Abstract
The process of carbon capture and sequestration has been proposed as a method of mitigating the build-up of greenhouse gases in the atmosphere. If implemented, the cost of electricity generated by a fossil fuel-burning power plant would rise substantially, owing to the expense of removing CO2 from the effluent stream. There is therefore an urgent need for more efficient gas separation technologies, such as those potentially offered by advanced solid adsorbents. Here we show that diamine-appended metal-organic frameworks can behave as ‘phase-change’ adsorbents, with unusual step-shaped CO2 adsorption isotherms that shift markedly with temperature. Results from spectroscopic, diffraction and computational studies show that the origin of the sharp adsorption step is an unprecedented cooperative process in which, above a metal-dependent threshold pressure, CO2 molecules insert into metal-amine bonds, inducing a reorganization of the amines into well-ordered chains of ammonium carbamate. As a consequence, large CO2 separation capacities can be achieved with small temperature swings, and regeneration energies appreciably lower than achievable with state-of-the-art aqueous amine solutions become feasible. The results provide a mechanistic framework for designing highly efficient adsorbents for removing CO2 from various gas mixtures, and yield insights into the conservation of Mg2+ within the ribulose-1,5-bisphosphate carboxylase/oxygenase family of enzymes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
International Energy Agency. CO 2 Emissions from Fuel Combustion: Highlights <http://www.iea.org/publications/freepublications/publication/CO2EmissionsFromFuelCombustionHighlights2013.pdf> (IEA, 2013)
IPCC. in Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change (eds Stocker, T. F. et al.) 11–14 (Cambridge Univ. Press, 2013)
Orr, J. C. et al. Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 437, 681–686 (2005)
Haszeldine, R. S. Carbon capture and storage: how green can black be? Science 325, 1647–1652 (2009)
Boot-Handford, M. E. et al. Carbon capture and storage update. Energy Environ. Sci. 7, 130–189 (2014)
Choi, S., Drese, J. H. & Jones, C. W. Adsorbent materials for carbon dioxide capture from large anthropogenic point sources. ChemSusChem 2, 796–854 (2009)
Lin, L. C. et al. In silico screening of carbon-capture materials. Nature Mater. 11, 633–641 (2012)
Sumida, K. et al. Carbon dioxide capture in metal-organic frameworks. Chem. Rev. 112, 724–781 (2012)
Zhou, H. C., Long, J. R. & Yaghi, O. M. Introduction to metal-organic frameworks. Chem. Rev. 112, 673–674 (2012)
Furukawa, H., Cordova, K. E., O’Keeffe, M. & Yaghi, O. M. The chemistry and applications of metal-organic frameworks. Science 341, 123044 (2013)
Demessence, A., D’Alessandro, D. M., Foo, M. L. & Long, J. R. Strong CO2 binding in a water stable triazolate-bridged metal-organic framework functionalized with ethylenediamine. J. Am. Chem. Soc. 131, 8784–8786 (2009)
McDonald, T. M., D’Alessandro, D. M., Krishna, R. & Long, J. R. Enhanced carbon dioxide capture upon incorporation of N,N′-dimethylethylenediamine in the metal-organic framework CuBTTri. Chem. Sci. 2, 2022–2028 (2011)
McDonald, T. M. et al. Capture of carbon dioxide from air and flue gas in the alkylamine-appended metal-organic framework mmen-Mg2(dobpdc). J. Am. Chem. Soc. 134, 7056–7065 (2012)
Hong, C. S. et al. Diamine-functionalized metal-organic framework: exceptionally high CO2 capacities from ambient air and flue gas, ultrafast CO2 uptake rate, and adsorption mechanism. Energy Environ. Sci. 7, 744–751 (2014)
