 7102–7107 (2002).
Ricci-Vitiani, L. et al. Absence of caspase 8 and high expression of PED protect primitive neural cells from cell death. J. Exp. Med. 200, 1257–1266 (2004).
Hahn, H. et al. Rhabdomyosarcomas and radiation hypersensitivity in a mouse model of Gorlin syndrome. Nature Med. 4, 619–622 (1998).
Acknowledgements
This work was supported by the Associazione Italiana per la Ricerca sul Cancro, Telethon Grant GGP04168, the Ministry of University & Research and National Research Council, the Ministry of Health, the Center of Excellence for Biology and Molecular Medicine and the Rome Oncogenomic Center. E.D.S. is supported by an Fondazione Italiana per la Ricerca sul Cancro (FIRC) fellowship. We thank all mentioned colleagues and R. De Maria for reagents, G. Canettieri and R. Paolini for sharing data and advice, M. Levrero for critical discussion and C. Fragomeli, T. Natale and S. Espinola for experimental assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures S1, S2, S3, S4 and S5 (PDF 542 kb)
Rights and permissions
About this article
Cite this article
Marcotullio, L., Ferretti, E., Greco, A. et al. Numb is a suppressor of Hedgehog signalling and targets Gli1 for Itch-dependent ubiquitination. Nat Cell Biol 8, 1415–1423 (2006). https://doi.org/10.1038/ncb1510
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1510
This article is cited by
-
SALL4 is a CRL3REN/KCTD11 substrate that drives Sonic Hedgehog-dependent medulloblastoma
Cell Death & Differentiation (2024)
-
Ubiquilin-4 induces immune escape in gastric cancer by activating the notch signaling pathway
Cellular Oncology (2023)
-
The role of Hedgehog and Notch signaling pathway in cancer
Molecular Biomedicine (2022)
-
Numb exon 9 inclusion regulates Integrinβ5 surface expression and promotes breast cancer metastasis
Oncogene (2022)
-
NUMB facilitates autophagy initiation through targeting SCFβ-TrCP2 complex
Cell Death & Differentiation (2022)