Abstract
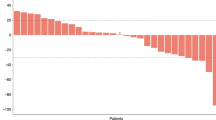
Fifty patients affected by histologically confirmed gastrointestinal tract cancer (GTC) were treated with oral tegafur (TG) 1,000 mg m-2 p.o. on days 1-14 repeated after a 14 day interval. Out of 42 evaluable patients seven patients had a partial response (PR. 17%) with a median duration of 20.5 weeks, three had a minimal response (7%) with a median duration of 23.7 weeks, nine showed a stabilisation which lasted a median of 31.3 weeks, and 23 progressed (55%). No response was obtained in patients affected by carcinoma of the pancreas and the hepatobiliary system. All PRs were achieved in patients with metastatic disease to the liver. No response was seen in patients with bone, lung or nodal metastasis. Three PRs were obtained in patients resistant to 5-fluorouracil. The difference in survival between patients who achieved PR and those who had a stabilisation was not statistically significant. On the other hand the survival of patients with PR was significantly longer than that of patients who progressed. Oral TG was well tolerated by most patients. WHO grade 1-2 gastrointestinal and neurological toxicities were seen respectively in 36% and 25% of cases. Five patients had grade 3 nausea/vomiting and one had grade 3 diarrhoea. Our data suggest that oral TG is effective in the treatment of stomach and colorectal cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Palmeri, S., Gebbia, V., Russo, A. et al. Oral tegafur in the treatment of gastrointestinal tract cancers: a phase II study. Br J Cancer 61, 475–478 (1990). https://doi.org/10.1038/bjc.1990.105
Issue Date:
DOI: https://doi.org/10.1038/bjc.1990.105
This article is cited by
-
Tegafur and 5-fluorouracil pelvic tissue concentrations in rectal cancer patients receiving preoperative chemoradiation
Clinical and Translational Oncology (2006)
-
The expression of thymidine phosphorylase correlates with angiogenesis and the efficacy of chemotherapy using fluorouracil derivatives in advanced gastric carcinoma
British Journal of Cancer (1999)
-
Chronic active hepatitis and liver cirrhosis in association with combined tamoxifen/tegafur adjuvant therapy
Digestive Diseases and Sciences (1995)
-
A case of allergic liver injury induced by tegafur
Journal of Gastroenterology (1994)