Abstract
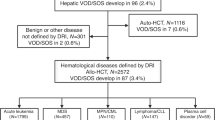
This retrospective study was conducted in Japan to determine the incidence, risk factors and outcomes of sinusoidal obstruction syndrome (SOS) after allogeneic hematopoietic stem cell transplantation (HSCT). Among 4290 patients undergoing allogeneic HSCT between 1999 and 2010, 462 were diagnosed with SOS according to the Seattle criteria (cumulative incidence, 10.8%). The cumulative incidence of SOS diagnosed by the modified Seattle criteria was 9.3%. Of 462 patients, 107 met the Baltimore criteria and 168 had severe SOS with renal and/or respiratory failure. The median onset for SOS was 12 days after HSCT (range, −2–30). Overall survival at day 100 was 32% for SOS and 15% for severe SOS. Multivariate analyses showed that significant independent risk factors for SOS were the number of HSCTs, age, performance status, hepatitis C virus-seropositivity, advanced disease status and myeloablative regimen. SOS was highly associated with overall mortality (hazard ratio, 2.09; P<0.001). Our retrospective survey showed that the cumulative incidence of SOS in Japan was 10.8%, similar to that previously reported in Western countries, and that the overall survival of patients who developed SOS was low. Furthermore, several risk factors were identified. Preventive and therapeutic strategies for high-risk SOS patients must be established to improve overall survival.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Gooley TA, Chien JW, Pergam SA, Hingorani S, Sorror ML, Boeckh M et al. Reduced mortality after allogeneic hematopoietic-cell transplantation. N Engl J Med 2010; 363: 2091–2101.
Horan JT, Logan BR, Agovi-Johnson MA, Lazarus HM, Bacigalupo AA, Ballen KK et al. Reducing the risk for transplantation-related mortality after allogeneic hematopoietic cell transplantation: how much progress has been made? J Clin Oncol 2011; 29: 805–813.
Remberger M, Ackefors M, Berglund S, Blennow O, Dahllof G, Dlugosz A et al. Improved survival after allogeneic hematopoietic stem cell transplantation in recent years. A single-center study. Biol Blood Marrow Transplant 2011; 17: 1688–1697.
Kurosawa S, Yakushijin K, Yamaguchi T, Atsuta Y, Nagamura-Inoue T, Akiyama H et al. Recent decrease in non-relapse mortality due to GVHD and infection after allogeneic hematopoietic cell transplantation in non-remission acute leukemia. Bone Marrow Transplant 2013; 48: 1198–1204.
Kurosawa S, Yakushijin K, Yamaguchi T, Atsuta Y, Nagamura-Inoue T, Akiyama H et al. Changes in incidence and causes of non-relapse mortality after allogeneic hematopoietic cell transplantation in patients with acute leukemia/myelodysplastic syndrome: an analysis of the Japan Transplant Outcome Registry. Bone Marrow Transplant 2013; 48: 529–536.
McDonald GB, Sharma P, Matthews DE, Shulman HM, Thomas ED . Venocclusive disease of the liver after bone marrow transplantation: diagnosis, incidence, and predisposing factors. Hepatology 1984; 4: 116–122.
McDonald GB, Hinds MS, Fisher LD, Schoch HG, Wolford JL, Banaji M et al. Veno-occlusive disease of the liver and multiorgan failure after bone marrow transplantation: a cohort study of 355 patients. Ann Intern Med 1993; 118: 255–267.
Jones RJ, Lee KS, Beschorner WE, Vogel VG, Grochow LB, Braine HG et al. Venoocclusive disease of the liver following bone marrow transplantation. Transplantation 1987; 44: 778–783.
Carreras E, Bertz H, Arcese W, Vernant JP, Tomas JF, Hagglund H et al. Incidence and outcome of hepatic veno-occlusive disease after blood or marrow transplantation: a prospective cohort study of the European Group for Blood and Marrow Transplantation. European Group for Blood and Marrow Transplantation Chronic Leukemia Working Party. Blood 1998; 92: 3599–3604.
DeLeve LD, Shulman HM, McDonald GB . Toxic injury to hepatic sinusoids: sinusoidal obstruction syndrome (veno-occlusive disease). Semin Liver Dis 2002; 22: 27–42.
