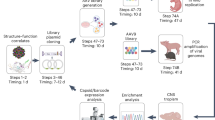
Abstract
Adeno-associated virus (AAV) vectors have achieved clinical efficacy in treating several diseases. However, enhanced vectors are required to extend these landmark successes to other indications and protein engineering approaches may provide the necessary vector improvements to address such unmet medical needs. To generate new capsid variants with potentially enhanced infectious properties and to gain insights into AAV’s evolutionary history, we computationally designed and experimentally constructed a putative ancestral AAV library. Combinatorial variations at 32 amino acid sites were introduced to account for uncertainty in their identities. We then analyzed the evolutionary flexibility of these residues, the majority of which have not been previously studied, by subjecting the library to iterative selection on a representative cell line panel. The resulting variants exhibited transduction efficiencies comparable to the most efficient extant serotypes and, in general, ancestral libraries were broadly infectious across the cell line panel, indicating that they favored promiscuity over specificity. Interestingly, putative ancestral AAVs were more thermostable than modern serotypes and did not use sialic acids, galactose or heparan sulfate proteoglycans for cellular entry. Finally, variants mediated 19- to 31-fold higher gene expression in the muscle compared with AAV1, a clinically used serotype for muscle delivery, highlighting their promise for gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Stackhouse J, Presnell SR, McGeehan GM, Nambiar KP, Benner SA . The ribonuclease from an extinct bovid ruminant. FEBS Lett 1990; 262: 104–106.
Cole MF, Gaucher EA . Utilizing natural diversity to evolve protein function: applications towards thermostability. Curr Opin Chem Biol 2011; 15: 399–406.
Gaucher EA, Govindarajan S, Ganesh OK . Palaeotemperature trend for Precambrian life inferred from resurrected proteins. Nature 2008; 451: 704–707.
Ortlund EA, Bridgham JT, Redinbo MR, Thornton JW . Crystal structure of an ancient protein: evolution by conformational epistasis. Science 2007; 317: 1544–1548.
Ugalde JA, Chang BS, Matz MV . Evolution of coral pigments recreated. Science 2004; 305: 1433.
Afriat-Jurnou L, Jackson CJ, Tawfik DS . Reconstructing a missing link in the evolution of a recently diverged phosphotriesterase by active-site loop remodeling. Biochemistry 2012; 51: 6047–6055.
Watanabe K, Ohkuri T, Yokobori S, Yamagishi A . Designing thermostable proteins: ancestral mutants of 3-isopropylmalate dehydrogenase designed by using a phylogenetic tree. J Mol Biol 2006; 355: 664–674.
Alcolombri U, Elias M, Tawfik DS . Directed evolution of sulfotransferases and paraoxonases by ancestral libraries. J Mol Biol 2011; 411: 837–853.
Chen F, Gaucher EA, Leal NA, Hutter D, Havemann SA, Govindarajan S et al. Reconstructed evolutionary adaptive paths give polymerases accepting reversible terminators for sequencing and SNP detection. Proc Natl Acad Sci USA 2010; 107: 1948–1953.
Gonzalez D, Hiblot J, Darbinian N, Miller JC, Gotthard G, Amini S et al. Ancestral mutations as a tool for solubilizing proteins: the case of a hydrophobic phosphate-binding protein. FEBS Open Bio 2014; 4: 121–127.
Kothe DL, Li Y, Decker JM, Bibollet-Ruche F, Zammit KP, Salazar MG et al. Ancestral and consensus envelope immunogens for HIV-1 subtype C. Virology 2006; 352: 438–449.
Ducatez MF, Bahl J, Griffin Y, Stigger-Rosser E, Franks J, Barman S et al. Feasibility of reconstructed ancestral H5N1 influenza viruses for cross-clade protective vaccine development. Proc Natl Acad Sci USA 2011; 108: 349–354.
Rolland M, Jensen MA, Nickle DC, Yan J, Learn GH, Heath L et al. Reconstruction and function of ancestral center-of-tree human immunodeficiency virus type 1 proteins. J Virol 2007; 81: 8507–8514.
Gullberg M, Tolf C, Jonsson N, Mulders MN, Savolainen-Kopra C, Hovi T et al. Characterization of a putative ancestor of coxsackievirus B5. J Virol 2010; 84: 9695–9708.
