Abstract
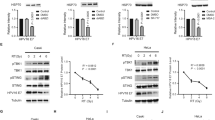
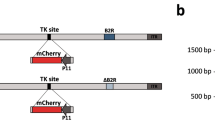
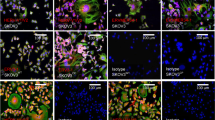
Genomes of all mammals encode apobec3 genes, which are thought to have a function in intrinsic cellular immunity to several viruses including human immunodeficiency virus type 1 (HIV-1)1. APOBEC3 (A3) proteins are packaged into virions and inhibit retroviral replication in newly infected cells, at least in part by deaminating cytidines on the negative strand DNA intermediates2. However, the role of A3 in innate resistance to mouse retroviruses is not understood. Here we show that A3 functions during retroviral infection in vivo and provides partial protection to mice against infection with mouse mammary tumour virus (MMTV). Both mouse A3 and human A3G proteins interacted with the MMTV nucleocapsid in an RNA-dependent fashion and were packaged into virions. In addition, mouse A3-containing and human A3G-containing virions showed a marked decrease in titre. Last, A3-/- mice were more susceptible to MMTV infection, because virus spread was more rapid and extensive than in their wild-type littermates.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Sheehy, A. M., Gaddis, N. C., Choi, J. D. & Malim, M. H. Isolation of a human gene that inhibits HIV-1 infection and is suppressed by the viral Vif protein. Nature 418, 646–650 (2002)
Mangeat, B. et al. Broad antiretroviral defense by human APOBEC3G through lethal editing of nascent reverse transcripts. Nature 424, 99–103 (2003)
Zheng, Y. H. et al. Human APOBEC3F is another host factor that blocks human immunodeficiency virus type 1 replication. J. Virol. 78, 6073–6076 (2004)
Holmes, R. K., Koning, F. A., Bishop, K. N. & Malim, M. H. APOBEC3F can inhibit the accumulation of HIV-1 reverse transcription products in the absence of hypermutation: Comparisons with APOBEC3G. J. Biol. Chem. advance online publication, doi:10.1074/jbc.M607298200 (22 November 2006)
Harris, R. S. et al. DNA deamination mediates innate immunity to retroviral infection. Cell 113, 803–809 (2003)
Bishop, K. N. et al. Cytidine deamination of retroviral DNA by diverse APOBEC proteins. Curr. Biol. 14, 1392–1396 (2004)
Doehle, B. P., Schafer, A., Wiegand, H. L., Bogerd, H. P. & Cullen, B. R. Differential sensitivity of murine leukemia virus to APOBEC3-mediated inhibition is governed by virion exclusion. J. Virol. 79, 8201–8207 (2005)
Kobayashi, M. et al. APOBEC3G targets specific virus species. J. Virol. 78, 8238–8244 (2004)
Esnault, C. et al. APOBEC3G cytidine deaminase inhibits retrotransposition of endogenous retroviruses. Nature 433, 430–433 (2005)
Cen, S. et al. The interaction between HIV-1 Gag and APOBEC3G. J. Biol. Chem. 279, 33177–33184 (2004)
Indik, S., Gunzburg, W. H., Salmons, B. & Rouault, F. Mouse mammary tumor virus infects human cells. Cancer Res. 65, 6651–6659 (2005)
Zhang, Y., Rassa, J. C., deObaldia, E. M., Albritton, L. & Ross, S. R. Identification of the mouse mammary tumor virus envelope receptor-binding domain. J. Virol. 77, 10468–10478 (2003)
Nandi, S. & McGrath, C. M. Mammary neoplasia in mice. Adv. Cancer Res. 17, 353–414 (1973)
Mikl, M. C. et al. Mice deficient in APOBEC2 and APOBEC3. Mol. Cell. Biol. 25, 7270–7277 (2005)
Su, A. I. et al. A gene atlas of the mouse and human protein-encoding transcriptomes. Proc. Natl Acad. Sci. USA 101, 6062–6067 (2004)
Svarovskaia, E. S. et al. Human apolipoprotein B mRNA-editing enzyme-catalytic polypeptide-like 3G (APOBEC3G) is incorporated into HIV-1 virions through interactions with viral and nonviral RNAs. J. Biol. Chem. 279, 35822–35828 (2004)
Ross, S. R. Using genetics to probe host–virus interactions: the mouse mammary tumor virus model. Microbes Infect. 2, 1215–1223 (2000)
