Abstract
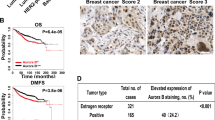
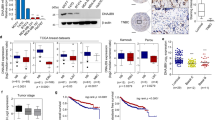
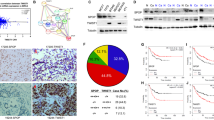
Epithelial–mesenchymal transition (EMT) is an essential step for tumor progression, although the mechanisms driving EMT are still not fully understood. In an effort to investigate these mechanisms, we observed that heregulin (HRG)-mediated activation of HER2, or HER2 overexpression, resulted in EMT, which is accompanied with increased expression of a known EMT regulator Slug, but not TWIST or Snail. We then investigated how HER2 induced Slug expression and found, for the first time, that there are four consensus HSF sequence-binding elements (HSEs), the binding sites for heat shock factor-1 (HSF-1), located in the Slug promoter. HSF-1 bound to and transactivated the Slug promoter independent of heat shock, leading to Slug expression in breast cancer cells. Mutation of the putative HSEs ablated Slug transcriptional activation induced by HRG or HSF-1 overexpression. Knockdown of HSF-1 expression by siRNA reduced Slug expression and HRG-induced EMT. The positive association between HSF-1 and Slug was confirmed by immunohistochemical staining of a cohort of 100 invasive breast carcinoma specimens. While investigating how HER2 activated HSF-1 independent of heat shock, we observed that HER2 activation resulted in concurrent phosphorylation of Akt and HSF-1. We then observed, also for the first time, that Akt directly interacted with HSF-1 and phosphorylated HSF-1 at S326. Inhibition of Akt using siRNA, dominant-negative Akt mutant, or small molecule inhibitors prevented HRG-induced HSF-1 activation and Slug expression. Conversely, constitutively active Akt induced HSF-1 phosphorylation and Slug expression. HSF-1 knockdown reduced the ability of Akt to induce Slug expression, indicating an essential role that HSF-1 plays in Akt-induced Slug upregulation. Altogether, our study uncovered the existence of a novel Akt-HSF-1 signaling axis that leads to Slug upregulation and EMT, and potentially contributes to progression of HER2-positive breast cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Thiery JP, Acloque H, Huang RY, Nieto MA . Epithelial-mesenchymal transitions in development and disease. Cell 2009; 139: 871–890.
De Craene B, Berx G . Regulatory networks defining EMT during cancer initiation and progression. Nat Rev Cancer 2013; 13: 97–110.
Rhim AD, Mirek ET, Aiello NM, Maitra A, Bailey JM, McAllister F et al. EMT and dissemination precede pancreatic tumor formation. Cell 2012; 148: 349–361.
Trimboli AJ, Fukino K, de Bruin A, Wei G, Shen L, Tanner SM et al. Direct evidence for epithelial-mesenchymal transitions in breast cancer. Cancer Res 2008; 68: 937–945.
Hajra KM, Chen DY, Fearon ER . The SLUG zinc-finger protein represses E-cadherin in breast cancer. Cancer Res 2002; 62: 1613–1618.
Savagner P, Yamada KM, Thiery JP . The zinc-finger protein slug causes desmosome dissociation, an initial and necessary step for growth factor-induced epithelial-mesenchymal transition. J Cell Biol 1997; 137: 1403–1419.
Cobaleda C, Perez-Caro M, Vicente-Duenas C, Sanchez-Garcia I . Function of the zinc-finger transcription factor SNAI2 in cancer and development. Annu Rev Genet 2007; 41: 41–61.
Batlle E, Sancho E, Franci C, Dominguez D, Monfar M, Baulida J et al. The transcription factor snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nat Cell Biol 2000; 2: 84–89.
Cano A, Perez-Moreno MA, Rodrigo I, Locascio A, Blanco MJ, del Barrio MG et al. The transcription factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nat Cell Biol 2000; 2: 76–83.
Lo HW, Hsu SC, Xia W, Cao X, Shih JY, Wei Y et al. Epidermal growth factor receptor cooperates with signal transducer and activator of transcription 3 to induce epithelial-mesenchymal transition in cancer cells via up-regulation of TWIST gene expression. Cancer Res 2007; 67: 9066–9076.
Yang J, Mani SA, Donaher JL, Ramaswamy S, Itzykson RA, Come C et al. Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell 2004; 117: 927–939.
Hynes NE, MacDonald G . ErbB receptors and signaling pathways in cancer. Curr Opin Cell Biol 2009; 21: 177–184.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL . Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 1987; 235: 177–182.
Slamon DJ, Godolphin W, Jones LA, Holt JA, Wong SG, Keith DE et al. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 1989; 244: 707–712.
Yarden Y, Sliwkowski MX . Untangling the ErbB signalling network. Nat Rev Mol Cell Biol 2001; 2: 127–137.
Ursini-Siegel J, Schade B, Cardiff RD, Muller WJ . Insights from transgenic mouse models of ERBB2-induced breast cancer. Nat Rev Cancer 2007; 7: 389–397.
Cheng JC, Qiu X, Chang HM, Leung PC . HER2 mediates epidermal growth factor-induced down-regulation of E-cadherin in human ovarian cancer cells. Biochem Biophys Res Commun 2013; 434: 81–86.