Berns KI, Linden RM . The cryptic life style of adeno-associated virus. BioEssays 1995; 17: 237–245.
Ellis BL, Hirsch ML, Barker JC, Connelly JP, Steininger RJ 3rd, Porteus MH . A survey of ex vivo/in vitro transduction efficiency of mammalian primary cells and cell lines with Nine natural adeno-associated virus (AAV1-9) and one engineered adeno-associated virus serotype. Virol J 2013; 10: 74.
Asokan A, Schaffer DV, Samulski RJ . The AAV vector toolkit: poised at the clinical crossroads. Mol Ther 2012; 20: 699–708.
Kotterman MA, Schaffer DV . Engineering adeno-associated viruses for clinical gene therapy. Nat Rev Genet 2014; 15: 445–451.
Gaudet D, Methot J, Dery S, Brisson D, Essiembre C, Tremblay G et al. Efficacy and long-term safety of alipogene tiparvovec (AAV1-LPLS447X) gene therapy for lipoprotein lipase deficiency: an open-label trial. Gene Therapy 2013; 20: 361–369.
Sonntag F, Schmidt K, Kleinschmidt JA . A viral assembly factor promotes AAV2 capsid formation in the nucleolus. Proc Natl Acad Sci USA 2010; 107: 10220–10225.
Khersonsky O, Rosenblat M, Toker L, Yacobson S, Hugenmatter A, Silman I et al. Directed evolution of serum paraoxonase PON3 by family shuffling and ancestor/consensus mutagenesis, and its biochemical characterization. Biochemistry 2009; 48: 6644–6654.
Benson DA, Clark K, Karsch-Mizrachi I, Lipman DJ, Ostell J, Sayers EW . GenBank. Nucleic Acids Res 2014; 42: D32–D37.
Huelsenbeck JP, Ronquist F . MRBAYES: Bayesian inference of phylogeny. Bioinformatics 2001; 17: 754–755.
Wu Z, Miller E, Agbandje-McKenna M, Samulski RJ . Alpha2,3 and alpha2,6 N-linked sialic acids facilitate efficient binding and transduction by adeno-associated virus types 1 and 6. J Virol 2006; 80: 9093–9103.
Gao GP, Alvira MR, Wang L, Calcedo R, Johnston J, Wilson JM . Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proc Natl Acad Sci USA 2002; 99: 11854–11859.
Westesson O, Barquist L, Holmes I . HandAlign: Bayesian multiple sequence alignment, phylogeny and ancestral reconstruction. Bioinformatics 2012; 28: 1170–1171.
Koerber JT, Maheshri N, Kaspar BK, Schaffer DV . Construction of diverse adeno-associated viral libraries for directed evolution of enhanced gene delivery vehicles. Nat Protoc 2006; 1: 701–706.
Bloom JD, Romero PA, Lu Z, Arnold FH . Neutral genetic drift can alter promiscuous protein functions, potentially aiding functional evolution. Biol Direct 2007; 2: 17.
Aharoni A, Gaidukov L, Khersonsky O, Mc QGS, Roodveldt C, Tawfik DS . The ‘evolvability’ of promiscuous protein functions. Nat Genet 2005; 37: 73–76.
Thornton JW . Resurrecting ancient genes: experimental analysis of extinct molecules. Nat Rev Genet 2004; 5: 366–375.
Rayaprolu V, Kruse S, Kant R, Venkatakrishnan B, Movahed N, Brooke D et al. Comparative analysis of adeno-associated virus capsid stability and dynamics. J Virol 2013; 87: 13150–13160.
Klimczak RR, Koerber JT, Dalkara D, Flannery JG, Schaffer DV . A novel adeno-associated viral variant for efficient and selective intravitreal transduction of rat Muller cells. PLoS One 2009; 4: e7467.
Hauck B, Xiao W . Characterization of tissue tropism determinants of adeno-associated virus type 1. J Virol 2003; 77: 2768–2774.
Vandenberghe LH, Breous E, Nam HJ, Gao G, Xiao R, Sandhu A et al. Naturally occurring singleton residues in AAV capsid impact vector performance and illustrate structural constraints. Gene Therapy 2009; 16: 1416–1428.
