Abstract
Episodic ataxia type 6 (EA6) is caused by mutations in SLC1A3 that encodes excitatory amino acid transporter 1 (EAAT1), a glial glutamate transporter. EAAT1 regulates the extent and durations of glutamate-mediated signal by the clearance of glutamate after synaptic release. In addition, EAAT1 also has an anion channel activity that prevents additional glutamate release. We identified a missense mutation in SLC1A3 in a family with EA. The proband exhibited typical EA2-like symptoms such as recurrent ataxia, slurred speech with a duration of several hours, interictal nystagmus and response to acetazolamide, but had late-onset age of sixth decade. Whole-exome sequencing detected a heterozygous c.1177G>A mutation in SLC1A3. This mutation predicted a substitution of isoleucine for a highly conserved valine residue in the seventh transmembrane domain of EAAT1. The mutation was not present in 100 controls, a large panel of in-house genome data and various mutation databases. Most functional prediction scores revealed to be deleterious. Same heterozygous mutation was identified in one clinically affected family member and two asymptomatic members. Our data expand the mutation spectrum of SLC1A3 and the clinical phenotype of EA6.
Similar content being viewed by others
Introduction
Episodic ataxia (EA) is a clinically heterogeneous group characterized by recurrent spells of truncal ataxia and incoordination.1 There are seven subtypes defined by genetic background and clinical features. The most common subtypes are EA1 and EA2, caused by mutations in KCNA1 and CACNA1A, respectively. They have well-defined clinical features and been reported in ethnically multiple families. EA1 is characterized by brief episodes of ataxia and constant myokymia, whereas EA2 usually presents longer episodes of ataxia with interictal nystagmus.1 The onset is typically in early childhood, and episodes are commonly triggered by physical and emotional stress. In EA2, acetazolamide can reduce the frequency and severity of the attacks. Other EA subtypes have been reported in one or two families, but only two genes (CACNB4 in EA5 and SLC1A3 in EA6) have been identified. In the other three subtypes, the known EA genes were excluded (EA4) or distinct disease loci were mapped (EA3 and EA7).
EA6 is caused by mutations in SLC1A3 that encodes excitatory amino acid transporter 1 (EAAT1), a glial glutamate transporter.1, 2, 3 EAAT1 is present in the cerebellum, diencephalon and caudal brainstem.4 It is responsible for the clearance of glutamate after synaptic release, but also exhibits a sodium- and glutamate-gated anion conductance.5, 6 So far, EA6 has been reported in only two families, and their clinical phenotype was different from each other according to the glutamate reuptake capability.2, 3
We identified a missense mutation in SLC1A3 using whole-exome sequencing in a Korean family, and report their clinical characteristics.
Materials and methods
Subjects
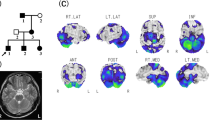
Seven members from three consecutive generations of a Korean family with EA (two clinically affected and five unaffected, Figure 1a) were enrolled. The proband was a 62-year-old man who had suffered from episodes of ataxia since 7 years back (Table 1). Initially, he experienced transient episodes of unsteadiness and dizziness lasting a few minutes. The severity and frequency of episodes increased over time, and truncal ataxia and slurred speech became more prominent lasting for several hours. The episodes were usually provoked by stress, heat or smoking. Interictal examination revealed mild truncal ataxia, hypermetric saccades and downbeat nystagmus. Brain magnetic resonance imagings were normal. He had no pathogenic mutations causing spinocerebellar ataxia 1–3, 6–8, 17. The administration of acetazolamide markedly reduced the severity and frequency of the episodes. His 36-year-old son (III-1) reported recurrent episodes of ataxia and dizziness with nausea lasting for 30 min since childhood, but the severity was milder than that of the proband. The episodes were provoked by physical and emotional stress. Interictal neurologic examinations were unremarkable. The other individuals (II-3, II-5, II-7, III-2 and III-4) did not exhibit symptoms related to episodic ataxia, and neurological examinations were normal.
