Abstract
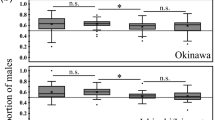
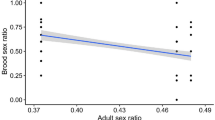
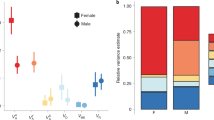
Mercurialis annua is a ruderal of pan-European distribution. In southern Spain, southern Portugal and northern Morocco, males coexist with monoecious (cosex) individuals at frequencies which vary between zero and about 30 per cent. Here, I report the results of two glasshouse experiments which aimed to characterize the mode of sex determination in one such androdioecious population. In a breeding experiment, cosexes isolated from males produced only cosexual progeny, whereas those allowed to mate with males produced both cosexual and male offspring. The results are consistent with a single-locus model of sex inheritance, with maleness determined by a dominant allele. In a density experiment, the frequency of males correlated positively with density, confirming field observations. Differential mortality or germination between the sexes was excluded by experimental design, so that the result supports the hypothesis of density-dependent gender choice. A negative correlation between density and the proportion of ‘late cosexes’ (males which turned cosexual late in their development), and the lack of any differences in the proportion of pure (unchanged) cosexes between density, suggest that only males were capable of sex change. This interpretation is consistent with the results of the breeding experiment, in which late cosexes occurred only in the offspring of cosexes allowed to mate with males. I argue that these findings help to explain the maintenance of androdioecy in M. annua.
Similar content being viewed by others
Article PDF
References
Armitage, P. 1971. Statistical Methods in Medical Research, 1st edn. Blackwell Scientific Publications, Oxford.
Aronne, G, and Wilcock, C C. 1994. Reproductive characteristics and breeding system of shrubs in the Mediterranean region. Funct Ecol, 8, 69–76.
Baker, H G. 1955. Self-compatibility and establishment after ‘long-distance’ dispersal. Evolution, 9, 347–348.
Baker, H G. 1967. Support for Baker's Law—as a rule. Evolution, 21, 853–856.
Charlesworth, D. 1984. Androdioecy and the evolution of dioecy. Biol J Linn Soc, 22, 333–348.
Charlesworth, D, and Charlesworth, B. 1978. A model for the evolution of dioecy and gynodioecy. Am Nat, 112, 975–997.
Charnov, E L. 1982. The Theory of Sex Allocation. Princeton University Press, Princeton, NJ.
Durand, B. 1963. Le complexe Mercurialis annua L. s.l.: une étude biosystematique. Ann Sci Nat Bot, 12, 579–736.
Durand, B, and Durand, R. 1991a. Male sterility and restored fertility in annual mercuries: relations with sex differentiation. Plant Sci, 80, 107–118.
Durand, B, and Durand, R. 1991b. Sex determination and reproductive organ differentiation in Mercurialis. Plant Sci, 80, 49–66.
Durand, R, and Durand, B. 1992. Dioecy, monoecy, polyploidy and speciation in annual Mercuries. Bull Soc Bot Fr Lett Bot, 139, 377–399.
Freeman, D C, and Vitale, J J. 1985. The influence of environment on the sex ratio and fitness of spinach. Bot Gaz, 146, 137–142.
Fritsch, P, and Rieseberg, L H. 1992. High outcrossing rates maintain male and hermaphrodite individuals in populations of the flowering plant Datisca glomerata. Nature, 359, 633–636.
Gouyon, P-H, and Couvet, D. 1987. A conflict between two sexes, females and hermaphrodites. In: Stearns, S. C. (ed.) The Evolution of Sex and its Consequences, pp. 245–261. Birkhäuser Verlag, Basel.
Irish, E E, and Nelson, T. 1989. Sex determination in monoecious and dioecious plants. Pl Cell, 1, 737–744.
Jarne, P, and Charlesworth, D. 1993. The evolution of the selfing rate in functionally hermaphrodite plants and animals. Ann Rev Ecol Syst, 24, 441–466.
Kohn, J R. 1989. Sex ratio, seed production, biomass allocation, and the cost of female function in Curcurbita foetidissima HBK (Curcurbitaceae). Evolution, 43, 1424–1434.
Lepart, J, and Dommée, B. 1992. Is Phillyrea angustifolia L. (Oleaceae) an androdioecious species? Bot J Linn Soc, 108, 375–387.