Wu Z, Asokan A, Grieger JC, Govindasamy L, Agbandje-McKenna M, Samulski RJ . Single amino acid changes can influence titer, heparin binding, and tissue tropism in different adeno-associated virus serotypes. J Virol 2006; 80: 11393–11397.
Thornton JW, Need E, Crews D . Resurrecting the ancestral steroid receptor: ancient origin of estrogen signaling. Science 2003; 301: 1714–1717.
Voordeckers K, Brown CA, Vanneste K, van der Zande E, Voet A, Maere S et al. Reconstruction of ancestral metabolic enzymes reveals molecular mechanisms underlying evolutionary innovation through gene duplication. PLoS Biol 2012; 10: e1001446.
Lanfear R, Kokko H, Eyre-Walker A . Population size and the rate of evolution. Trends Ecol Evol 2014; 29: 33–41.
Bowles DE, McPhee SW, Li C, Gray SJ, Samulski JJ, Camp AS et al. Phase 1 gene therapy for Duchenne muscular dystrophy using a translational optimized AAV vector. Mol Ther 2012; 20: 443–455.
Lochrie MA, Tatsuno GP, Christie B, McDonnell JW, Zhou S, Surosky R et al. Mutations on the external surfaces of adeno-associated virus type 2 capsids that affect transduction and neutralization. J Virol 2006; 80: 821–834.
Schmidt DM, Mundorff EC, Dojka M, Bermudez E, Ness JE, Govindarajan S et al. Evolutionary potential of (beta/alpha)8-barrels: functional promiscuity produced by single substitutions in the enolase superfamily. Biochemistry 2003; 42: 8387–8393.
Asuri P, Bartel MA, Vazin T, Jang JH, Wong TB, Schaffer DV . Directed evolution of adeno-associated virus for enhanced gene delivery and gene targeting in human pluripotent stem cells. Mol Ther 2012; 20: 329–338.
Varadarajan N, Gam J, Olsen MJ, Georgiou G, Iverson BL . Engineering of protease variants exhibiting high catalytic activity and exquisite substrate selectivity. Proc Natl Acad Sci USA 2005; 102: 6855–6860.
Zincarelli C, Soltys S, Rengo G, Rabinowitz JE . Analysis of AAV serotypes 1-9 mediated gene expression and tropism in mice after systemic injection. Mol Ther 2008; 16: 1073–1080.
Wu Z, Asokan A, Samulski RJ . Adeno-associated virus serotypes: vector toolkit for human gene therapy. Mol Ther 2006; 14: 316–327.
Schenk-Braat EA, van Mierlo MM, Wagemaker G, Bangma CH, Kaptein LC . An inventory of shedding data from clinical gene therapy trials. J Gene Med 2007; 9: 910–921.
Toromanoff A, Cherel Y, Guilbaud M, Penaud-Budloo M, Snyder RO, Haskins ME et al. Safety and efficacy of regional intravenous (r.i.) versus intramuscular (i.m.) delivery of rAAV1 and rAAV8 to nonhuman primate skeletal muscle. Mol Ther 2008; 16: 1291–1299.
Ciron C, Cressant A, Roux F, Raoul S, Cherel Y, Hantraye P et al. Human alpha-iduronidase gene transfer mediated by adeno-associated virus types 1, 2, and 5 in the brain of nonhuman primates: vector diffusion and biodistribution. Hum Gene Ther 2009; 20: 350–360.
Gray SJ, Foti SB, Schwartz JW, Bachaboina L, Taylor-Blake B, Coleman J et al. Optimizing promoters for recombinant adeno-associated virus-mediated gene expression in the peripheral and central nervous system using self-complementary vectors. Hum Gene Ther 2011; 22: 1143–1153.
Qiao C, Yuan Z, Li J, He B, Zheng H, Mayer C et al. Liver-specific microRNA-122 target sequences incorporated in AAV vectors efficiently inhibits transgene expression in the liver. Gene Therapy 2011; 18: 403–410.
Geisler A, Jungmann A, Kurreck J, Poller W, Katus HA, Vetter R et al. microRNA122-regulated transgene expression increases specificity of cardiac gene transfer upon intravenous delivery of AAV9 vectors. Gene Therapy 2011; 18: 199–209.
