Abstract
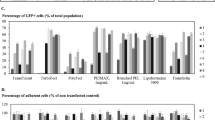
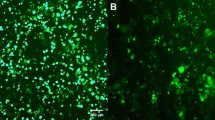
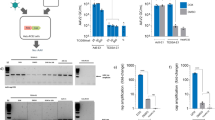
Several patients with severe combined immunodeficiency-X1 disease and adenosine deaminase deficiency have been cured by retroviral-mediated gene therapy. Despite the earlier success, the production of retroviral vectors for clinical gene therapy is cumbersome, costly and lacks safety features because of the adherent nature of packaging cells and the necessity to supplement the culture media with bovine serum. The aim of this study was to generate a retrovirus packaging cell line that could be used for the production of large clinical batch vectors. Bicistronic vectors containing an internal ribosomal entry site followed by a selection gene were used to express Moloney murine leukemia gag-pol and amphotropic envelope viral proteins in HEK293 cells. The candidate clone (293GP-A2) that was selected as the packaging cell line could release recombinant green fluorescent protein retroviruses at 4 × 107 infectious viral particles per ml. Similar titers were achieved after these cells were adapted to grow in suspension and serum-free media. Furthermore, using the same culture conditions viral titers proved to be stable for a 3-month culture period. The 293GP-A2 packaging cell line has the potential to be cultured in bioreactors, opening the possibility for large-scale use of retroviral vectors in late stage clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Cavazzana-Calvo M, Hacein-Bey S, de Saint Basile G, Gross F, Yvon E, Nusbaum P et al. Gene therapy of human severe combined immunodeficiency (SCID)-X1 disease. Science 2000; 288: 669–672.
Aiuti A, Slavin S, Aker M, Ficara F, Deola S, Mortellaro A et al. Correction of ADA-SCID by stem cell gene therapy combined with nonmyeloablative conditioning. Science 2002; 296: 2410–2413.
Gaspar HB, Parsley KL, Howe S, King D, Gilmour KC, Sinclair J et al. Gene therapy of X-linked severe combined immunodeficiency by use of a pseudotyped gammaretroviral vector. Lancet 2004; 364: 2181–2187.
Sadelain M, Riviere I, Brentjens R . Targeting tumours with genetically enhanced T lymphocytes. Nat Rev Cancer 2003; 3: 35–45.
Morgan RA, Dudley ME, Wunderlich JR, Hughes MS, Yang JC, Sherry RM et al. Cancer regression in patients after transfer of genetically engineered lymphocytes. Science 2006; 314: 126–129.
Mann R, Mulligan RC, Baltimore D . Construction of a retrovirus packaging mutant and its use to produce helper-free defective retrovirus. Cell 1983; 33: 153–159.
Miller AD, Buttimore C . Redesign of retrovirus packaging cell lines to avoid recombination leading to helper virus production. Mol Cell Biol 1986; 6: 2895–2902.
Markowitz D, Goff S, Bank A . A safe packaging line for gene transfer: separating viral genes on two different plasmids. J Virol 1988; 62: 1120–1124.
Markowitz D, Goff S, Bank A . Construction and use of a safe and efficient amphotropic packaging cell line. Virology 1988; 167: 400–406.
Danos O, Mulligan RC . Safe and efficient generation of recombinant retroviruses with amphotropic and ecotropic host ranges. Proc Natl Acad Sci USA 1988; 85: 6460–6464.
Rigg RJ, Chen J, Dando JS, Forestell SP, Plavec I, Bohnlein E . A novel human amphotropic packaging cell line: high titer, complement resistance, and improved safety. Virology 1996; 218: 290–295.
Otto E, Jones-Trower A, Vanin EF, Stambaugh K, Mueller SN, Anderson WF et al. Characterization of a replication-competent retrovirus resulting from recombination of packaging and vector sequences. Hum Gene Ther 1994; 5: 567–575.
Chong H, Vile RG . Replication-competent retrovirus produced by a ‘split-function’ third generation amphotropic packaging cell line. Gene Therapy 1996; 3: 624–629.
Cosset FL, Takeuchi Y, Battini JL, Weiss RA, Collins MK . High-titer packaging cells producing recombinant retroviruses resistant to human serum. J Virol 1995; 69: 7430–7436.
Forestell SP, Dando JS, Chen J, de Vries P, Bohnlein E, Rigg RJ . Novel retroviral packaging cell lines: complementary tropisms and improved vector production for efficient gene transfer. Gene Therapy 1997; 4: 600–610.
Sheridan PL, Bodner M, Lynn A, Phuong TK, DePolo NJ, de la Vega Jr DJ et al. Generation of retroviral packaging and producer cell lines for large-scale vector production and clinical application: improved safety and high titer. Mol Ther 2000; 2: 262–275.
Zhao Y, Low W, Collins MK . Improved safety and titre of murine leukaemia virus (MLV)-based retroviral vectors. Gene Therapy 2000; 7: 300–305.
Yu SS, Kim JM, Kim S . High efficiency retroviral vectors that contain no viral coding sequences. Gene Therapy 2000; 7: 797–804.
Tuschong L, Soenen SL, Blaese RM, Candotti F, Muul LM . Immune response to fetal calf serum by two adenosine deaminase-deficient patients after T cell gene therapy. Hum Gene Ther 2002; 13: 1605–1610.
Gerin PA, Gilligan MG, Searle PF, Al-Rubeai M . Improved titers of retroviral vectors from the human FLYRD18 packaging cell line in serum- and protein-free medium. Hum Gene Ther 1999; 10: 1965–1974.