Liston, A, Rieseberg, L H, and Elias, T S. 1990. Functional androdioecy in the flowering plant Datisca glomerata. Nature, 343, 641–642.
Lloyd, D G. 1975. The maintenance of gynodioecy and androdioecy in angiosperms. Genetica, 45, 325–339.
Lloyd, D G, and Bawa, K S. 1984. Modification of the gender of seed plants in varying conditions. Evol Biol, 17, 255–336.
Louis, J P, Augur, C, and Teller, G. 1990. Cytokinins and differentiation processes in Mercurialis annua. Genetic regulation, regulations with auxins, indoleacetic acid oxidases, and sexual expression patterns. Pl Physiol, 94, 1535–1541.
Love, A, and Kapoor, B M. 1967. A chromosome atlas of the collective genus Rumex. Cytologia, 32, 328–342.
Meagher, T R. 1988. Sex determination in plants. In: Lovett Doust, J. and Lovett Doust, L. (eds) Plant Reproductive Ecology: Patterns and Strategies, pp. 125–138. Oxford University Press, New York.
Otto, S P, Sassaman, C, and Feldman, M W. 1993. Evolution of sex determination in the conchostracan shrimp Eulimnadia texana. Am Nat, 141, 327–337.
Pannell, J R. 1995. Models of Androdioecy and Studies on Mercurialis annua L. D.Phil. Thesis, University of Oxford.
Pannell, J R. 1997a. Variation in sex ratios and sex allocation in androdioecious Mercurialis annua. J Ecol, (in press).
Pannell, J R. 1997b. Widespread functional androdioecy in Mercurialis annua L. (Euphorbiaceae). Biol J Linn Soc, (in press).
Pannell, J R. 1997c. The maintenance of gynodioecy and androdioecy in a metapopulation. Evolution, (in press).
Reboud, X, and Zeyl, C. 1994. Organelle inheritance in plants. Heredity, 72, 132–140.
Sakai, A K, and Weller, S G. 1991. Ecological aspects of sex expression in subdioecious Schiedea globosa (Caryo-phyllaceae). Am J Bot, 78, 1280–1288.
Sassaman, C. 1989. Inbreeding and sex ratio variation in female-biased populations of a clam shrimp, Eulimnadia texana. Bull Mar Sci, 45, 425–432.
Sassaman, C. 1991. Sex ratio variation in female-biased populations of Notostracans. Hydrobiologia, 212, 169–179.
Sassaman, C, and Weeks, S C. 1993. The genetic mechanism of sex determination in the conchostracan shrimp Eulimnadia texana. Am Nat, 141, 314–328.
Traveset, A. 1994. Reproductive biology of Phillyrea angustifolia L. (Oleaceae) and effect of galling-insects on its reproductive output. Bot J Linn Soc, 114, 153–166.
Tutin, T G, Heywood, V H, Burges, N A, Moore, D M, Valentine, D H, Walters, S M, and Webb, D A. 1968. Flora Europaea, vol. 2. Cambridge University Press, Cambridge.
Valdes, B, Talavera, S, and Fernandez-Galiano, E. 1987. Flora Vascular de Andalucía Occidental, vol. 2. Ketres Editora, Barcelona.
Van Damme, J M M. 1991. A restorer gene in gynodioe-cious Plantago coronopus subject to selection in the gametophytic and seedling stage. Heredity, 66, 19–27.
Westergaard, M. 1958. The mechanism of sex determination in flowering plants. Adv Genet, 9, 217–281.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pannell, J. Mixed genetic and environmental sex determination in an androdioecious population of Mercurialis annua. Heredity 78, 50–56 (1997). https://doi.org/10.1038/hdy.1997.6
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1997.6
Keywords
This article is cited by
-
Highly sex specific gene expression in Jojoba
BMC Plant Biology (2023)
-
Two floral forms in the same species—distyly
Planta (2023)
-
The mysterious sex chromosomes of haploid plants
Heredity (2022)
-
Selection drives convergent gene expression changes during transitions to co-sexuality in haploid sexual systems
Nature Ecology & Evolution (2022)
-
Anatomy and RNA-Seq reveal important gene pathways regulating sex differentiation in a functionally Androdioecious tree, Tapiscia sinensis
BMC Plant Biology (2019)