Bershtein S, Goldin K, Tawfik DS . Intense neutral drifts yield robust and evolvable consensus proteins. J Mol Biol 2008; 379: 1029–1044.
Tseng YS, Agbandje-McKenna M . Mapping the AAV capsid host antibody response toward the development of second generation gene delivery vectors. Front Immunol 2014; 5: 9.
Gurda BL, DiMattia MA, Miller EB, Bennett A, McKenna R, Weichert WS et al. Capsid antibodies to different adeno-associated virus serotypes bind common regions. J Virol 2013; 87: 9111–9124.
Wobus CE, Hugle-Dorr B, Girod A, Petersen G, Hallek M, Kleinschmidt JA . Monoclonal antibodies against the adeno-associated virus type 2 (AAV-2) capsid: epitope mapping and identification of capsid domains involved in AAV-2-cell interaction and neutralization of AAV-2 infection. J Virol 2000; 74: 9281–9293.
Maheshri N, Koerber JT, Kaspar BK, Schaffer DV . Directed evolution of adeno-associated virus yields enhanced gene delivery vectors. Nat Biotechnol 2006; 24: 198–204.
Gao G, Vandenberghe LH, Alvira MR, Lu Y, Calcedo R, Zhou X et al. Clades of adeno-associated viruses are widely disseminated in human tissues. J Virol 2004; 78: 6381–6388.
Takeuchi Y, Myers R, Danos O . Recombination and population mosaic of a multifunctional viral gene, adeno-associated virus cap. PLoS One 2008; 3: e1634.
Westesson O, Holmes I . Developing and applying heterogeneous phylogenetic models with XRate. PLoS One 2012; 7: e36898.
Koerber JT, Jang JH, Schaffer DV . DNA shuffling of adeno-associated virus yields functionally diverse viral progeny. Mol Ther 2008; 16: 1703–1709.
Deleyrolle LP, Harding A, Cato K, Siebzehnrubl FA, Rahman M, Azari H et al. Evidence for label-retaining tumour-initiating cells in human glioblastoma. Brain 2011; 134: 1331–1343.
Huson DH, Scornavacca C . Dendroscope 3: an interactive tool for rooted phylogenetic trees and networks. Syst Biol 2012; 61: 1061–1067.
Schrodinger, LLC. The PyMOL Molecular Graphics System, Version 1.3r1 2010.
Acknowledgements
We are grateful to Professor Brent Reynolds (University of Florida) for kindly providing the L0 human GBM tumor-initiating cells. This work was supported by the National Institutes of Health grant (R01EY022975). DSO is supported by a National Science Foundation Graduate Fellowship and JSO is supported by a National Science Foundation Graduate Fellowship and a UC Berkeley Graduate Division Fellowship. IH and OW were supported by the National Human Genome Research Institute grant (HG004483).
Author Contributions
DSO and JSO designed the project and carried out the experimental work, analyzed and interpreted the data, and wrote and edited the manuscript. OW and JRW designed the project and edited the manuscript. SYW carried out GBM cell culture and edited the manuscript. AS assisted with molecular cloning. SK edited the manuscript. IH designed the project, analyzed and interpreted the data, supervised the project and edited the manuscript. DVS designed the project, interpreted the data, supervised the project through all stages and edited the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
DVS is an inventor on patents involving AAV directed evolution.
Additional information
Supplementary Information accompanies this paper on Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Santiago-Ortiz, J., Ojala, D., Westesson, O. et al. AAV ancestral reconstruction library enables selection of broadly infectious viral variants. Gene Ther 22, 934–946 (2015). https://doi.org/10.1038/gt.2015.74
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2015.74
This article is cited by
-
Optimization of AAV vectors to target persistent viral reservoirs
Virology Journal (2021)
-
Advances in designing Adeno-associated viral vectors for development of anti-HBV gene therapeutics
Virology Journal (2021)
-
Engineering adeno-associated virus vectors for gene therapy
Nature Reviews Genetics (2020)
-
Viral Cre-LoxP tools aid genome engineering in mammalian cells
Journal of Biological Engineering (2017)
-
AAV-based dual-reporter circuit for monitoring cell signaling in living human cells
Journal of Biological Engineering (2017)