Abstract
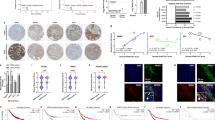
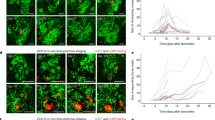
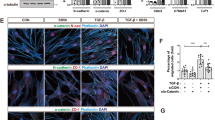
Patient data suggest that colony-stimulating factor-1 (CSF1) and its receptor (CSF1R) have critical roles during breast cancer progression. We have previously shown that in human breast tumors expressing both CSF1 and CSF1R, invasion in vivo is dependent both on a paracrine interaction with tumor-associated macrophages and an autocrine regulation of CSF1R in the tumor cells themselves. Although the role of the paracrine interaction between tumor cells and macrophages has been extensively studied, very little is known about the mechanism by which the autocrine CSF1R signaling contributes to tumor progression. We show here that breast cancer patients of the claudin-low subtype have significantly increased expression of CSF1R. Using a panel of breast cancer cell lines, we confirm that CSF1R expression is elevated and regulated by TGFβ specifically in claudin-low cell lines. Abrogation of autocrine CSF1R signaling in MDA-MB-231 xenografts (a claudin-low cell line) leads to increased tumor size by enhanced proliferation, but significantly reduced invasion, dissemination and metastasis. Indeed, we show that proliferation and invasion are oppositely regulated by CSF1R downstream of TGFβ only in claudin-low cell lines. Intravital multiphoton imaging revealed that inhibition of CSF1R in the tumor cells leads to decreased in vivo motility and a more cohesive morphology. We show that, both in vitro and in vivo, CSF1R inhibition results in a reversal of claudin-low marker expression by significant upregulation of luminal keratins and tight-junction proteins such as claudins. Finally, we show that artificial overexpression of claudins in MDA-MB-231 cells is sufficient to tip the cells from an invasive state to a proliferative state. Our results suggest that autocrine CSF1R signaling is essential in maintaining low claudin expression and that it mediates a switch between the proliferative and the invasive state in claudin-low tumor cells downstream of TGFβ.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA et al. Molecular portraits of human breast tumours. Nature 2000; 406: 747–752.
Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 2001; 98: 10869–10874.
Sorlie T, Tibshirani R, Parker J, Hastie T, Marron JS, Nobel A et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci USA 2003; 100: 8418–8423.
Sotiriou C, Neo SY, McShane LM, Korn EL, Long PM, Jazaeri A et al. Breast cancer classification and prognosis based on gene expression profiles from a population-based study. Proc Natl Acad Sci USA 2003; 100: 10393–10398.
Hu Z, Fan C, Oh DS, Marron JS, He X, Qaqish BF et al. The molecular portraits of breast tumors are conserved across microarray platforms. BMC Genomics 2006; 7: 96.
Herschkowitz JI, Simin K, Weigman VJ, Mikaelian I, Usary J, Hu Z et al. Identification of conserved gene expression features between murine mammary carcinoma models and human breast tumors. Genome Biol 2007; 8: R76.
Prat A, Parker JS, Karginova O, Fan C, Livasy C, Herschkowitz JI et al. Phenotypic and molecular characterization of the claudin-low intrinsic subtype of breast cancer. Breast Cancer Res 2010; 12: R68.
Sotiriou C, Pusztai L . Gene-expression signatures in breast cancer. N Engl J Med 2009; 360: 790–800.
Weigelt B, Pusztai L, Ashworth A, Reis-Filho JS . Challenges translating breast cancer gene signatures into the clinic. Nat Rev Clin Oncol 2012; 9: 58–64.
Kacinski BM, Carter D, Mittal K, Kohorn EI, Bloodgood RS, Donahue J et al. High level expression of fms proto-oncogene mRNA is observed in clinically aggressive human endometrial adenocarcinomas. Int J Radiat Oncol Biol Phys 1988; 15: 823–829.
