Abstract
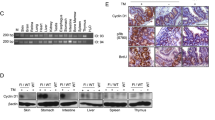
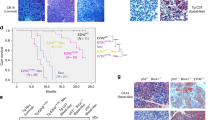
We have previously shown that increased and deregulated estrogen receptor α expression in the mammary gland leads to the development of proliferative disease and cancer. To address the importance of cyclin D1 in ERα-mediated mammary tumorigenesis, we crossed ERα-overexpressing mice with cyclin D1 knockout mice. Mammary gland morphogenesis was completely interrupted in the ERα-overexpressing cyclin D1-deficient triple transgenic mice. In addition to a highly significant reduction in mammary epithelial cell proliferation, cyclin E was upregulated resulting in DNA damage checkpoint activation and apoptosis. This imbalance between proliferative and apoptotic rates in conjunction with remarkable structural defects and cellular disorganization in the terminal end buds interrupted ductal morphogenesis. Interestingly, the structure of the mammary fat pad was fundamentally altered by the consequences of overexpressing ERα in the epithelial cells in the absence of cyclin D1 illustrating how alterations in the epithelial compartment can impact surrounding stromal composition. Transplantation of embryonic ERα-overexpressing and cyclin D1-deficient mammary epithelium into the cleared fat pad of wild-type mice did not rescue the aberrant mammary gland phenotype indicating that it was intrinsic to the mammary epithelial cells. In conclusion, although cyclin D1 is not essential for proliferation of normal mammary epithelial cells, ERα-overexpressing cells are absolutely dependent on cyclin D1 for proliferation. This differential requirement for cyclin D1 in normal vs abnormal mammary epithelial cells supports the application of cyclin D1 inhibitors as therapeutic interventions in ERα-overexpressing breast cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bartkova J, Horejsi Z, Koed K, Kramer A, Tort F, Zieger K et al. (2005). DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434: 864–870.
Carthon BC, Neumann CA, Das M, Pawlyk B, Li T, Geng Y et al. (2005). Genetic replacement of cyclin D1 function in mouse development by cyclin D2. Mol Cell Biol 25: 1081–1088.
Daniel CW, Strickland P, Friedmann Y . (1995). Expression and functional role of E- and P-cadherins in mouse mammary ductal morphogenesis and growth. Dev Biol 169: 511–519.
Deugnier MA, Teuliere J, Faraldo MM, Thiery JP, Glukhova MA . (2002). The importance of being a myoepithelial cell. Breast Cancer Res 4: 224–230.
Ekholm-Reed S, Mendez J, Tedesco D, Zetterberg A, Stillman B, Reed SI . (2004). Deregulation of cyclin E in human cells interferes with prereplication complex assembly. J Cell Biol 165: 789–800.
Fantl V, Stamp G, Andrews A, Rosewell I, Dickson C . (1995). Mice lacking cyclin D1 are small and show defects in eye and mammary gland development. Genes Dev 9: 2364–2372.
Frech MS, Halama ED, Tilli MT, Singh B, Gunther EJ, Chodosh LA et al. (2005). Deregulated estrogen receptor alpha expression in mammary epithelial cells of transgenic mice results in the development of ductal carcinoma in situ. Cancer Res 65: 681–685.
Geng Y, Whoriskey W, Park MY, Bronson RT, Medema RH, Li T et al. (1999). Rescue of cyclin D1 deficiency by knockin cyclin E. Cell 97: 767–777.
Gorgoulis VG, Vassiliou LV, Karakaidos P, Zacharatos P, Kotsinas A, Liloglou T et al. (2005). Activation of the DNA damage checkpoint and genomic instability in human precancerous lesions. Nature 434: 907–913.
Hinck L, Silberstein GB . (2005). Key stages in mammary gland development: the mammary end bud as a motile organ. Breast Cancer Res 7: 245–251.
Keyomarsi K, Tucker SL, Buchholz TA, Callister M, Ding Y, Hortobagyi GN et al. (2002). Cyclin E and survival in patients with breast cancer. N Engl J Med 347: 1566–1575.
Minella AC, Grim JE, Welcker M, Clurman BE . (2007). p53 and SCF(Fbw7) cooperatively restrain cyclin E-associated genome instability. Oncogene 48: 6948–6953.
Oh YL, Choi JS, Song SY, Ko YH, Han BK, Nam SJ et al. (2001). Expression of p21Waf1, p27Kip1 and cyclin D1 proteins in breast ductal carcinoma in situ: relation with clinicopathologic characteristics and with p53 expression and estrogen receptor status. Pathol Int 51: 94–99.
Peng SY, Chou SP, Hsu HC . (1998). Association of downregulation of cyclin D1 and of overexpression of cyclin E with p53 mutation, high tumor grade and poor prognosis in hepatocellular carcinoma. J Hepatol 29: 281–289.
Robinson GW, Accili D, Hennighausen L . (2000). Rescue of mammary epithelium of early phenotypes by embryonic mammary gland transplantation as exemplified with insulin receptor null mice. Methods in Mammary Gland Biology and Breast Cancer Research, In: Ip MM, Asch BB (eds). Kluwer Academics/Plenun Publishers: New York, pp 301–316.
Shoker BS, Jarvis C, Davies MP, Iqbal M, Sibson DR, Sloane JP . (2001). Immunodetectable cyclin D(1)is associated with oestrogen receptor but not Ki67 in normal, cancerous and precancerous breast lesions. Br J Cancer 84: 1064–1069.
Shoker BS, Jarvis C, Sibson DR, Walker C, Sloane JP . (1999). Oestrogen receptor expression in the normal and pre-cancerous breast. J Pathol 188: 237–244.
Sicinski P, Donaher JL, Parker SB, Li T, Fazeli A, Gardner H et al. (1995). Cyclin D1 provides a link between development and oncogenesis in the retina and breast. Cell 82: 621–630.
Silberstein GB . (2001). Tumour-stromal interactions. Role of the stroma in mammary development. Breast Cancer Res 3: 218–223.
Spruck CH, Won KA, Reed SI . (1999). Deregulated cyclin E induces chromosome instability. Nature 401: 297–300.
Sutherland RL, Musgrove EA . (2004). Cyclins and breast cancer. J Mammary Gland Biol Neoplasia 9: 95–104.
Wiseman BS, Werb Z . (2002). Stromal effects on mammary gland development and breast cancer. Science 296: 1046–1049.
Yu Q, Geng Y, Sicinski P . (2001). Specific protection against breast cancers by cyclin D1 ablation. Nature 411: 1017–1021.
Acknowledgements
This work was supported by grant NCI, NIH 1RO1CA112176 (PAF) and DOD Breast Cancer Research Predoctoral Award W81XWH-05-1-0302 (MSF).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Frech, M., Torre, K., Robinson, G. et al. Loss of cyclin D1 in concert with deregulated estrogen receptor α expression induces DNA damage response activation and interrupts mammary gland morphogenesis. Oncogene 27, 3186–3193 (2008). https://doi.org/10.1038/sj.onc.1210974
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210974
Keywords
This article is cited by
-
Hormone-Sensing Mammary Epithelial Progenitors: Emerging Identity and Hormonal Regulation
Journal of Mammary Gland Biology and Neoplasia (2015)