Rosi, N. L. et al. Rod packings and metal-organic frameworks constructed from rod-shaped secondary building units. J. Am. Chem. Soc. 127, 1504–1518 (2005)
Dietzel, P. D. C., Panella, B., Hirscher, M., Blom, R. & Fjellvåg, H. Hydrogen adsorption in a nickel based coordination polymer with open metal sites in the cylindrical cavities of the desolvated framework. Chem. Commun. 959–961 (2006)
Caskey, S. R., Wong-Foy, A. G. & Matzger, A. J. Dramatic tuning of carbon dioxide uptake via metal substitution in a coordination polymer with cylindrical pores. J. Am. Chem. Soc. 130, 10870–10871 (2008)
Mason, J. A. et al. Evaluating metal-organic frameworks for post-combustion carbon dioxide capture via temperature swing adsorption. Energy Environ. Sci 4, 3030–3040 (2011)
Coelho, A. A. Whole-profile structure solution from powder diffraction powder using simulated annealing. J. Appl. Crystallogr. 33, 899–908 (2000)
Planas, N. et al. The mechanism of carbon dioxide adsorption in an alkylamine-functionalized metal-organic framework. J. Am. Chem. Soc. 135, 7402–7405 (2013)
Tiritiris, I. & Kantlehner, W. Orthoamide und Iminiumsalze, LXX [1]. Zur Fixierung von Kohlendioxid mit organischen Basen (Teil 1): Reaktionen von Diaminen mit Kohlendioxid. Z. Naturforsch. 66b, 164–176 (2011)
Drisdell, W. S. et al. Probing adsorption interactions in metal-organic frameworks using X-ray spectroscopy. J. Am. Chem. Soc. 135, 18183–18190 (2013)
Weiss, J. N. The Hill equation revisited: uses and misuses. FASEB J. 11, 835–841 (1997)
Irving, H. & Williams, R. J. P. The stability of transition-metal complexes. J. Chem. Soc. 637, 3192–3210 (1953)
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996)
Walton, K. S. et al. Understanding inflections and steps in carbon dioxide adsorption isotherms in metal-organic frameworks. J. Am. Chem. Soc. 130, 406–407 (2008)
Horike, S., Shimomura, S. & Kitagawa, S. Soft porous crystals. Nature Chem. 1, 695–704 (2009)
Serre, C. et al. Very large breathing effect in the first nanoporous chromium(III)-based solids: MIL-53 or CrIII(OH)·{O2C–C6H4–CO2}·{HO2C–C6H4–CO2H}x·{H2O}y . J. Am. Chem. Soc. 124, 13519–13526 (2002)
Henke, S. et al. Multiple phase-transitions upon selective CO2 adsorption in an alkyl ether functionalized metal-organic framework—an in situ X-ray diffraction study. CrystEngComm 13, 6399–6404 (2011)
Seo, J., Matsuda, R., Sakamoto, H., Bonneau, C. & Kitagawa, S. A pillared-layer coordination polymer with a rotable pillar acting as a molecular gate for guest molecules. J. Am. Chem. Soc. 131, 12792–12800 (2009)
Thomy, A. & Duval, X. Stepwise isotherms and phase transitions in physisorbed films. Surf. Sci. 299–300, 415–425 (1994)
Jessop, P. G., Mercer, S. M. & Heldebrant, D. J. CO2-triggered switchable solvents, surfactants, and other materials. Energy Environ. Sci. 5, 7240–7253 (2012)
Liebenthal, U. et al. Overall process analysis and optimisation for CO2 capture from coal fired power plants based on phase change solvents forming two liquid phases. Energy Procedia 37, 1844–1854 (2013)
Ma’mun, S. & Kim, I. Selection and characterization of phase-change solvent for carbon dioxide capture: precipitating system. Energy Procedia 37, 331–339 (2013)
Choi, S., Watanabe, T., Bae, T.-H., Sholl, D. S. & Jones, C. W. Modification of the Mg/DOBDC MOF with amines to enhance CO2 adsorption from ultradilute gases. J. Phys. Chem. Lett. 3, 1136–1141 (2012)
Rochelle, G. et al. Aqueous piperazine as the new standard for CO2 capture technology. Chem. Eng. J. 171, 725–733 (2011)