(a) Pedigree of patients with SLC1A3 mutations. Solid symbols (squares=males, circles=females) indicate clinically affected individuals; open symbols, unaffected individuals; and slashed symbols, deceased individuals. The probands are indicated by arrows. (b) The chromatograms show a heterozygous c.1177G>A mutation (V393I) in exon 8 of SLC1A3. Same mutation is also identified in clinically affected family individual (III-1), and two asymptomatic family individuals (II-3, III-2). The other three asymptomatic individuals (II-5, II-7 and III-4) have no V393I mutation. A full color version of this figure is available at the Journal of Human Genetics journal online.
Molecular analysis
Genomic DNA was extracted from the patient’s blood samples. Whole-exome sequencings were performed on the proband using the Ion Torrent platform (Life Technologies, Carlsbad, CA, USA) according to the manufacturer’s specifications. To identify the pathogenic mutation responsible for EA, sequence variants were screened for presence in four known genes including KCNA1, CACNA1A, CACNB4 and SLC1A3. Nonsynonymous homozygous or heterozygous variants in coding regions and exon/intron boundaries were selected. Common variants with allele frequency of >0.01 in dbSNP were excluded. The pathogenicity of nonsynonymous variants was evaluated by MutationTaster, Likelihood Ratio Test, Polyphen2 and Sorting Intolerant From Tolerant. The suspicious variant was further confirmed by PCR-based direct sequencing.
Results
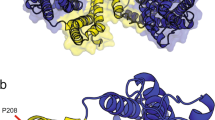
Through whole-exome sequencing on the proband, we detected 20 variants on four EA genes (Supplementary Data). After excluding variants in noncoding region and synonymous variants, we only found a heterozygous c.1177G>A mutation in exon 8 of SLC1A3 (NM_004172.4) (Figure 1b). This mutation predicted a substitution of isoleucine for a highly conserved valine residue in the seventh transmembrane domain (TMD) of EAAT1 (Figure 2). The mutation was not present in 100 Korean controls, and a large panel of in-house genome data (∼700 individuals). It was also absent in the 1000 genome projects, exome sequencing project and the other public databases (KPGP and HGMD). Most functional prediction scores revealed to be deleterious (MutationTaster: disease_causing; Likelihood Ratio Test: deleterious; Polyphen2: possibly damaging; Sorting Intolerant From Tolerant: tolerated). Same heterozygous mutation was identified in the other affected member (III-1) and two asymptomatic members (II-3 and III-2), but was absent in remaining asymptomatic members (II-5, II-7 and III-4).
(a) Schematic representation of the EAAT1 protein and localization of the mutations (the structure is adapted from Yernool et al.9). V393I mutations is located in transmembrane domain 7. (b) Conservation of the mutated residues in different species. The alignment of amino acids around p.393 reveals a highly conserved valine residue. The protein sequences were obtained from GenBank (Homo sapiens, NP_004163; Bos taurus, NP_46411; Mus musculus, NP_683740; Rattus norvegicus, NP_062098; salamander, O57321; Danio rerio, NP_997805; Drosophila melanogaster, NP_477428; human EAAT2, AY066021; human EAAT3, AB008536; human EAAT4, NP_005062; human EAAT5, NP_006662). A full color version of this figure is available at the Journal of Human Genetics journal online.
Discussion
We identified a missense mutation (c.1177G>A, V393I, NM_004172.4) of SLC1A3 in a Korean family with EA using whole-exome sequencing. This mutation was not present in 100 Korean controls, a large panel of in-house genome data and other public mutation databases. Furthermore, it is located in a highly conserved residue in the TMDs of EAAT1 among different organisms, and most functional prediction scores revealed to be deleterious. No pathogenic mutations were identified in other EA genes including KCNA1, CACNA1A and CACNB4. Thus, our SLC1A3 mutation is worthy of causative variant responsible for EA.
