Abstract
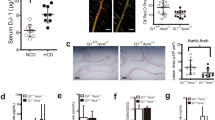
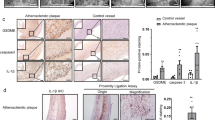
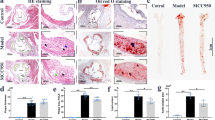
Programmed cell death 4 (Pdcd4) is a newly defined inhibitor of transcription and translation and a tumor suppressor. Recent studies have suggested that Pdcd4 may also be involved in some inflammatory diseases. However, its role in atherosclerosis, a chronic inflammation of the arterial wall, remains to be investigated. Here, we found that Pdcd4 deficiency in mice increased the expression of IL-10 in macrophages and decreased the expression of IL-17 in T cells in the presence of an atherosclerosis-associated stimulator in vitro and in high fat-induced atherosclerotic plaques. Importantly, knocking out Pdcd4 led to a decrease in atherosclerotic lesions in Apoe−/− mice fed a high fat diet. This effect could be partly reversed by blocking IL-10 with a neutralizing antibody but not by the application of exogenous IL-17. Further mechanistic studies revealed that Pdcd4 negatively regulated the expression of IL-10 in an ERK1/2- and p38-dependent manner. These results demonstrate that Pdcd4 deficiency attenuates atherosclerosis in hyperlipidemic mice in part through the upregulation of the anti-inflammatory cytokine IL-10. This indicates that endogenous Pdcd4 promotes atherosclerosis and therefore represents a potential therapeutic target for patients with atherosclerosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Yang HS, Jansen AP, Komar AA, Zheng X, Merrick WC, Costes S et al. The transformation suppressor Pdcd4 is a novel eukaryotic translation initiation factor 4A binding protein that inhibits translation. Mol Cell Biol 2003; 23: 26–37.
Shibahara K, Asano M, Ishida Y, Aoki T, Koike T, Honjo T . Isolation of a novel mouse gene MA-3 that is induced upon programmed cell death. Gene 1995; 166: 297–301.
Singh P, Wedeken L, Waters LC, Carr MD, Klempnauer KH . Pdcd4 directly binds the coding region of c-myb mRNA and suppresses its translation. Oncogene 2011; 30: 4864–4873.
Wang Q, Sun Z, Yang HS . Downregulation of tumor suppressor Pdcd4 promotes invasion and activates both beta-catenin/Tcf and AP-1-dependent transcription in colon carcinoma cells. Oncogene 2008; 27: 1527–1535.
Waters LC, Veverka V, Bohm M, Schmedt T, Choong PT, Muskett FW et al. Structure of the C-terminal MA-3 domain of the tumour suppressor protein Pdcd4 and characterization of its interaction with eIF4A. Oncogene 2007; 26: 4941–4950.
Chen Y, Knosel T, Kristiansen G, Pietas A, Garber ME, Matsuhashi S et al. Loss of PDCD4 expression in human lung cancer correlates with tumour progression and prognosis. J Pathol 2003; 200: 640–646.
Mudduluru G, Medved F, Grobholz R, Jost C, Gruber A, Leupold JH et al. Loss of programmed cell death 4 expression marks adenoma-carcinoma transition, correlates inversely with phosphorylated protein kinase B, and is an independent prognostic factor in resected colorectal cancer. Cancer 2007; 110: 1697–1707.
Gao F, Zhang P, Zhou C, Li J, Wang Q, Zhu F et al. Frequent loss of PDCD4 expression in human glioma: possible role in the tumorigenesis of glioma. Oncol Rep 2007; 17: 123–128.
Wei ZT, Zhang X, Wang XY, Gao F, Zhou CJ, Zhu FL et al. PDCD4 inhibits the malignant phenotype of ovarian cancer cells. Cancer Sci 2009; 100: 1408–1413.
Zhang X, Wang X, Song X, Liu C, Shi Y, Wang Y et al. Programmed cell death 4 enhances chemosensitivity of ovarian cancer cells by activating death receptor pathway in vitro and in vivo. Cancer Sci 2010; 101: 2163–2170.
Hilliard A, Hilliard B, Zheng SJ, Sun H, Miwa T, Song W et al. Translational regulation of autoimmune inflammation and lymphoma genesis by programmed cell death 4. J Immunol 2006; 177: 8095–8102.
Sheedy FJ, Palsson-McDermott E, Hennessy EJ, Martin C, O'Leary JJ, Ruan Q et al. Negative regulation of TLR4 via targeting of the proinflammatory tumor suppressor PDCD4 by the microRNA miR-21. Nat Immunol 2010; 11: 141–147.