Coppell JA, Richardson PG, Soiffer R, Martin PL, Kernan NA, Chen A et al. Hepatic veno-occlusive disease following stem cell transplantation: incidence, clinical course, and outcome. Biol Blood Marrow Transplant 2010; 16: 157–168.
Carreras E, Diaz-Beya M, Rosinol L, Martinez C, Fernandez-Aviles F, Rovira M . The incidence of veno-occlusive disease following allogeneic hematopoietic stem cell transplantation has diminished and the outcome improved over the last decade. Biol Blood Marrow Transplant 2011; 17: 1713–1720.
Corbacioglu S, Cesaro S, Faraci M, Valteau-Couanet D, Gruhn B, Rovelli A et al. Defibrotide for prophylaxis of hepatic veno-occlusive disease in paediatric haemopoietic stem-cell transplantation: an open-label, phase 3, randomised controlled trial. Lancet 2012; 379: 1301–1309.
Richardson PG, Murakami C, Jin Z, Warren D, Momtaz P, Hoppensteadt D et al. Multi-institutional use of defibrotide in 88 patients after stem cell transplantation with severe veno-occlusive disease and multisystem organ failure: response without significant toxicity in a high-risk population and factors predictive of outcome. Blood 2002; 100: 4337–4343.
Richardson PG, Soiffer RJ, Antin JH, Uno H, Jin Z, Kurtzberg J et al. Defibrotide for the treatment of severe hepatic veno-occlusive disease and multiorgan failure after stem cell transplantation: a multicenter, randomized, dose-finding trial. Biol Blood Marrow Transplant 2010; 16: 1005–1017.
Chopra R, Eaton JD, Grassi A, Potter M, Shaw B, Salat C et al. Defibrotide for the treatment of hepatic veno-occlusive disease: results of the European compassionate-use study. Br J Haematol 2000; 111: 1122–1129.
Richardson PG, Elias AD, Krishnan A, Wheeler C, Nath R, Hoppensteadt D et al. Treatment of severe veno-occlusive disease with defibrotide: compassionate use results in response without significant toxicity in a high-risk population. Blood 1998; 92: 737–744.
Mohty M, Malard F, Abecassis M, Aerts E, Alaskar AS, Aljurf M et al. Sinusoidal obstruction syndrome/veno-occlusive disease: current situation and perspectives-a position statement from the European Society for Blood and Marrow Transplantation (EBMT). Bone Marrow Transplant 2015; 50: 781–789.
Atsuta Y, Suzuki R, Yoshimi A, Gondo H, Tanaka J, Hiraoka A et al. Unification of hematopoietic stem cell transplantation registries in Japan and establishment of the TRUMP System. Int J Hematol 2007; 86: 269–274.
Giralt S, Ballen K, Rizzo D, Bacigalupo A, Horowitz M, Pasquini M et al. Reduced-intensity conditioning regimen workshop: defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2009; 15: 367–369.
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant 2009; 15: 1628–1633.
Morishima Y, Kawase T, Malkki M, Morishima S, Spellman S, Kashiwase K et al. Significance of ethnicity in the risk of acute graft-versus-host disease and leukemia relapse after unrelated donor hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2013; 19: 1197–1203.
Bearman SI . The syndrome of hepatic veno-occlusive disease after marrow transplantation. Blood 1995; 85: 3005–3020.
Lee JH, Lee KH, Kim S, Seol M, Park CJ, Chi HS et al. Plasminogen activator inhibitor-1 is an independent diagnostic marker as well as severity predictor of hepatic veno-occlusive disease after allogeneic bone marrow transplantation in adults conditioned with busulphan and cyclophosphamide. Br J Haematol 2002; 118: 1087–1094.
Heikinheimo M, Halila R, Fasth A . Serum procollagen type III is an early and sensitive marker for veno-occlusive disease of the liver in children undergoing bone marrow transplantation. Blood 1994; 83: 3036–3040.
Tabbara IA, Ghazal CD, Ghazal HH . Early drop in protein C and antithrombin III is a predictor for the development of venoocclusive disease in patients undergoing hematopoietic stem cell transplantation. J Hematother 1996; 5: 79–84.
Cesaro S, Pillon M, Talenti E, Toffolutti T, Calore E, Tridello G et al. A prospective survey on incidence, risk factors and therapy of hepatic veno-occlusive disease in children after hematopoietic stem cell transplantation. Haematologica 2005; 90: 1396–1404.