Kacinski BM, Carter D, Mittal K, Yee LD, Scata KA, Donofrio L et al. Ovarian adenocarcinomas express fms-complementary transcripts and fms antigen, often with coexpression of CSF-1. Am J Pathol 1990; 137: 135–147.
Kacinski BM, Scata KA, Carter D, Yee LD, Sapi E, King BL et al. FMS (CSF-1 receptor) and CSF-1 transcripts and protein are expressed by human breast carcinomas in vivo and in vitro. Oncogene 1991; 6: 941–952.
Scholl SM, Lidereau R, de la Rochefordiere A, Le-Nir CC, Mosseri V, Nogues C et al. Circulating levels of the macrophage colony stimulating factor CSF-1 in primary and metastatic breast cancer patients. A pilot study. Breast Cancer Res Treat 1996; 39: 275–283.
Kacinski BM, Stanley ER, Carter D, Chambers JT, Chambers SK, Kohorn EI et al. Circulating levels of CSF-1 (M-CSF) a lymphohematopoietic cytokine may be a useful marker of disease status in patients with malignant ovarian neoplasms. Int J Radiat Oncol Biol Phys 1989; 17: 159–164.
Scholl SM, Bascou CH, Mosseri V, Olivares R, Magdelenat H, Dorval T et al. Circulating levels of colony-stimulating factor 1 as a prognostic indicator in 82 patients with epithelial ovarian cancer. Br J Cancer 1994; 69: 342–346.
Wyckoff J, Wang W, Lin EY, Wang Y, Pixley F, Stanley ER et al. A paracrine loop between tumor cells and macrophages is required for tumor cell migration in mammary tumors. Cancer Res 2004; 64: 7022–7029.
Roussos ET, Balsamo M, Alford SK, Wyckoff JB, Gligorijevic B, Wang Y et al. Mena invasive (MenaINV) promotes multicellular streaming motility and transendothelial migration in a mouse model of breast cancer. J Cell Sci 2011; 124: 2120–2131.
Patsialou A, Bravo-Cordero JJ, Wang Y, Entenberg D, Liu H, Clarke M et al. Intravital multiphoton imaging reveals multicellular streaming as a crucial component of in vivo cell migration in human breast tumors. Intravital 2013; 2: e25294.
Lin EY, Nguyen AV, Russell RG, Pollard JW . Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy. J Exp Med 2001; 193: 727–740.
Aharinejad S, Paulus P, Sioud M, Hofmann M, Zins K, Schafer R et al. Colony-stimulating factor-1 blockade by antisense oligonucleotides and small interfering RNAs suppresses growth of human mammary tumor xenografts in mice. Cancer Res 2004; 64: 5378–5384.
Paulus P, Stanley ER, Schafer R, Abraham D, Aharinejad S . Colony-stimulating factor-1 antibody reverses chemoresistance in human MCF-7 breast cancer xenografts. Cancer Res 2006; 66: 4349–4356.
DeNardo DG, Brennan DJ, Rexhepaj E, Ruffell B, Shiao SL, Madden SF et al. Leukocyte complexity predicts breast cancer survival and functionally regulates response to chemotherapy. Cancer Discov 2011; 1: 54–67.
Kacinski BM . CSF-1 and its receptor in breast carcinomas and neoplasms of the female reproductive tract. Mol Reprod Dev 1997; 46: 71–74.
Filderman AE, Bruckner A, Kacinski BM, Deng N, Remold HG . Macrophage colony-stimulating factor (CSF-1) enhances invasiveness in CSF-1 receptor-positive carcinoma cell lines. Cancer Res 1992; 52: 3661–3666.
Sapi E, Flick MB, Rodov S, Gilmore-Hebert M, Kelley M, Rockwell S et al. Independent regulation of invasion and anchorage-independent growth by different autophosphorylation sites of the macrophage colony-stimulating factor 1 receptor. Cancer Res 1996; 56: 5704–5712.
Wrobel CN, Debnath J, Lin E, Beausoleil S, Roussel MF, Brugge JS . Autocrine CSF-1R activation promotes Src-dependent disruption of mammary epithelial architecture. J Cell Biol 2004; 165: 263–273.
