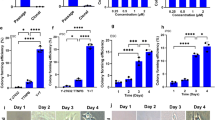
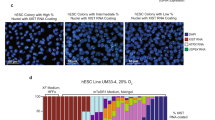
Abstract
The application of human embryonic stem (hES) cells in regenerative medicine will require rigorous quality control measures to ensure the safety of hES cell–derived grafts. During propagation in vitro, hES cells can acquire cytogenetic abnormalities1,2,3 as well as submicroscopic genetic lesions, such as small amplifications or deletions4. Many of the genetic abnormalities that arise in hES cell cultures are also implicated in human cancer development. The causes of genetic instability of hES cells in culture are poorly understood, and commonly used cytogenetic methods for detection of abnormal cells are capable only of low-throughput analysis on small numbers of cells. The identification of biomarkers of genetic instability in hES cells would greatly facilitate the development of culture methods that preserve genomic integrity. Here we show that CD30, a member of the tumor necrosis factor receptor superfamily, is expressed on transformed but not normal hES cells, and that CD30 expression protects hES cells against apoptosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Draper, J.S. et al. Recurrent gain of chromosomes 17q and 12 in cultured human embryonic stem cells. Nat. Biotechnol. 22, 53–54 (2004).
Cowan, C.A. et al. Derivation of embryonic stem-cell lines from human blastocysts. N. Engl. J. Med. 350, 1353–1356 (2004).
Zeng, X. et al. BG01V: a variant human embryonic stem cell line which exhibits rapid growth after passaging and reliable dopaminergic differentiation. Restor. Neurol. Neurosci. 22, 421–428 (2004).
Maitra, A. et al. Genomic alterations in cultured human embryonic stem cells. Nat. Genet. 37, 1099–1103 (2005).
Durkop, H. et al. Molecular cloning and expression of a new member of the nerve growth factor receptor family that is characteristic for Hodgkin's disease. Cell 68, 421–427 (1992).
Horie, R. & Watanabe, T. CD30: expression and function in health and disease. Semin. Immunol. 10, 457–470 (1998).
Durkop, H. et al. Expression of the CD30 antigen in non-lymphoid tissues and cells. J. Pathol. 190, 613–618 (2000).
Pera, M.F., Bennett, W. & Cerretti, D.P. Expression of CD30 and CD30 ligand in cultured cell lines from human germ-cell tumors. Lab. Invest. 76, 497–504 (1997).
Horie, R. et al. A variant CD30 protein lacking extracellular and transmembrane domains is induced in HL-60 by tetradecanoylphorbol acetate and is expressed in alveolar macrophages. Blood 88, 2422–2432 (1996).
Croager, E.J., Gout, A.M. & Abraham, L.J. Involvement of Sp1 and microsatellite repressor sequences in the transcriptional control of the human CD30 gene. Am. J. Pathol. 156, 1723–1731 (2000).
Amit, M. et al. Clonally derived human embryonic stem cell lines maintain pluripotency and proliferative potential for prolonged periods of culture. Dev. Biol. 227, 271–278 (2000).
Xu, C. et al. Feeder-free growth of undifferentiated human embryonic stem cells. Nat. Biotechnol. 19, 971–974 (2001).
Oosterhuis, J.W. & Looijenga, L.H. Testicular germ-cell tumours in a broader perspective. Nat. Rev. Cancer 5, 210–222 (2005).
Henegariu, O. et al. Characterization of gains, losses, and regional amplification in testicular germ cell tumor cell lines by comparative genomic hybridization. Cancer Genet. Cytogenet. 148, 14–20 (2004).
Pallesen, G. & Hamilton-Dutoit, S.J. Ki-1 (CD30) antigen is regularly expressed by tumor cells of embryonal carcinoma. Am. J. Pathol. 133, 446–450 (1988).
Pera, M.F., Bennett, W. & Cerretti, D.P. CD30 and its ligand: possible role in regulation of teratoma stem cells. APMIS 106, 169–173 (1998).
Amakawa, R. et al. Impaired negative selection of T cells in Hodgkin's disease antigen CD30-deficient mice. Cell 84, 551–562 (1996).
Reubinoff, B.E., Pera, M.F., Fong, C.Y., Trounson, A. & Bongso, A. Embryonic stem cell lines from human blastocysts: somatic differentiation in vitro. Nat. Biotechnol. 18, 399–404 (2000).