Dignan FL, Wynn RF, Hadzic N, Karani J, Quaglia A, Pagliuca A et al. BCSH/BSBMT guideline: diagnosis and management of veno-occlusive disease (sinusoidal obstruction syndrome) following haematopoietic stem cell transplantation. Br J Haematol 2013; 163: 444–457.
Nakasone H, Kurosawa S, Yakushijin K, Taniguchi S, Murata M, Ikegame K et al. Impact of hepatitis C virus infection on clinical outcome in recipients after allogeneic hematopoietic cell transplantation. Am J Hematol 2013; 88: 477–484.
Wadleigh M, Richardson PG, Zahrieh D, Lee SJ, Cutler C, Ho V et al. Prior gemtuzumab ozogamicin exposure significantly increases the risk of veno-occlusive disease in patients who undergo myeloablative allogeneic stem cell transplantation. Blood 2003; 102: 1578–1582.
McKoy JM, Angelotta C, Bennett CL, Tallman MS, Wadleigh M, Evens AM et al. Gemtuzumab ozogamicin-associated sinusoidal obstructive syndrome (SOS): an overview from the research on adverse drug events and reports (RADAR) project. Leuk Res 2007; 31: 599–604.
Yakushijin K, Matsui T, Okamura A, Yamamoto K, Ito M, Chihara K . Successful treatment with defibrotide for sinusoidal obstruction syndrome after hematopoietic stem cell transplantation. Kobe J Med Sci 2005; 51: 55–65.
Ikezoe T, Takeuchi A, Taniguchi A, Togitani K, Yokoyama A . Recombinant human soluble thrombomodulin counteracts capillary leakage associated with engraftment syndrome. Bone Marrow Transplant 2011; 46: 616–618.
Nakamura D, Yoshimitsu M, Kawada H, Inoue H, Kuroki T, Kaieda T et al. Recombinant human soluble thrombomodulin for the treatment of hepatic sinusoidal obstructive syndrome post allogeneic hematopoietic SCT. Bone Marrow Transplant 2012; 47: 463–464.
Tsubokura M, Yamashita T, Inagaki L, Kobayashi T, Kakihana K, Wakabayashi S et al. Fatal intracranial hemorrhage following administration of recombinant thrombomodulin in a patient after cord blood transplantation. Bone Marrow Transplant 2011; 46: 1030–1031.
Chao N . How I treat sinusoidal obstruction syndrome. Blood 2014; 123: 4023–4026.
Carreras E . How I manage sinusoidal obstruction syndrome after haematopoietic cell transplantation. Br J Haematol 2015; 168: 481–491.
Acknowledgements
We appreciate the contributions of all of the physicians and data managers at the centers who provided important data regarding transplantation to the JSHCT, the Japan Marrow Donor Program (JMDP) and the Japan Cord Blood Bank Network (JCBBN). We also thank all of the members of the TRUMP committees of the JSHCT, JMDP and JCBBN for their dedication and contribution to data management.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Yakushijin, K., Atsuta, Y., Doki, N. et al. Sinusoidal obstruction syndrome after allogeneic hematopoietic stem cell transplantation: Incidence, risk factors and outcomes. Bone Marrow Transplant 51, 403–409 (2016). https://doi.org/10.1038/bmt.2015.283
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2015.283
This article is cited by
-
Autotaxin is a potential predictive marker for the development of veno-occlusive disease/sinusoidal obstruction syndrome after allogeneic hematopoietic cell transplantation
Annals of Hematology (2024)
-
Potential factors for and the prognostic impact of ascites after allogeneic hematopoietic stem cell transplantation
Scientific Reports (2023)
-
HokUS-10 scoring system predicts the treatment outcome for sinusoidal obstruction syndrome after allogeneic hematopoietic stem cell transplantation
Scientific Reports (2023)
-
Rifaximin Prevents Intestinal Barrier Dysfunction and Alleviates Liver Injury in MCT-induced HSOS Mice
Current Medical Science (2023)
-
Diagnosis and severity criteria for sinusoidal obstruction syndrome/veno-occlusive disease in adult patients: a refined classification from the European society for blood and marrow transplantation (EBMT)
Bone Marrow Transplantation (2023)