Sayari, A. & Blemabkhout, Y. Stabilization of amine-containing CO2 adsorbents: dramatic effect of water vapor. J. Am. Chem. Soc. 132, 6312–6314 (2010)
Nugent, P. et al. Porous materials with optimal adsorption thermodynamics and kinetics for CO2 separation. Nature 495, 80–84 (2013)
van Lare, C. E. J. Mass Transfer in Gas Fluidized Beds: Scaling, Modeling, and Particle Size Influence 141–142. PhD thesis, Tech. Univ. Eindhoven (1991) <http://alexandria.tue.nl/repository/books/348157.pdf>
Lorimer, G. The carboxylation and oxygenation of ribulose 1,5–bisphosphate: the primary events in photosynthesis and photorespiration. Annu. Rev. Plant Physiol. 32, 349–382 (1981)
Taylor, T. C. & Andersson, I. Structural transitions during activation and ligand binding in hexadecameric Rubisco inferred from the crystal structure of activated unliganded spinach enzyme. Nature Struct. Biol. 3, 95–101 (1996)
Assche, F. & Clijsters, H. Effects of metals on enzyme activity in plants. Plant Cell Environ. 13, 195–206 (1990)
Coelho, A. A. Indexing of powder diffraction patterns by iterative use of singular value decomposition. J. Appl. Crystallogr. 36, 86–95 (2003)
Coelho, A. A. TOPAS-Academic, Version 4.1 (Coelho Software, 2007)
Stephens, P. W. Phenomenological model of anisotropic peak broadening in power diffraction. J. Appl. Crystallogr. 32, 281–289 (1999)
Drisdell, W. S. & Kortright, J. B. Gas cell for in situ soft X-ray transmission-absorption spectroscopy of materials. Rev. Sci. Instrum. 85, 074103 (2014)
Allen, M. P. & Tildesley, D. J. Computer Simulation of Liquids (Clarendon Press, 1987)
Parrinello, M. & Rahman, A. Crystal structure and pair potentials: a molecular-dynamics study. Phys. Rev. Lett. 45, 1196–1199 (1980)
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17978 (1994)
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999)
Kresse, G. & Furthmuller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996)
Kresse, G. & Hafner, J. Ab initio molecular-dynamics for liquid-metals. Phys. Rev. B 47, 558–561 (1993)
Lee, K., Murray, E. D., Kong, L., Lundqvist, B. I. & Langreth, D. C. Higher-accuracy van der Waals density functional. Phys. Rev. B 82, 081101 (2010)
Vanderbilt, D. Soft self-consistent pseudopotentials in a generalized eigenvalue formalism. Phys. Rev. B 41, 7892–7895 (1990)
Giannozzi, P. et al. QUANTUM ESPRESSO: a modular and open-source software project for quantum simulations of materials. J. Phys. Condens. Matter 21, 395502 (2009)
Shirley, E. L. Optimal basis sets for detailed Brillouin-zone integrations. Phys. Rev. B 54, 16464–16469 (1996)
Prendergast, D. & Louie, S. G. Bloch-state-based interpolation: an efficient generalization of the Shirley approach to interpolating electronic structure. Phys. Rev. B 80, 235126 (2009)
Taillefumier, M., Cabaret, D., Flank, A.-M. & Mauri, F. X-ray absorption near-edge structure calculations with the pseudopotentials: application to the K edge in diamond and α-quartz. Phys. Rev. B 66, 195107 (2002)
Kresse, G. & Furthmuller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996)
Zhao, Y. & Truhlar, D. G. A new local density functional for main-group thermochemistry, transition metal bonding, thermochemical kinetics, and noncovalent interactions. J. Chem. Phys. 125, 194101 (2006)
Liechtenstein, A. I., Anisimov, V. I. & Zaanen, J. Density-functional theory and strong interactions: orbital ordering in Mott–Hubbard insulators. Phys. Rev. B 52, R5467–R5470 (1995)