Gerin PA, Searle PF, Al-Rubeai M . Production of retroviral vectors for gene therapy with the human packaging cell line FLYRD18. Biotechnol Prog 1999; 15: 941–948.
Glimm H, Flugge K, Mobest D, Hofmann VM, Postmus J, Henschler R et al. Efficient serum-free retroviral gene transfer into primitive human hematopoietic progenitor cells by a defined, high-titer, nonconcentrated vector-containing medium. Hum Gene Ther 1998; 9: 771–778.
McTaggart S, Al-Rubeai M . Effects of culture parameters on the production of retroviral vectors by a human packaging cell line. Biotechnol Prog 2000; 16: 859–865.
Chan LM, Coutelle C, Themis M . A novel human suspension culture packaging cell line for production of high-titre retroviral vectors. Gene Therapy 2001; 8: 697–703.
Schilz AJ, Kuhlcke K, Fauser AA, Eckert HG . Optimization of retroviral vector generation for clinical application. J Gene Med 2001; 3: 427–436.
Budak-Alpdogan T, Przybylowski M, Gonen M, Sadelain M, Bertino J, Riviere I . Functional assessment of the engraftment potential of gammaretrovirus-modified CD34+ cells, using a short serum-free transduction protocol. Hum Gene Ther 2006; 17: 780–794.
Kotani H, Newton PBR, Zhang S, Chiang YL, Otto E, Weaver L et al. Improved methods of retroviral vector transduction and production for gene therapy. Hum Gene Ther 1994; 5: 19–28.
Pan D, Whitley CB . Closed hollow-fiber bioreactor: a new approach to retroviral vector production. J Gene Med 1999; 1: 433–440.
Merten OW . State-of-the-art of the production of retroviral vectors. J Gene Med 2004; 6 (Suppl 1): S105–S124.
Przybylowski M, Hakakha A, Stefanski J, Hodges J, Sadelain M, Riviere I . Production scale-up and validation of packaging cell clearance of clinical-grade retroviral vector stocks produced in cell factories. Gene Therapy 2006; 13: 95–100.
Merten OW, Cruz PE, Rochette C, Geny-Fiamma C, Bouquet C, Goncalves D et al. Comparison of different bioreactor systems for the production of high titer retroviral vectors. Biotechnol Prog 2001; 17: 326–335.
Ghani K, Garnier A, Coelho H, Transfiguracion J, Trudel P, Kamen A . Retroviral vector production using suspension-adapted 293GPG cells in a 3L acoustic filter-based perfusion bioreactor. Biotechnol Bioeng 2006; 95: 653–660.
Pizzato M, Merten OW, Blair ED, Takeuchi Y . Development of a suspension packaging cell line for production of high titre, serum-resistant murine leukemia virus vectors. Gene Therapy 2001; 8: 737–745.
Miller AD, Garcia JV, von Suhr N, Lynch CM, Wilson C, Eiden MV . Construction and properties of retrovirus packaging cells based on gibbon ape leukemia virus. J Virol 1991; 65: 2220–2224.
Young WB, Link Jr CJ . Chimeric retroviral helper virus and picornavirus IRES sequence to eliminate DNA methylation for improved retroviral packaging cells. J Virol 2000; 74: 5242–5249.
Reeves L, Smucker P, Cornetta K . Packaging cell line characteristics and optimizing retroviral vector titer: the National Gene Vector Laboratory experience. Hum Gene Ther 2000; 11: 2093–2103.
Kelly PF, Vandergriff J, Nathwani A, Nienhuis AW, Vanin EF . Highly efficient gene transfer into cord blood nonobese diabetic/severe combined immunodeficiency repopulating cells by oncoretroviral vector particles pseudotyped with the feline endogenous retrovirus (RD114) envelope protein. Blood 2000; 96: 1206–1214.
Relander T, Brun AC, Olsson K, Pedersen L, Richter J . Overexpression of gibbon ape leukemia virus (GALV) receptor (GLVR1) on human CD34(+) cells increases gene transfer mediated by GALV pseudotyped vectors. Mol Ther 2002; 6: 400–406.
Relander T, Johansson M, Olsson K, Ikeda Y, Takeuchi Y, Collins M et al. Gene transfer to repopulating human CD34+ cells using amphotropic-, GALV-, or RD114-pseudotyped HIV-1-based vectors from stable producer cells. Mol Ther 2005; 11: 452–459.
Cote J, Garnier A, Massie B, Kamen A . Serum-free production of recombinant proteins and adenoviral vectors by 293SF-3F6 cells. Biotechnol Bioeng 1998; 59: 567–575.
Qiao J, Roy V, Girard M-H, Caruso M . High translation efficiency is mediated by the encephalomyocarditis virus IRES if the natural sequence surrounding the 11th AUG is retained. Hum Gene Ther 2002; 13: 881–887.
Acknowledgements
We are grateful to Pedro Otavio de Campos-Lima for critical reading of the manuscript and to Stephen Goff for the MLV provirus used to generate the 3T3 chronically infected cell line. This study was initiated with a grant from the Canadian Institute of Health Research (CIHR) (IC074582). MC is a Senior Research Scholar of the Fonds de la Recherche en Santé du Québec (FRSQ).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghani, K., Cottin, S., Kamen, A. et al. Generation of a high-titer packaging cell line for the production of retroviral vectors in suspension and serum-free media. Gene Ther 14, 1705–1711 (2007). https://doi.org/10.1038/sj.gt.3303039
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3303039
Keywords
This article is cited by
-
Manufacture of tumor- and virus-specific T lymphocytes for adoptive cell therapies
Cancer Gene Therapy (2015)