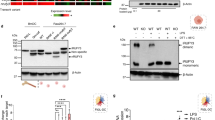
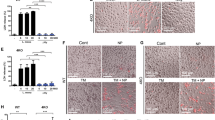
Abstract
Homologues of signal peptide peptidase (SPPLs) are putative aspartic proteases that may catalyse regulated intramembrane proteolysis of type II membrane-anchored signalling factors. Here, we show that four human SPPLs are each sorted to a different compartment of the secretory pathway. We demonstrate that SPPL2a and SPPL2b, which are sorted to endosomes and the plasma membrane, respectively, are functional proteases that catalyse intramembrane cleavage of tumour necrosis factor alpha (TNFα). The two proteases promoted the release of the TNFα intracellular domain, which in turn triggers expression of the pro-inflammatory cytokine interleukin-12 by activated human dendritic cells. Our study reveals a critical function for SPPL2a and SPPL2b in the regulation of innate and adaptive immunity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Brown, M. S., Ye, J., Rawson, R. B. & Goldstein, J. L. Regulated intramembrane proteolysis: a control mechanism conserved from bacteria to humans. Cell 100, 391–398 (2000).
Rawson, R. B. Regulated intramembrane proteolysis: from the endoplasmic reticulum to the nucleus. Essays Biochem. 38, 155–168 (2002).
Brunkan, A. L. & Goate, A. M. Presenilin function and γ-secretase activity. J. Neurochem. 93, 769–792 (2005).
Weihofen, A. & Martoglio, B. Intramembrane-cleaving proteases: controlled liberation of proteins and bioactive peptides. Trends Cell Biol. 13, 71–78 (2003).
Haass, C. & Steiner, H. Alzheimer disease γ-secretase: a complex story of GxGD-type presenilin proteases. Trends Cell Biol. 12, 556–562 (2002).
Rawson, R. B. et al. Complementation cloning of S2P, a gene encoding a putative metalloprotease required for intramembrane cleavage of SREBPs. Mol. Cell 1, 47–57 (1997).
Ye, J. et al. ER stress induces cleavage of membrane-bound ATF6 by the same proteases that process SREBPs. Mol. Cell 6, 1355–1364 (2000).
Eissner, G., Kolch, W. & Scheurich, P. Ligands working as receptors: reverse signaling by members of the TNF superfamily enhance the plasticity of the immune system. Cytokine Growth Factor Rev. 15, 353–366 (2004).
Laudon, H. et al. A nine-transmembrane domain topology for presenilin 1. J. Biol. Chem. 280, 35352–35360 (2005).
Martoglio, B. & Golde, T. E. Intramembrane-cleaving aspartic proteases and disease: presenilins, signal peptide peptidase and their homologs. Hum. Mol. Genet. 12, R201–R206 (2003).
Friedmann, E. et al. Consensus analysis of signal peptide peptidase and homologous human aspartic proteases reveals opposite topology of catalytic domains compared with presenilins. J. Biol. Chem. 279, 50790–50798 (2004).
Weihofen, A., Binns, K., Lemberg, M. K., Ashman, K. & Martoglio, B. Identification of signal peptide peptidase, a presenilin-type aspartic protease. Science 296, 2215–2218 (2002).
Lemberg, M. K. & Martoglio, B. Requirements for signal peptide peptidase-catalyzed intramembrane proteolysis. Mol. Cell 10, 735–744 (2002).
Krawitz, P. et al. Differential localization and identification of a critical aspartate suggest non-redundant proteolytic functions of the presenilin in homologues SPPL2b and SPPL3. J. Biol. Chem. 280, 39515–39523 (2005).
Aggarwal, B. B. Signalling pathways of the TNF superfamily: a double-edged sword. Nature Rev. Immunol. 3, 745–756 (2003).
Weihofen, A. et al. Targeting presenilin-type aspartic protease signal peptide peptidase with γ-secretase inhibitors. J. Biol. Chem. 278, 16528–16533 (2003).
Watts, A. D., Hunt, N. H., Madigan, M. C. & Chaudhri, G. Soluble TNF-α receptors bind and neutralize over-expressed transmembrane TNF-α on macrophages, but do not inhibit its processing. J. Leukoc. Biol. 66, 1005–1013 (1999).
Banchereau, J. & Steinman, R. M. Dendritic cells and the control of immunity. Nature 392, 245–252 (1998).
Cella, M., Sallusto, F. & Lanzavecchia, A. Origin, maturation and antigen presenting function of dendritic cells. Curr. Opin. Immunol. 9, 10–16 (1997).
Trinchieri, G., Pflanz, S. & Kastelein, R. A. The IL-12 family of heterodimeric cytokines: new players in the regulation of T cell responses. Immunity 19, 641–644 (2003).