Wang, L., Maxisch, T. & Ceder, G. Oxidation energies of transition metal oxides within the GGA+U framework. Phys. Rev. B 73, 195107 (2006)
Acknowledgements
We thank A. S. Bhown and A. H. Berger of EPRI, H. Krutka, C. M. Brown and. K. S. Suslick for discussions, and L. Ribaud and the 11-BM staff at the Advanced Photon Source at Argonne National Laboratory for assisting with powder X-ray diffraction experiments. The work presented here pertaining to the synthesis and gas adsorption properties of metal-organic frameworks was funded by the Advanced Research Projects Agency–Energy (ARPA-E), US Department of Energy (DOE), under award numbers DE-AR0000103 and DE-AR0000402. Funding pertaining to the characterization of materials by spectroscopy and X-ray diffraction and the computational work performed by W.S.D., B.V., R.P., S.K.S., K.L., J.B.N., B.S. and J.B.K. was provided by the Center for Gas Separations Relevant to Clean Energy Technologies, an Energy Frontier Research Center funded by the DOE, Office of Science, Office of Basic Energy Sciences under award DE-SC0001015. Experiments performed in Turin were supported by grant MIUR-PRIN 2010-2011. Work at SIMAP was performed using computer resources from GENCI (CINES grant 2014-c2015097211). The computational work performed by S.O.O., A.L.D., N.P. and L.G. was supported through the Nanoporous Materials Genome Center of the DOE, Office of Basic Energy Sciences, Division of Chemical Sciences, Geosciences, and Biosciences, under award number DE-FG02-12ER16362. This research used resources of the Advanced Photon Source, a DOE Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357. Portions of this work (use of beamline 6.3.2 at the Advanced Light Source; a user project at The Molecular Foundry, facilitated by T.P., L.F.W. and D.P., and use of its computer cluster vulcan, managed by the High Performance Computing Services Group; use of the National Energy Research Scientific Computing Center) were performed at Lawrence Berkeley National Laboratory, which is supported by the Director, Office of Science, Office of Basic Energy Sciences, of the DOE under contract no. DE-AC02-05CH11231. For fellowship support, we further thank the National Science Foundation (J.A.M.), Gerald K. Branch and Arkema (E.D.B.) and the Research Council of Norway (grant 230534 to S.K.S.).
Author information
Authors and Affiliations
Contributions
T.M.McD. and J.R.L. formulated the project. T.M.McD., E.D.B. and D.G. synthesized the compounds. T.M.McD. collected and analysed the gas adsorption data. J.A.M. collected and analysed the X-ray diffraction data. X.K. collected the NMR data. X.K. and J.A.R. analysed the NMR data. T.M.McD., A.D., V.C., F.G. and S.B. collected and analysed the infrared data. W.S.D. and J.B.K. collected X-ray absorption spectroscopy data and performed analysis, with assistance from R.P., T.P., L.F.W. and D.P. S.O.O., B.V., A.L.D., R.P., S.K.S., N.P. and K.L. performed the computations and analysed the results. J.B.N., B.S. and L.G. helped with the computational analyses. T.M.McD., J.A.M. and J.R.L. wrote the paper, and all authors contributed to revising the paper.
Corresponding author
Ethics declarations
Competing interests
The authors and the University of California have filed for a patent on some of the results contained herein.
Additional information
Metrical data for the solid-state structures of mmen-Mn2(dobpdc) without and with adsorbed CO2 are available free of charge from the Cambridge Crystallographic Data Centre under reference numbers CCDC 994497 and 994498, respectively.