Uz-Zaman, T., Ignatowicz, L. & Sarkar, N. H. Mouse mammary tumor viruses expressed by RIII/Sa mice with a high incidence of mammary tumors interact with the Vβ-2- and Vβ-8-specific T cells during viral infection. Virology 314, 294–304 (2003)
Chiu, Y. L. et al. Cellular APOBEC3G restricts HIV-1 infection in resting CD4+ T cells. Nature 435, 108–114 (2005)
Newman, E. N. et al. Antiviral function of APOBEC3G can be dissociated from cytidine deaminase activity. Curr. Biol. 15, 166–170 (2005)
Cullen, B. R. Role and mechanism of action of the APOBEC3 family of antiretroviral resistance factors. J. Virol. 80, 1067–1076 (2006)
Doehle, B. P. et al. The betaretrovirus Mason–Pfizer Monkey Virus selectively excludes simian APOBEC3G from virion particles. J. Virol. 80, 12102–12108 (2006)
Baillie, G. J., van de Lagemaat, L. N., Baust, C. & Mager, D. L. Multiple groups of endogenous betaretroviruses in mice, rats and other mammals. J. Virol. 78, 5784–5798 (2004)
Morris, V. L., Medeiros, E., Ringold, G. M., Bishop, J. M. & Varmus, H. E. Comparison of mouse mammary tumor virus-specific DNA in inbred, wild and Asian mice, and in tumors and normal organs from inbred mice. J. Mol. Biol. 114, 73–91 (1977)
Berkhout, B., Grigoriev, A., Bakker, M. & Lukashov, V. V. Codon and amino acid usage in retroviral genomes is consistent with virus-specific nucleotide pressure. AIDS Res. Hum. Retroviruses 18, 133–141 (2002)
Golovkina, T. V., Dudley, J. P., Jaffe, A. & Ross, S. R. Mouse mammary tumor viruses with functional superantigen genes are selected during in vivo infection. Proc. Natl Acad. Sci. USA 92, 4828–4832 (1995)
Rassa, J. C., Meyers, J. L., Zhang, Y., Kudaravalli, R. & Ross, S. R. Murine retroviruses activate B cells via interaction with Toll-like receptor 4. Proc. Natl Acad. Sci. USA 99, 2281–2286 (2002)
Golovkina, T. V. et al. A novel membrane protein is a mouse mammary tumor virus receptor. J. Virol. 72, 3066–3071 (1998)
Dzuris, J. L., Golovkina, T. V. & Ross, S. R. Both T and B cells shed infectious MMTV. J. Virol. 71, 6044–6048 (1997)
Acknowledgements
The CGRES6 cells and pGR102ES plasmid were a gift from F. Roualt and W. Günzburg. This study was supported by grants from the National Institutes of Health to S.R.R. and M.B.P. C.O. was supported by a training grant from the National Cancer Institute of the National Institutes of Health and N.L. was funded in part by the Slovenian Research Agency.
Author Contributions C.M.O, N.L., B.M.P. and S.R.R. designed the research. C.M.O. and N.L. performed the research. C.M.O, N.L., B.M.P. and S.R.R. wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures S1-S7 with legends and Supplementary Methods. (PDF 445 kb)
Rights and permissions
About this article
Cite this article
Okeoma, C., Lovsin, N., Peterlin, B. et al. APOBEC3 inhibits mouse mammary tumour virus replication in vivo. Nature 445, 927–930 (2007). https://doi.org/10.1038/nature05540
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05540
This article is cited by
-
Addressing the benefits of inhibiting APOBEC3-dependent mutagenesis in cancer
Nature Genetics (2022)
-
Molecular analysis of γ1, γ3, and α class switch recombination junctions in APOBEC3-deficient mice using high-throughput sequencing
Cellular & Molecular Immunology (2020)
-
Genomic heterogeneity in bladder cancer: challenges and possible solutions to improve outcomes
Nature Reviews Urology (2020)
-
Treatment resistance in urothelial carcinoma: an evolutionary perspective
Nature Reviews Clinical Oncology (2018)
-
Type I interferon signaling is required for the APOBEC3/Rfv3-dependent neutralizing antibody response but not innate retrovirus restriction
Retrovirology (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.