In the mammalian central nervous system, EAATs regulate the extent and durations of glutamate-mediated signal by the clearance of glutamate after synaptic release.5, 6 In addition, EAATs also have an anion channel activity that prevents additional glutamate release.6, 7 Thus, the mutation in EAAT1-encoding SLC1A3 may lead to excessive extracellular concentration of glutamate and neurotoxic insults. EAAT1 contain eight α-helical TMDs and re-entrant hairpin loops (HP) 1 and 2 flanking TMD7 (Figure 2a).5, 8, 9 The first six TMDs form a scaffold that surrounds a C-terminal core domain comprising HP1, TMD7, HP2 and TMD8. C-terminal domain is known to play an important role in transporting glutamate by inducing conformational rearrangements. Among them, TMD7 is critical binding site for glutamate and various coupled ions, Na+, H+ and K+.5, 10, 11 Furthermore, several residues in TMD7 contribute to anion permeation and selectivity.5, 12, 13 Hence, our V393I mutation located in TMD7 may influence glutamate binding and anion conductance. This expectation may be reasonable, considering that mutation of a nearby glycine at position 392 of GLT-1 (equivalent to reside 394 of EAAT1) inhibited glutamate transport.14
To date, three mutations of SLC1A3 have been identified in the patients with cerebellar ataxia (Table 1 and Figure 2), but one of them (R454Q mutation, NP_001160167.1) was found in a family with progressive cerebellar ataxia, and two affected members of this family showed adult-onset ataxia, dysarthria and jerky ocular pursuit.15 The other two mutations have been reported in EA patients, and clinical severity appeared to be well correlated with glutamate reuptake capability of mutant EAAT1.2, 3 The de novo P290R mutation (NP_004163.3) in sporadic case led to complete loss of glutamate reuptake and was associated with severe phenotype including episodic ataxia, seizures, migraine and alternating hemiplegia.3 On the other hand, the C186S mutation (NP_004163.3), which was identified in three symptomatic and one asymptomatic members, showed a mild reduction of glutamate reuptake and was correlated with milder clinical manifestations similar to EA2.2 Our proband also exhibited typical EA2-like symptoms including recurrent ataxia with a duration of several hours, interictal nystagmus and good response to acetazolamide, but had late-onset age of sixth decade. Although the onset of EA is typically early in life, late onset has been reported in some EA patients.16, 17, 18, 19, 20 Furthermore, the proband’s son did not complain of slurred speech and severe imbalance during an attack, and two relatives were asymptomatic, suggesting incomplete penetrance. In a family with the C186S mutation, clinical heterogeneity and reduced penetrance were also observed. Variable phenotype and incomplete penetrance can be explained by the existence of additional EAAT glutamate transporters in cerebellum. Indeed, glutamate uptake capability in homozygous GLAST knockout mice was only reduced by half.21 Nevertheless, it is still possible that truly causative mutation exists in other genes because of the presence of asymptomatic carriers in our family. We also need to accumulate other families that show late-onset EA with mutation in SLC1A3.
In conclusion, we identified a missense mutation (c.1177G>A, V393I) of SLC1A3 in a Korean EA family. Our data expand the mutation spectrum of SLC1A3 and the clinical phenotype of EA6.
References
Jen, J. C., Graves, T. D., Hess, E. J., Hanna, M. G., Griggs, R. C., Baloh, R. W. et al. Primary episodic ataxias: diagnosis, pathogenesis and treatment. Brain 130, 2484–2493 (2007).
de Vries, B., Mamsa, H., Stam, A. H., Wan, J., Bakker, S. L., Vanmolkot, K. R. et al. Episodic ataxia associated with EAAT1 mutation C186S affecting glutamate reuptake. Arch. Neurol. 66, 97–101 (2009).
Jen, J. C., Wan, J., Palos, T. P., Howard, B. D. & Baloh, R. W. Mutation in the glutamate transporter EAAT1 causes episodic ataxia, hemiplegia, and seizures. Neurology 65, 529–534 (2005).
Banner, S. J., Fray, A. E., Ince, P. G., Steward, M., Cookson, M. R. & Shaw, P. J. The expression of the glutamate re-uptake transporter excitatory amino acid transporter 1 (EAAT1) in the normal human CNS and in motor neurone disease: an immunohistochemical study. Neuroscience 109, 27–44 (2002).
Jiang, J. & Amara, S. G. New views of glutamate transporter structure and function: advances and challenges. Neuropharmacology 60, 172–181 (2011).
Fairman, W. A., Vandenberg, R. J., Arriza, J. L., Kavanaugh, M. P. & Amara, S. G. An excitatory amino-acid transporter with properties of a ligand-gated chloride channel. Nature 375, 599–603 (1995).
Winter, N., Kovermann, P. & Fahlke, C. A point mutation associated with episodic ataxia 6 increases glutamate transporter anion currents. Brain 135, 3416–3425 (2012).
Seal, R. P. & Amara, S. G. A reentrant loop domain in the glutamate carrier EAAT1 participates in substrate binding and translocation. Neuron 21, 1487–1498 (1998).