Ruan Q, Wang T, Kameswaran V, Wei Q, Johnson DS, Matschinsky F et al. The microRNA-21-PDCD4 axis prevents type 1 diabetes by blocking pancreatic beta cell death. Proc Natl Acad Sci U S A 2011; 108: 12030–12035.
Wang Q, Dong Z, Liu X, Song X, Song Q, Shang Q et al. Programmed cell death-4 deficiency prevents diet-induced obesity, adipose tissue inflammation, and insulin resistance. Diabetes 2013; 62: 4132–4143.
Zhong B, Yang X, Sun Q, Liu L, Lan X, Tian J et al. Pdcd4 modulates markers of macrophage alternative activation and airway remodeling in antigen-induced pulmonary inflammation. J Leukoc Biol 2014; 96: 1065–1075.
Wang X, Zhang L, Wei Z, Zhang X, Gao Q, Ma Y et al. The inhibitory action of PDCD4 in lipopolysaccharide/D-galactosamine-induced acute liver injury. Lab Invest 2013; 93: 291–302.
Di Tullio MR, Russo C, Jin Z, Sacco RL, Mohr JP, Homma S et al. Aortic arch plaques and risk of recurrent stroke and death. Circulation 2009; 119: 2376–2382.
Hansson GK . Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med 2005; 352: 1685–1695.
Moore KJ, Tabas I . Macrophages in the pathogenesis of atherosclerosis. Cell 2011; 145: 341–355.
Tedgui A, Mallat Z . Anti-inflammatory mechanisms in the vascular wall. Circ Res 2001; 88: 877–887.
Hansson GK, Hermansson A . The immune system in atherosclerosis. Nat Immunol 2011; 12: 204–212.
Huber SA, Sakkinen P, David C, Newell MK, Tracy RP . T helper-cell phenotype regulates atherosclerosis in mice under conditions of mild hypercholesterolemia. Circulation 2001; 103: 2610–2616.
Zhou X, Nicoletti A, Elhage R, Hansson GK . Transfer of CD4(+) T cells aggravates atherosclerosis in immunodeficient apolipoprotein E knockout mice. Circulation 2000; 102: 2919–2922.
Whitman SC, Ravisankar P, Elam H, Daugherty A . Exogenous interferon-gamma enhances atherosclerosis in apolipoprotein E−/− mice. Am J Pathol 2000; 157: 1819–1824.
Gao Q, Jiang Y, Ma T, Zhu F, Gao F, Zhang P et al. A critical function of Th17 proinflammatory cells in the development of atherosclerotic plaque in mice. J Immunol 2010; 185: 5820–5827.
Smith E, Prasad KM, Butcher M, Dobrian A, Kolls JK, Ley K et al. Blockade of interleukin-17A results in reduced atherosclerosis in apolipoprotein E-deficient mice. Circulation 2010; 121: 1746–1755.
Groux H . Type 1 T-regulatory cells: their role in the control of immune responses. Transplantation 2003; 75: 8S-12S.
Halvorsen B, Waehre T, Scholz H, Clausen OP, von der Thusen JH, Muller F et al. Interleukin-10 enhances the oxidized LDL-induced foam cell formation of macrophages by antiapoptotic mechanisms. J Lipid Res 2005; 46: 211–219.
Arjuman A, Chandra NC . Effect of IL-10 on LOX-1 expression, signalling and functional activity: an atheroprotective response. Diab Vasc Dis Res 2013; 10: 442–451.
Mallat Z, Besnard S, Duriez M, Deleuze V, Emmanuel F, Bureau MF et al. Protective role of interleukin-10 in atherosclerosis. Circ Res 1999; 85: e17–e24.
Pinderski Oslund LJ, Hedrick CC, Olvera T, Hagenbaugh A, Territo M, Berliner JA et al. Interleukin-10 blocks atherosclerotic events in vitro and in vivo. Arterioscler Thromb Vasc Biol 1999; 19: 2847–2853.
Almer G, Frascione D, Pali-Scholl I, Vonach C, Lukschal A, Stremnitzer C et al. Interleukin-10: an anti-inflammatory marker to target atherosclerotic lesions via PEGylated liposomes. Mol Pharm 2013; 10: 175–186.
Caligiuri G, Rudling M, Ollivier V, Jacob MP, Michel JB, Hansson GK et al. Interleukin-10 deficiency increases atherosclerosis, thrombosis, and low-density lipoproteins in apolipoprotein E knockout mice. Mol Med 2003; 9: 10–17.
