Abstract
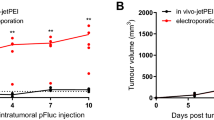
Electroporation was applied to enhance gene transfer into subcutaneous MC2 murine breast tumors. Cultured MC2 cells were also transfected by electroporation or by cationic liposomes in the presence of serum using pSV-luc plasmids. Electroporation parameters and liposome formulation were optimized to achieve the highest relative levels of transfection. An electric field threshold for successful electrotransfection in cultured cells appeared around 800–900 V/cm. The liposomes used contained the cationic lipid dioleoyl-3-trimethylammonium propane (DOTAP). Multilamellar vesicles (MLV) had a 10-fold advantage over small unilamellar vesicles (SUV) in cell culture transfection. For in vivo gene delivery, the plasmids were injected either alone, or in complex with MLV or SUV DOTAP liposomes. A series of six electric pulses 1 ms long were applied across tumors, using caliper electrodes on the skin surface. Electric field strengths ranged from 400–2300 V/cm. Luciferase expression was approximately two orders of magnitude higher than controls in tumors treated with pulses ⩾800 V/cm. Differences between enhanced relative levels of transfection using uncomplexed plasmid and lipoplexes were not statistically significant. Distribution of DNA into tumor tissues was monitored by fluorescence in situ PCR. The highest numbers of fluorescent cells were found in tumors electroporated following the injection of plasmid. The significant transfection improvement shows that in vivo electroporation is a powerful tool for local gene delivery to tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Chang BM, Chassy JA, Saunders, Sowers AE (eds) . Handbook of Electroporation and Electrofusion Academic Press: New York 1992
Prausnitz MR, Bose VG, Langer R, Weaver JC . Electroporation of mammalian skin: a mechanism to enhance transdermal drug delivery Proc Natl Acad Sci USA 1993 90: 10504–10508
Gallo SA, Oseroff AR, Johnson PG, Hui SW . Characterization of the electric pulse induced permeabilization of porcine skin using surface electrodes Biophys J 1997 72: 2805–2811
Mir LM, Orlowski S, Belehradek J, Paoletti C . Electrochemotherapy: potentiation of antitumor effect of bleomycin by electric pulses Eur J Cancer 1991 27: 68–72
Jaroszeski MJ, Gilbert RA, Heller R . In vivo antitumor effects in a hepatoma model Biochim Biophys Acta 1997 133: 15–18
Heller R, Jaroszeski MJ, Glass LF, Messina JL . Phase I/II trial for the treatment of cutaneous and subcutaneous tumors using electrochemotherapy Cancer 1996 77: 964–971
Titomirov AV, Sukharev S, Kistanova E . Electroporation and stable transformation of skin cells of newborn mice by plasmid DNA Biochim Biophys Acta 1991 1088: 131–134
Zhang L, Li L, Hoffmann GA, Hoffman RM . Depth targeted efficient gene delivery and expression in the skin by pulsed electric fields: an approach to gene therapy of skin aging and other diseases Biochem Biophys Res Com 1996 220: 633–636
Heller R, Jaroszeski MJ, Atkin A, Moradpour D . In vivo gene electroinjection and expression in rat liver FEBS Lett 1996 389: 225–228
Nishi T, Yoshizato K, Yamashiro S, Takeshima H . High efficiency in vivo gene transfer using intra-arterial plasmid DNA injection following in vivo electroporation Cancer Res 1996 56: 1050–1055
Rols MP et al. In vivo electrically mediated protein and gene transfer in murine melanoma Nat Biotech 1998 16: 168–171
Ross P, Henson ML, Supabhol R, Hui SW . Multilamellar cationic liposomes are efficient vectors for in vitro gene transfer in serum J Liposome Res 1998 8: 499–520
Li LH, Ross P, Hui SW . Improving electrotransfection efficiency by post-pulse centrifugation Gene Therapy 1999 6: 364–372
Hui SW, Langner M, Zhao YL, Ross P . The role of helper lipids in cationic liposome mediated gene transfer Biophys J 1996 71: 590–599
Wolff JA, Ludtke JJ, Acsadi G, Williams P, Jani A . Long-term persistence of plasmid DNA and foreign gene expression in mouse muscle Hum Mol Genet 1992 1: 363–369
Acknowledgements
This work is supported by a grant GM 30969 from the National Institutes of Health. We thank Dr John Yates, Department of Cancer Genetics, for the use of the luminometer, and Dorothy Donovan for her help in starting the mouse and MC2 cell colonies. Statistical analysis was performed with the help of Dr William Greco, Biostatistics Facility, which is supported by the CCSR grant CA16056 from the National Cancer Institute.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wells, J., Li, L., Sen, A. et al. Electroporation-enhanced gene delivery in mammary tumors. Gene Ther 7, 541–547 (2000). https://doi.org/10.1038/sj.gt.3301141
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301141
Keywords
This article is cited by
-
Cell uptake mechanisms of PAMAM G4-FITC dendrimer in human myometrial cells
Journal of Nanoparticle Research (2013)
-
Cell-Specific Targeting Strategies for Electroporation-Mediated Gene Delivery in Cells and Animals
The Journal of Membrane Biology (2013)
-
Physical Non-Viral Gene Delivery Methods for Tissue Engineering
Annals of Biomedical Engineering (2013)
-
Enhancement of electric field-mediated gene delivery through pretreatment of tumors with a hyperosmotic mannitol solution
Cancer Gene Therapy (2011)
-
The effect of the histological properties of tumors on transfection efficiency of electrically assisted gene delivery to solid tumors in mice
Gene Therapy (2007)