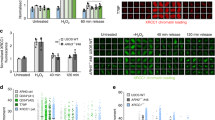
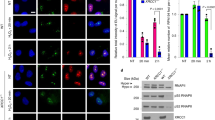
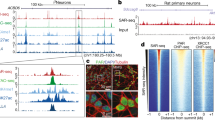
Abstract
Ataxia oculomotor apraxia-1 (AOA1) is a neurological disorder caused by mutations in the gene (APTX) encoding aprataxin1,2. Aprataxin is a member of the histidine triad (HIT) family of nucleotide hydrolases and transferases3, and inactivating mutations are largely confined to this HIT domain. Aprataxin associates with the DNA repair proteins XRCC1 and XRCC4, which are partners of DNA ligase III and ligase IV, respectively4,5,6,7, suggestive of a role in DNA repair. Consistent with this, APTX-defective cell lines are sensitive to agents that cause single-strand breaks and exhibit an increased incidence of induced chromosomal aberrations4,5,8. It is not, however, known whether aprataxin has a direct or indirect role in DNA repair, or what the physiological substrate of aprataxin might be. Here we show, using purified aprataxin protein and extracts derived from either APTX-defective chicken DT40 cells or Aptx-/- mouse primary neural cells, that aprataxin resolves abortive DNA ligation intermediates. Specifically, aprataxin catalyses the nucleophilic release of adenylate groups covalently linked to 5′-phosphate termini at single-strand nicks and gaps, resulting in the production of 5′-phosphate termini that can be efficiently rejoined. These data indicate that neurological disorders associated with APTX mutations may be caused by the gradual accumulation of unrepaired DNA strand breaks resulting from abortive DNA ligation events.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Date, H. et al. Early-onset ataxia with ocular motor apraxia and hypoalbuminemia is caused by mutations in a new HIT superfamily gene. Nature Genet. 29, 184–188 (2001)
Moreira, M. C. et al. The gene mutated in ataxia-ocular apraxia 1 encodes the new HIT/Zn-finger protein aprataxin. Nature Genet. 29, 189–193 (2001)
Brenner, C. Hint, Fhit, and GalT: Function, structure, evolution and mechanism of three branches of the histidine triad superfamily of nucleotide hydrolases and transferases. Biochemistry 41, 9003–9014 (2002)
Clements, P. M. et al. The ataxia-oculomotor apraxia 1 gene product has a role distinct from ATM and interacts with the DNA strand break repair proteins XRCC1 and XRCC4. DNA Repair (Amst.) 3, 1493–1502 (2004)
Gueven, N. et al. Aprataxin, a novel protein that protects against genotoxic stress. Hum. Mol. Genet. 13, 1081–1093 (2004)
Sano, Y. et al. Aprataxin, the causative protein for EAOH is a nuclear protein with a potential role as a DNA repair protein. Ann. Neurol. 55, 241–249 (2004)
Luo, H. et al. A new XRCC1-containing complex and its role in cellular survival of methyl methanesulfonate treatment. Mol. Cell. Biol. 24, 8356–8365 (2004)
Mosesso, P. et al. The novel human gene aprataxin is directly involved in DNA single-strand break repair. Cell. Mol. Life Sci. 62, 485–491 (2005)
Tomkinson, A. E., Vijayakumar, S., Pascal, J. M. & Ellenberger, T. DNA ligases: structure, reaction mechanism and function. Chem. Rev. 106, 687–699 (2006)
Kijas, A. W., Harris, J. L., Harris, J. M. & Lavin, M. F. Aprataxin forms a discrete branch in the HIT (histidine triad) superfamily of proteins with both DNA/RNA binding and nucleotide hydrolase activities. J. Biol. Chem. 281, 13939–13948 (2006)
Bieganowski, P. et al. Adenosine monophosphoramidase activity of Hint and Hnt1 supports function of Kin28, Ccl1, and Tfb3. J. Biol. Chem. 277, 10852–10860 (2002)
Brenner, C. et al. Crystal structures of Hint demonstrate that histidine triad proteins are GalT-related nucleotide-binding proteins. Nature Struct. Biol. 4, 231–238 (1997)
Krakowiak, A. et al. Biochemical, crystallographic, and mutagenic characterization of Hint, the AMP-lysine hydrolase, with novel substrates and inhibitors. J. Biol. Chem. 279, 18711–18716 (2004)
Barnes, L. D. et al. Fhit, a putative tumor suppressor in humans, is a dinucleoside 5′,5‴-P1,P3-triphosphate hydrolase. Biochemistry 35, 11529–11535 (1996)
Lima, C. D., Klein, M. G. & Hendrickson, W. A. Structure-based analysis of catalysis and substrate definition in the HIT protein family. Science 278, 286–290 (1997)
Draganescu, A., Hodawadekar, S. C., Gee, K. R. & Brenner, C. Fhit-nucleotide specificity probed with novel fluorescent and fluorogenic substrates. J. Biol. Chem. 275, 4555–4560 (2000)
Ghaemmaghami, S. et al. Global analysis of protein expression in yeast. Nature 425, 737–741 (2003)
Plateau, P., Fromant, M., Schmitter, J. M., Buhler, J. M. & Blanquet, S. Isolation, characterization, and inactivation of the Apa1 gene encoding yeast diadenosine 5′,5‴-P1,P4-tetraphosphate phosphorylase. J. Bacteriol. 171, 6437–6445 (1989)