Extended data figures and tables
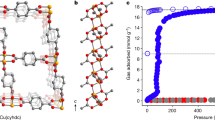
Extended Data Figure 1 Powder X-ray diffraction.
a–c, Rietveld refinement of mmen-Mn2(dobpdc) at 100 K (a), CO2-mmen-Mn2(dobpdc) at 100 K (b) and CO2-mmen-Mn2(dobpdc) at 295 K (c). The blue and red lines represent the experimental and calculated diffraction patterns, respectively; the grey line represents the difference between experimental and calculated patterns; the green tick marks represent the calculated Bragg peak positions. d, Plot of the diffraction data for CO2-mmen-Mn2(dobpdc) at 100 K (blue), where the calculated pattern (red) is based on the mmen-Mn2(dobpdc) structural model. The grey line represents the difference between the experimental and calculated patterns. Note that the intensity differences indicate a structural transition when CO2 is adsorbed. e, Fourier difference map for mmen-Mn2(dobpdc) at 100 K. Purple, brown and red spheres represent Mn, C and O atoms, respectively; yellow blobs represent excess electron density that is not accounted for in the Mn2(dobpdc) structural model and that is due to the mmen bound to each Mn2+ site. f, The coordination environment around Mg2+ in the active form of the Rubisco enzyme is structurally similar to the coordination environment around the metal cations of mmen-Mg2(dobpdc) after adsorption of CO2.
Extended Data Figure 2 Infrared spectroscopy.
a, On cooling mmen-Mg2(dobpdc) from 150 °C (red) to 30 °C (blue) under 5% CO2, changes to the aliphatic C–H vibrations of mmen are apparent on adsorption of CO2. Furthermore, ammonium formation from neutral secondary amines is indicated by the appearance of a new, broad feature centred on 3,000 cm−1. b, Room temperature, in situ infrared spectroscopy measurements of mmen-M2(dobpdc) (M = Mg, Mn, Fe, Co, Zn, Ni) under N2 (blue) and CO2 (green) atmospheres. Grey lines mark diagnostic carbamate bands at 1,690 and 1,334 cm−1. c, On cooling under flowing 5% CO2 in N2 from 150 to 30 °C, the normalized mass increase of mmen-Mg2(dobpdc) measured by thermogravimetric analysis (black line) can be compared with the normalized integrated area of the infrared active bands at 1,330 and 660 cm−1. The bands at 1,330 and 660 cm−1 can be assigned to ν(C–N) and [β(OCO) + β(NCO)] modes that are characteristic of the highly ordered ammonium carbamate chains. d, In contrast to carbamate that is coordinated to a metal site through a nitrogen atom (top), changes in electron resonance configurations give rise to a feature at 1,334 cm−1 characteristic of CO2 insertion into the metal–nitrogen bond (bottom). e, Infrared spectroscopy clearly indicates that CO2 adsorption via metal-amine insertion occurs even in the presence of H2O owing to the presence of the aforementioned band at 1,334 cm−1.
Extended Data Figure 3 X-ray absorption spectroscopy.
Experimental N K-edge NEXAFS spectra of mmen-Mg2(dobpdc) in vacuum and under increasing pressures of CO2 gas. The broad feature between 411 and 419 eV, a signature of N–C bond formation, appears before the pre-edge peak at 402.3 eV, which is characteristic of CO2 insertion.
Extended Data Figure 4 Thermodynamics of CO2 adsorption.
a, Isosteric heat of CO2 adsorption plots for the mmen-M2(dopbdc) series. b, Plot of the entropy of gaseous CO2 against pressure at 298 K. c, A linear correlation was found to exist for each mmen-M2(dobpdc) material between the step pressure at any temperature and the gas-phase entropy of CO2, for mmen-Mg2(dobpdc) (green, R2 = 0.99946), mmen-Mn2(dobpdc) (blue, R2 = 0.99918), mmen-Fe2(dobpdc) (orange, R2 = 0.99934), mmen-Co2(dobpdc) (purple, R2 = 0.99244) and mmen-Zn2(dobpdc) (black, R2 = 0.99932).