Patsialou A, Wyckoff J, Wang Y, Goswami S, Stanley ER, Condeelis JS . Invasion of human breast cancer cells in vivo requires both paracrine and autocrine loops involving the colony-stimulating factor-1 receptor. Cancer Res 2009; 69: 9498–9506.
Taube JH, Herschkowitz JI, Komurov K, Zhou AY, Gupta S, Yang J et al. Core epithelial-to-mesenchymal transition interactome gene-expression signature is associated with claudin-low and metaplastic breast cancer subtypes. Proc Natl Acad Sci USA 2010; 107: 15449–15454.
Arteaga CL, Tandon AK, Von Hoff DD, Osborne CK . Transforming growth factor beta: potential autocrine growth inhibitor of estrogen receptor-negative human breast cancer cells. Cancer Res 1988; 48: 3898–3904.
Zugmaier G, Ennis BW, Deschauer B, Katz D, Knabbe C, Wilding G et al. Transforming growth factors type beta 1 and beta 2 are equipotent growth inhibitors of human breast cancer cell lines. J Cell Physiol 1989; 141: 353–361.
Morandi A, Barbetti V, Riverso M, Dello Sbarba P, Rovida E . The colony-stimulating factor-1 (CSF-1) receptor sustains ERK1/2 activation and proliferation in breast cancer cell lines. PLoS One 2011; 6: e27450.
Qian BZ, Pollard JW . Macrophage diversity enhances tumor progression and metastasis. Cell 2010; 141: 39–51.
Farina AR, Coppa A, Tiberio A, Tacconelli A, Turco A, Colletta G et al. Transforming growth factor-beta1 enhances the invasiveness of human MDA-MB-231 breast cancer cells by up-regulating urokinase activity. Int J Cancer 1998; 75: 721–730.
Cross M, Nguyen T, Bogdanoska V, Reynolds E, Hamilton JA . A proteomics strategy for the enrichment of receptor-associated complexes. Proteomics 2005; 5: 4754–4763.
Ikenouchi J, Matsuda M, Furuse M, Tsukita S . Regulation of tight junctions during the epithelium-mesenchyme transition: direct repression of the gene expression of claudins/occludin by Snail. J Cell Sci 2003; 116: 1959–1967.
Martinez-Estrada OM, Culleres A, Soriano FX, Peinado H, Bolos V, Martinez FO et al. The transcription factors Slug and Snail act as repressors of Claudin-1 expression in epithelial cells. Biochem J 2006; 394: 449–457.
Aigner K, Dampier B, Descovich L, Mikula M, Sultan A, Schreiber M et al. The transcription factor ZEB1 (deltaEF1) promotes tumour cell dedifferentiation by repressing master regulators of epithelial polarity. Oncogene 2007; 26: 6979–6988.
Myal Y, Leygue E, Blanchard AA . Claudin 1 in breast tumorigenesis: revelation of a possible novel ‘claudin high’ subset of breast cancers. J Biomed Biotechnol 2010; 2010: 956897.
Lanigan F, McKiernan E, Brennan DJ, Hegarty S, Millikan RC, McBryan J et al. Increased claudin-4 expression is associated with poor prognosis and high tumour grade in breast cancer. Int J Cancer 2009; 124: 2088–2097.
Resnick MB, Gavilanez M, Newton E, Konkin T, Bhattacharya B, Britt DE et al. Claudin expression in gastric adenocarcinomas: a tissue microarray study with prognostic correlation. Hum Pathol 2005; 36: 886–892.
Dhawan P, Singh AB, Deane NG, No Y, Shiou SR, Schmidt C et al. Claudin-1 regulates cellular transformation and metastatic behavior in colon cancer. J Clin Invest 2005; 115: 1765–1776.
Agarwal R, Mori Y, Cheng Y, Jin Z, Olaru AV, Hamilton JP et al. Silencing of claudin-11 is associated with increased invasiveness of gastric cancer cells. PLoS One 2009; 4: e8002.