Henle, E. S. & Linn, S. Formation, prevention, and repair of DNA damage by iron/hydrogen peroxide. J. Biol. Chem. 272, 19095–19098 (1997)
Pogozelski, W. K. & Tullius, T. D. Oxidative strand scission of nucleic acids: routes initiated by hydrogen abstraction from the sugar moiety. Chem. Rev. 98, 1089–1108 (1998)
Ward, J. F. in DNA Damage and Repair Vol. 2 (eds Nicoloff, J. A. & Hoekstra, M. F.) 65–85 (Humana, Totowa, New Jersey, 1998)
Loizou, J. I. et al. The protein kinase CK2 facilitates repair of chromosomal DNA single-strand breaks. Cell 117, 17–28 (2004)
Whitehouse, C. J. et al. XRCC1 stimulates human polynucleotide kinase activity at damaged DNA termini and accelerates DNA single-strand break repair. Cell 104, 107–117 (2001)
Caldecott, K. W. DNA single-strand break repair and spinocerebellar ataxia. Cell 112, 7–10 (2003)
Seidle, H. F., Bieganowski, P. & Brenner, C. Disease-associated mutations inactivate AMP-lysine hydrolase activity of Aprataxin. J. Biol. Chem. 280, 20927–20931 (2005)
Barnes, D. E. DNA damage: Air-breaks? Curr. Biol. 12, R262–R264 (2002)
Barnes, D. E., Stamp, G., Rosewell, I., Denzel, A. & Lindahl, T. Targeted disruption of the gene encoding DNA ligase IV leads to lethality in embryonic mice. Curr. Biol. 8, 1395–1398 (1998)
Gao, Y. et al. A critical role for DNA end-joining proteins in both lymphogenesis and neurogenesis. Cell 95, 891–902 (1998)
Abner, C. W. & McKinnon, P. J. The DNA double-strand break response in the nervous system. DNA Repair (Amst.) 3, 1141–1147 (2004)
El Khamisy, S. F. et al. Defective DNA single-strand break repair in spinocerebellar ataxia with axonal neuropathy-1. Nature 434, 108–113 (2005)
Acknowledgements
We thank D. Barnes and T. Lindahl for discussions and the gift of DNA ligase III–XRCC1 complex, and A. Ciccia for advice and comments. We thank S. Rulten for the gift of recombinant His-tagged aprataxin, and M. Taylor for provision of AOA1 lymphoblastoid cells. This work was supported by Cancer Research UK (S.C.W.), the EU DNA Repair Consortium (S.C.W.), the Medical Research Council (K.W.C.) and the NIH (P.J.M.). I.A. is supported by a fellowship from the European Molecular Biology Organisation. Author Contributions I.A. made the initial discovery of DNA–adenylate activity, and I.A. and U.R. were responsible for experimental design and the generation of most of the experimental data. S.F.E.-K., S.K. and P.M.C. were responsible for the development of vertebrate models and performed some of the experiments. P.J.M. and K.W.C. provided critical resources and expertise, and contributed to writing the manuscript. S.C.W. was the project leader and produced the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Methods and Supplementary Figure Legends. (DOC 74 kb)
Supplementary Figure 1
Reaction mechanism of DNA ligases. (PDF 114 kb)
Supplementary Figure 2
Purification of recombinant human Aprataxin. (PDF 257 kb)
Supplementary Figure 3
Direct measurement of AMP release from the DNA-adenylate intermediate. (PDF 209 kb)
Supplementary Figure 4
Analysis of Aprataxin on the DNA ligase-adenylate complex. (PDF 133 kb)
Supplementary Figure 5
Phylogenetic tree showing that Aprataxin (APTX) forms a distinct branch of the HIT superfamily. (PDF 111 kb)
Supplementary Figure 6
Extracts from Aptx-disrupted DT40 cells exhibit reduced ligation activity with the DNA adenylate substrate. (PDF 355 kb)
Supplementary Figure 7
Generation and characterization of Aptx-/- mice. (PDF 210 kb)
Rights and permissions
About this article
Cite this article
Ahel, I., Rass, U., El-Khamisy, S. et al. The neurodegenerative disease protein aprataxin resolves abortive DNA ligation intermediates. Nature 443, 713–716 (2006). https://doi.org/10.1038/nature05164
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05164
This article is cited by
-
The threat of programmed DNA damage to neuronal genome integrity and plasticity
Nature Genetics (2022)
-
Senescence and impaired DNA damage responses in alpha-synucleinopathy models
Experimental & Molecular Medicine (2022)
-
A label-free T4 polynucleotide kinase fluorescence sensor based on split dimeric G-quadruplex and ligation-induced dimeric G-quadruplex/thioflavin T conformation
Analytical and Bioanalytical Chemistry (2022)
-
Molecular basis for DarT ADP-ribosylation of a DNA base
Nature (2021)
-
Novel Neuroprotective Potential of Crocin in Neurodegenerative Disorders: An Illustrated Mechanistic Review
Neurochemical Research (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.