Namiki M, Kawashima S, Yamashita T, Ozaki M, Sakoda T, Inoue N et al. Intramuscular gene transfer of interleukin-10 cDNA reduces atherosclerosis in apolipoprotein E-knockout mice. Atherosclerosis 2004; 172: 21–29.
Das A, Ganesh K, Khanna S, Sen CK, Roy S . Engulfment of apoptotic cells by macrophages: a role of microRNA-21 in the resolution of wound inflammation. J Immunol 2014; 192: 1120–1129.
van den Bosch MW, Palsson-Mcdermott E, Johnson DS, O’Neill LA . LPS induces the degradation of programmed cell death protein 4 (PDCD4) to release Twist2, activating c-Maf transcription to promote interleukin-10 production. J Biol Chem 2014; 289: 22980–22990.
Chi H, Barry SP, Roth RJ, Wu JJ, Jones EA, Bennett AM et al. Dynamic regulation of pro- and anti-inflammatory cytokines by MAPK phosphatase 1 (MKP-1) in innate immune responses. Proc Natl Acad Sci U S A 2006; 103: 2274–2279.
Jansen AP, Camalier CE, Colburn NH . Epidermal expression of the translation inhibitor programmed cell death 4 suppresses tumorigenesis. Cancer Res 2005; 65: 6034–6041.
Cheng Y, Liu X, Zhang S, Lin Y, Yang J, Zhang C . MicroRNA-21 protects against the H(2)O(2)-induced injury on cardiac myocytes via its target gene PDCD4. J Mol Cell Cardiol 2009; 47: 5–14.
Cheng Y, Zhu P, Yang J, Liu X, Dong S, Wang X et al. Ischaemic preconditioning-regulated miR-21 protects heart against ischaemia/reperfusion injury via anti-apoptosis through its target PDCD4. Cardiovasc Res 2010; 87: 431–439.
Sii-Felice K, Pouponnot C, Gillet S, Lecoin L, Girault JA, Eychene A et al. MafA transcription factor is phosphorylated by p38 MAP kinase. FEBS Lett 2005; 579: 3547–3554.
Benkhelifa S, Provot S, Nabais E, Eychene A, Calothy G, Felder-Schmittbuhl MP . Phosphorylation of MafA is essential for its transcriptional and biological properties. Mol Cell Biol 2001; 21: 4441–4452.
Taleb S, Tedgui A, Mallat Z . Interleukin-17: friend or foe in atherosclerosis? Curr Opin Lipidol 2010; 21: 404–408.
Yu XH, Jiang N, Zheng XL, Cayabyab FS, Tang ZB, Tang CK . Interleukin-17A in lipid metabolism and atherosclerosis. Clin Chim Acta 2014; 431: 33–39.
Acknowledgements
We are grateful to the Key Laboratory of Cardiovascular Remodeling and Function Research, Qilu Hospital, Shandong University for expert technical assistance. This work was supported by the National 973 Basic Research Program of China (Nos. 2011CB503906, 2012CB518603), grants from the National Natural Science Foundation of China (81172863, 31470856 and 91439124) and the Natural Science Foundation of Shandong (Z2008C02), and a grant from the China Postdoctoral Science Foundation (2014 M551912).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare that there is no conflict of interest in this article.
Additional information
Supplementary information accompanies the article can be found on the Cellular & Molecular Immunology’s website (http://www.nature.com/cmi).
Supplementary information
Rights and permissions
About this article
Cite this article
Jiang, Y., Gao, Q., Wang, L. et al. Deficiency of programmed cell death 4 results in increased IL-10 expression by macrophages and thereby attenuates atherosclerosis in hyperlipidemic mice. Cell Mol Immunol 13, 524–534 (2016). https://doi.org/10.1038/cmi.2015.47
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2015.47
Keywords
This article is cited by
-
Cold exposure protects against medial arterial calcification development via autophagy
Journal of Nanobiotechnology (2023)
-
Role of programmed cell death 4 in diseases: a double-edged sword
Cellular & Molecular Immunology (2017)
-
BRD7 plays an anti-inflammatory role during early acute inflammation by inhibiting activation of the NF-кB signaling pathway
Cellular & Molecular Immunology (2017)
-
Pdcd4 restrains the self-renewal and white-to-beige transdifferentiation of adipose-derived stem cells
Cell Death & Disease (2016)
-
Pdcd4 deficiency enhances macrophage lipoautophagy and attenuates foam cell formation and atherosclerosis in mice
Cell Death & Disease (2016)