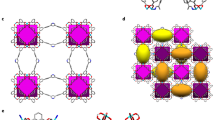
Extended Data Figure 5 Theoretical calculations.
a, Representation of the mapping of the hexagonal channel to a two-dimensional lattice in which each site consists of an amine that can interact with six neighbouring sites. Each amine can have one or zero CO2 molecules adsorbed. A single site with a CO2 adsorbed is shown in green. Pairs are allowed to form in both the crystallographic c direction and in the a–b plane (yellow); to model the chain mechanism, an amine in the middle of the chain is shown in red, and the amine at the end of the chain is in blue. b, Calculated CO2 adsorption isotherms indicate that only a chain model of interactions rather than pairwise adsorption interactions can give rise to the experimentally stepped isotherm shape. c, On the basis of calculated adsorption enthalpies, the relative positions of adsorption isotherms can be predicted from the chain model. d, e, DFT calculations reflect the experimentally observed trend that CO2 adsorption enthalpy (d) is related to the strength of the nitrogen-amine bond, as reflected by the calculated metal-amine bond length (e).
Extended Data Figure 6 Volumetric gas adsorption.
a, High-pressure excess CO2 adsorption isotherm at 25 °C for mmen-Ni2(dobpdc) indicates that Langmuir-type adsorption behaviour is maintained even at high pressures. b, Isothermal adsorption measurements of water onto a sample of mmen-Mg2(dobpdc) at 40, 75 and 100 °C. c, Four isothermal adsorption measurements of CO2 at 75 °C onto a sample of mmen-Mg2(dobpdc) before exposure to water and after water isotherms at 40, 75 and 100 °C. No changes in the CO2 adsorption isotherms were apparent from exposure of the sample to water.
Extended Data Figure 7 Dynamic gas adsorption and regeneration energy.
a, Isobaric, variable temperature (ramp rate of 1 °C min−1) gravimetric adsorption experiments for mmen-Mg2(dobpdc) under 100% CO2. Cooling from 150 to 30 °C is shown as the blue line, and heating from 30 to 150 °C as the red line. Desorption hysteresis was minimal because the phase-transition temperature and pressure are unchanged between adsorption and desorption. b, Regeneration energy calculations for mmen-Mg2(dobpdc) (green) and mmen-Mn2(dobpdc) (blue) indicate that effecting adsorption at high temperatures can be considerably more efficient than adsorption at 40 °C. c, Transient breakthrough of 15% CO2 (green), 4% O2 (red), 1.5% water (black) and balance N2 (blue) through an adsorbent bed packed with mmen-Mg2(dobpdc) at 25 °C. The adsorbent bed was under Ar (purple) before adsorption; a breakthrough CO2 capacity of 2.7 mmol g−1 was calculated.
Supplementary information
Supplementary Information
This file contains Supplementary Text, Supplementary Figures 1-8 and Supplementary Tables 1-7. (PDF 2027 kb)
Rights and permissions
About this article
Cite this article
McDonald, T., Mason, J., Kong, X. et al. Cooperative insertion of CO2 in diamine-appended metal-organic frameworks. Nature 519, 303–308 (2015). https://doi.org/10.1038/nature14327
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14327
This article is cited by
-
Engineering porous crystals to do different things
Nature Materials (2024)
-
Guidelines for the Design of Solid CO2 Adsorbents for Mobile Carbon Capture in Heavy-Duty Vehicles: A Review
Korean Journal of Chemical Engineering (2024)
-
Solvent-derived defects suppress adsorption in MOF-74
Nature Communications (2023)
-
Imidazolium-functionalized Mo3P nanoparticles with an ionomer coating for electrocatalytic reduction of CO2 to propane
Nature Energy (2023)
-
A ligand insertion mechanism for cooperative NH3 capture in metal–organic frameworks
Nature (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.