Condeelis J, Pollard JW . Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 2006; 124: 263–266.
Pollard JW . Macrophages define the invasive microenvironment in breast cancer. J Leukoc Biol 2008; 84: 623–630.
Ruffell B, Affara NI, Coussens LM . Differential macrophage programming in the tumor microenvironment. Trends Immunol 2012; 33: 119–126.
Sudo T, Nishikawa S, Ogawa M, Kataoka H, Ohno N, Izawa A et al. Functional hierarchy of c-kit and c-fms in intramarrow production of CFU-M. Oncogene 1995; 11: 2469–2476.
Gil-Henn H, Patsialou A, Wang Y, Warren MS, Condeelis JS, Koleske AJ . Arg/Abl2 promotes invasion and attenuates proliferation of breast cancer in vivo. Oncogene 2013; 32: 2622–2630.
Patsialou A, Wang Y, Lin J, Whitney K, Goswami S, Kenny PA et al. Selective gene-expression profiling of migratory tumor cells in vivo predicts clinical outcome in breast cancer patients. Breast Cancer Res 2012; 14: R139.
Wyckoff JB, Segall JE, Condeelis JS . The collection of the motile population of cells from a living tumor. Cancer Res 2000; 60: 5401–5404.
Wyckoff JB, Jones JG, Condeelis JS, Segall JE . A critical step in metastasis: in vivo analysis of intravasation at the primary tumor. Cancer Res 2000; 60: 2504–2511.
Gligorijevic B, Wyckoff J, Yamaguchi H, Wang Y, Roussos ET, Condeelis J . N-WASP-mediated invadopodium formation is involved in intravasation and lung metastasis of mammary tumors. J Cell Sci 2012; 125: 724–734.
Kedrin D, Gligorijevic B, Wyckoff J, Verkhusha VV, Condeelis J, Segall JE et al. Intravital imaging of metastatic behavior through a mammary imaging window. Nat Methods 2008; 5: 1019–1021.
Entenberg D, Wyckoff J, Gligorijevic B, Roussos ET, Verkhusha VV, Pollard JW et al. Setup and use of a two-laser multiphoton microscope for multichannel intravital fluorescence imaging. Nat Protoc 2011; 6: 1500–1520.
Acknowledgements
We thank Drs Paraic Kenny, Richard Stanley, Jeffrey Segall and members of the Condeelis lab for helpful discussions. The AFS98, total and phospho-Y559 CSF1R antibodies were a generous gift from Dr Richard Stanley. For technical help at Albert Einstein College of Medicine, we thank the Histotechnology and Comparative Pathology Facility, the shRNA Core Facility, the FACS Facility (supported by NCI P30 CA 013330) and the Genomics Facility. This work was supported by NCI grants 5RO1CA164468 (AP, YW, XC, DE and JSC) and RO1CA170507 (JP, MO and JSC).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
JSC holds equity and is a consultant for MetaStat and for Deciphera Pharmaceuticals. DE is a consultant for MetaStat. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Patsialou, A., Wang, Y., Pignatelli, J. et al. Autocrine CSF1R signaling mediates switching between invasion and proliferation downstream of TGFβ in claudin-low breast tumor cells. Oncogene 34, 2721–2731 (2015). https://doi.org/10.1038/onc.2014.226
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.226
This article is cited by
-
Network pharmacology modeling identifies synergistic Aurora B and ZAK interaction in triple-negative breast cancer
npj Systems Biology and Applications (2019)
-
Claudin 11 regulates bone homeostasis via bidirectional EphB4-EphrinB2 signaling
Experimental & Molecular Medicine (2018)
-
High co-expression of IL-34 and M-CSF correlates with tumor progression and poor survival in lung cancers
Scientific Reports (2018)
-
KRT19 directly interacts with β-catenin/RAC1 complex to regulate NUMB-dependent NOTCH signaling pathway and breast cancer properties
Oncogene (2017)
-
In Vivo Visualization of Stromal Macrophages via label-free FLIM-based metabolite imaging
Scientific Reports (2016)