Thomson, J.A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 (1998).
Buzzard, J.J., Gough, N.M., Crook, J.M. & Colman, A. Karyotype of human ES cells during extended culture. Nat. Biotechnol. 22, 381–382, author reply, 382 (2004).
Pera, M.F., Cooper, S., Mills, J. & Parrington, J.M. Isolation and characterization of a multipotent clone of human embryonal carcinoma cells. Differentiation 42, 10–23 (1989).
Kadin, M.E. Regulation of CD30 antigen expression and its potential significance for human disease. Am. J. Pathol. 156, 1479–1484 (2000).
Enver, T. et al. Cellular differentiation hierarchies in normal and culture-adapted human embryonic stem cells. Hum. Mol. Genet. 14, 3129–3140 (2005).
Andrews, P.W. et al. The International Stem Cell Initiative: toward benchmarks for human embryonic stem cell research. Nat. Biotechnol. 23, 795–797 (2005).
Doronina, S.O. et al. Development of potent monoclonal antibody auristatin conjugates for cancer therapy. Nat. Biotechnol. 21, 778–784 (2003).
Pera, M.F., Blasco Lafita, M.J. & Mills, J. Cultured stem-cells from human testicular teratomas: the nature of human embryonal carcinoma, and its comparison with two types of yolk-sac carcinoma. Int. J. Cancer 40, 334–343 (1987).
Herszfeld, D. & Pera, M.F. Differentiation of human pluripotent teratocarcinoma cells induced by bone morphogenetic protein-2. Reprod. Fertil. Dev. 10, 551–555 (1998).
Pera, M.F. et al. Regulation of human embryonic stem cell differentiation by BMP-2 and its antagonist noggin. J. Cell Sci. 117, 1269–1280 (2004).
Roach, S., Schmid, W. & Pera, M.F. Hepatocytic transcription factor expression in human embryonal carcinoma and yolk sac carcinoma cell lines: expression of HNF-3α in models of early endodermal cell differentiation. Exp. Cell Res. 215, 189–198 (1994).
Pera, M.F., Filipczyk, A.A., Hawes, S.M. & Laslett, A.L. Isolation, characterization, and differentiation of human embryonic stem cells. Methods Enzymol. 365, 429–446 (2003).
Acknowledgements
Work in the Monash Institute of Medical Research and the Australian Stem Cell Centre was supported by the National Health and Medical Research Council, The Australian Stem Cell Centre and the National Institutes of Health (NIGMS GM68417). Work in the laboratory of L.H.J.L. was supported in part by the Dutch Cancer Society/KWF.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
M.P. and B.R. hold shares in ES Cell International.
Supplementary information
Supplementary Fig. 1
Immunostaining of diploid ES or EC cells with antibodies to CD30. (PDF 1390 kb)
Supplementary Fig. 2
Southern analysis of CD30 gene and PCR analysis of a repeat region in its promoter in normal and GCT DNA and EC cell lines. (PDF 307 kb)
Supplementary Fig. 3
Differentiation of diploid or karyotypically abnormal hESC in vitro. (PDF 27 kb)
Supplementary Fig. 4
Differentiation of diploid or karyotypically abnormal ES or EC cells in vivo. (PDF 1412 kb)
Supplementary Fig. 5
Semiquantitative RT-PCR analysis of the expression of CD30 and CD30 variant in control and BMP-4 treated hESC. (PDF 194 kb)
Supplementary Table 1
Karyotype analysis of ES cell lines grown under different conditions. (PDF 42 kb)
Rights and permissions
About this article
Cite this article
Herszfeld, D., Wolvetang, E., Langton-Bunker, E. et al. CD30 is a survival factor and a biomarker for transformed human pluripotent stem cells. Nat Biotechnol 24, 351–357 (2006). https://doi.org/10.1038/nbt1197
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1197
This article is cited by
-
Sustained intrinsic WNT and BMP4 activation impairs hESC differentiation to definitive endoderm and drives the cells towards extra-embryonic mesoderm
Scientific Reports (2021)
-
Comprehensive genetic analysis of pediatric germ cell tumors identifies potential drug targets
Communications Biology (2020)
-
Testicular cancer
Nature Reviews Disease Primers (2018)
-
Emerging innovation towards safety in the clinical application of ESCs and iPSCs
Nature Reviews Cardiology (2014)