Yernool, D., Boudker, O., Jin, Y. & Gouaux, E. Structure of a glutamate transporter homologue from Pyrococcus horikoshii. Nature 431, 811–818 (2004).
Amara, S. G. & Fontana, A. C. Excitatory amino acid transporters: keeping up with glutamate. Neurochem. Int. 41, 313–318 (2002).
Kanner, B. I. & Borre, L. The dual-function glutamate transporters: structure and molecular characterisation of the substrate-binding sites. Biochem. Biophys. Acta. 1555, 92–95 (2002).
Huang, S. & Vandenberg, R. J. Mutations in transmembrane domains 5 and 7 of the human excitatory amino acid transporter 1 affect the substrate-activated anion channel. Biochemistry 46, 9685–9692 (2007).
Ryan, R. M., Mitrovic, A. D. & Vandenberg, R. J. The chloride permeation pathway of a glutamate transporter and its proximity to the glutamate translocation pathway. J. Biol. Chem. 279, 20742–20751 (2004).
Grunewald, M. & Kanner, B. I. The accessibility of a novel reentrant loop of the glutamate transporter GLT-1 is restricted by its substrate. J. Biol. Chem. 275, 9684–9689 (2000).
Pyle, A., Smertenko, T., Bargiela, D., Griffin, H., Duff, J., Appleton, M. et al. Exome sequencing in undiagnosed inherited and sporadic ataxias. Brain 138, 276–283 (2015).
Cuenca-León, E., Banchs, I., Serra, S. A., Latorre, P., Fernàndez-Castillo, N. & Corominas, R. Late-onset episodic ataxia type 2 associated with a novel loss-of-function mutation in the CACNA1A gene. J. Neurol. Sci. 280, 10–14 (2009).
Imbrici, P., Eunson, L. H., Graves, T. D., Bhatia, K. P., Wadia, N. H. & Kullmann, D. M. Late-onset episodic ataxia type 2 due to an in-frame insertion in CACNA1A. Neurology 65, 944–946 (2005).
Choi, K. D., Yook, J. W., Kim, M. J., Kim, H. S., Park, Y. E., Kim, J. S. et al. Possible anticipation associated with a novel splice site mutation in episodic ataxia type 2. Neurol. Sci. 34, 1629–1632 (2013).
Damji, K. F., Allingham, R. R., Pollock, S. C., Small, K., Lewis, K. E. & Stajich, J. M. Periodic vestibulocerebellar ataxia, an autosomal dominant ataxia with defective smooth pursuit, is genetically distinct from other autosomal dominant ataxias. Arch. Neurol. 53, 338–344 (1996).
Escayg, A., De Waard, M., Lee, D. D., Bichet, D., Wolf, P. & Mayer, T. Coding and noncoding variation of the human calcium-channel beta4-subunit gene CACNB4 in patients with idiopathic generalized epilepsy and episodic ataxia. Am. J. Hum. Genet. 66, 1531–1539 (2000).
Watase, K., Hashimoto, K., Kano, M., Yamada, K., Watanabe, M. & Inoue, Y. Motor discoordination and increased susceptibility to cerebellar injury in GLAST mutant mice. Eur. J. Neurosci. 10, 976–988 (1998).
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea funded by the Ministry of Education (NRF-2015R1D1A1A01060559).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Journal of Human Genetics website
Supplementary information
Rights and permissions
About this article
Cite this article
Choi, KD., Jen, J., Choi, S. et al. Late-onset episodic ataxia associated with SLC1A3 mutation. J Hum Genet 62, 443–446 (2017). https://doi.org/10.1038/jhg.2016.137
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhg.2016.137
This article is cited by
-
Progress in Structural Biology of Solute Carriers
Current Molecular Biology Reports (2021)
-
A novel mutation in SLC1A3 causes episodic ataxia
Journal of Human Genetics (2018)
-
Draft de novo transcriptome assembly and proteome characterization of the electric lobe of Tetronarce californica: a molecular tool for the study of cholinergic neurotransmission in the electric organ
BMC Genomics (2017)
-
Genetic Variants Associated with Episodic Ataxia in Korea
Scientific Reports (2017)
-
Structure and allosteric inhibition of excitatory amino acid transporter 1
Nature (2017)