Fujii, S., Liu, K., Smith, C., Bonito, A. J. & Steinman, R. M. The linkage of innate to adaptive immunity via maturing dendritic cells in vivo requires CD40 ligation in addition to antigen presentation and CD80/86 costimulation. J. Exp. Med. 199, 1607–1618 (2004).
Ma, X. TNF-α and IL-12: a balancing act in macrophage functioning. Microbes Infect. 3, 121–129 (2001).
Cartstea, E. D., Hough, S., Wiederholt, K. & Welch, P. J. State-of-the-art modified RNAi compounds for therapeutics. IDrug 8, 642–647 (2005).
Yamada, Y., Kirillova, I., Peschon, J. J. & Fausto, N. Initiation of liver growth by tumor necrosis factor: deficient liver regeneration in mice lacking type I tumor necrosis factor receptor. Proc. Natl Acad. Sci. USA 94, 1441–1446 (1997).
Steiner, H. et al. Glycine 384 is required for presenilin-1 function and is conserved in bacterial polytopic aspartyl proteases. Nature Cell Biol. 2, 848–851 (2000).
Moss, M. L. et al. Cloning of a disintegrin metalloproteinase that processes precursor tumour-necrosis factor-α. Nature 385, 733–736 (1997).
Peck, R., Brockhaus, M. & Frey, J. R. Cell surface tumor necrosis factor (TNF) accounts for monocyte- and lymphocyte-mediated killing of TNF-resistant target cells. Cell. Immunol. 122, 1–10 (1989).
Perez, C. et al. A nonsecretable cell surface mutant of tumor necrosis factor (TNF) kills by cell-to-cell contact. Cell 63, 251–258 (1990).
Grell, M. et al. The transmembrane form of tumor necrosis factor is the prime activating ligand of the 80 kDa tumor necrosis factor receptor. Cell 83, 793–802 (1995).
Eissner, G. et al. Reverse signaling through transmembrane TNF confers resistance to lipopolysaccharide in human monocytes and macrophages. J. Immunol. 164, 6193–6198 (2000).
Domonkos, A., Udvardy, A., Laszlo, L., Nagy, T. & Duda, E. Receptor-like properties of the 26 kDa transmembrane form of TNF. Eur. Cytokine Netw. 12, 411–419 (2001).
Hauben, E., Roncarolo, M. G., Nevo, U. & Schwartz, M. Beneficial autoimmunity in Type 1 diabetes mellitus. Trends Immunol. 26, 248–253 (2005).
Hammond, C. & Helenius, A. Quality control in the secretory pathway: retention of a misfolded viral membrane glycoprotein involves cycling between the ER, intermediate compartment, and Golgi apparatus. J. Cell Biol. 126, 41–52 (1994).
Wiltfang, J. et al. Improved electrophoretic separation and immunoblotting of β-amyloid (Aβ) peptides 1-40, 1-42, and 1-43. Electrophoresis 18, 527–532 (1997).
Acknowledgements
We thank B. Cenni and C. Haass for critical comments on the manuscript, and E. Duda and A. Helenius for antibodies. This work was supported by ETH Zurich and grants from the Centre of Neuroscience Zurich, and the National Competence Centre for Research on Neuronal Plasticity and Repair to B.M. E.H. was supported by a fellowship from the International Human Frontier Science Program Organization.
Author information
Authors and Affiliations
Contributions
E.F. and E.H. performed the main experimental work and data analysis with HeLa cells and dendritic cells, respectively, and contributed to the writing of the manuscript. K.M., S.S., S.V., S.F.L., P.H.K and D.S. also performed experimental work. R.G. contributed to project planning and writing of the manuscript. B.M. was responsible for project planning and guidance, data analysis and writing the manuscript.
Note: Supplementary Information is available on the Nature Cell Biology website.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1 and S2 (PDF 384 kb)
Rights and permissions
About this article
Cite this article
Friedmann, E., Hauben, E., Maylandt, K. et al. SPPL2a and SPPL2b promote intramembrane proteolysis of TNFα in activated dendritic cells to trigger IL-12 production. Nat Cell Biol 8, 843–848 (2006). https://doi.org/10.1038/ncb1440
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1440
This article is cited by
-
Deep post-GWAS analysis identifies potential risk genes and risk variants for Alzheimer’s disease, providing new insights into its disease mechanisms
Scientific Reports (2021)
-
Genome-wide insights on gastrointestinal nematode resistance in autochthonous Tunisian sheep
Scientific Reports (2021)
-
A lysosomal enigma CLN5 and its significance in understanding neuronal ceroid lipofuscinosis
Cellular and Molecular Life Sciences (2021)
-
Intramembrane proteolysis of an extracellular serine protease, epithin/PRSS14, enables its intracellular nuclear function
BMC Biology (2020)
-
Heme binding of transmembrane signaling proteins undergoing regulated intramembrane proteolysis
Communications Biology (2020)