D'Souza B, Taylor-Papadimitriou J . Overexpression of ERBB2 in human mammary epithelial cells signals inhibition of transcription of the E-cadherin gene. Proc Natl Acad Sci USA 1994; 91: 7202–7206.
Giordano A, Gao H, Anfossi S, Cohen E, Mego M, Lee BN et al. Epithelial-mesenchymal transition and stem cell markers in patients with HER2-positive metastatic breast cancer. Mol Cancer Ther 2012; 11: 2526–2534.
Guettouche T, Boellmann F, Lane WS, Voellmy R . Analysis of phosphorylation of human heat shock factor 1 in cells experiencing a stress. BMC Biochem 2005; 6: 4.
Kroeger PE, Morimoto RI . Selection of new HSF1 and HSF2 DNA-binding sites reveals difference in trimer cooperativity. Mol Cell Biol 1994; 14: 7592–7603.
Ciocca DR, Arrigo AP, Calderwood SK . Heat shock proteins and heat shock factor 1 in carcinogenesis and tumor development: an update. Arch Toxicol 2013; 87: 19–48.
Mendillo ML, Santagata S, Koeva M, Bell GW, Hu R, Tamimi RM et al. HSF1 drives a transcriptional program distinct from heat shock to support highly malignant human cancers. Cell 2012; 150: 549–562.
Santagata S, Hu R, Lin NU, Mendillo ML, Collins LC, Hankinson SE et al. High levels of nuclear heat-shock factor 1 (HSF1) are associated with poor prognosis in breast cancer. Proc Natl Acad Sci USA 2011; 108: 18378–18383.
Xi C, Hu Y, Buckhaults P, Moskophidis D, Mivechi NF . Heat shock factor Hsf1 cooperates with ErbB2 (Her2/Neu) protein to promote mammary tumorigenesis and metastasis. J Biol Chem 2012; 287: 35646–35657.
Khaleque MA, Bharti A, Sawyer D, Gong J, Benjamin IJ, Stevenson MA et al. Induction of heat shock proteins by heregulin beta1 leads to protection from apoptosis and anchorage-independent growth. Oncogene 2005; 24: 6564–6573.
Meng L, Gabai VL, Sherman MY . Heat-shock transcription factor HSF1 has a critical role in human epidermal growth factor receptor-2-induced cellular transformation and tumorigenesis. Oncogene 2010; 29: 5204–5213.
Anckar J, Sistonen L . Regulation of HSF1 function in the heat stress response: implications in aging and disease. Annu Rev Biochem 2011; 80: 1089–1115.
Chou SD, Prince T, Gong J, Calderwood SK . mTOR is essential for the proteotoxic stress response, HSF1 activation and heat shock protein synthesis. PLoS One 2012; 7: e39679.
Sekulic A, Hudson CC, Homme JL, Yin P, Otterness DM, Karnitz LM et al. A direct linkage between the phosphoinositide 3-kinase-AKT signaling pathway and the mammalian target of rapamycin in mitogen-stimulated and transformed cells. Cancer Res 2000; 60: 3504–3513.
Gabai VL, Meng L, Kim G, Mills TA, Benjamin IJ, Sherman MY . Heat shock transcription factor Hsf1 is involved in tumor progression via regulation of hypoxia-inducible factor 1 and RNA-binding protein HuR. Mol Cell Biol 2012; 32: 929–940.
Yoon YJ, Kim JA, Shin KD, Shin DS, Han YM, Lee YJ et al. KRIBB11 inhibits HSP70 synthesis through inhibition of heat shock factor 1 function by impairing the recruitment of positive transcription elongation factor b to the hsp70 promoter. J Biol Chem 2011; 286: 1737–1747.
Ohnishi K, Takahashi A, Yokota S, Ohnishi T . Effects of a heat shock protein inhibitor KNK437 on heat sensitivity and heat tolerance in human squamous cell carcinoma cell lines differing in p53 status. Int J Radiat Biol 2004; 80: 607–614.
Westerheide SD, Kawahara TL, Orton K, Morimoto RI . Triptolide, an inhibitor of the human heat shock response that enhances stress-induced cell death. J Biol Chem 2006; 281: 9616–9622.
Lo HW, Cao X, Zhu H, Ali-Osman F . Constitutively activated STAT3 frequently coexpresses with epidermal growth factor receptor in high-grade gliomas and targeting STAT3 sensitizes them to Iressa and alkylators. Clin Cancer Res 2008; 14: 6042–6054.
Lo HW, Stephenson L, Cao X, Milas M, Pollock R, Ali-Osman F . Identification and Functional Characterization of the Human Glutathione S-Transferase P1 Gene as a Novel Transcriptional Target of the p53 Tumor Suppressor Gene. Mol Cancer Res 2008; 6: 843–850.
Zhao R, Yang HY, Shin J, Phan L, Fang L, Yeung SC et al. CDK inhibitor p57 (Kip2) is downregulated by Akt during HER2-mediated tumorigenicity. Cell Cycle 2013; 12: 935–943.
Wang X, Grammatikakis N, Siganou A, Calderwood SK . Regulation of molecular chaperone gene transcription involves the serine phosphorylation, 14-3-3 epsilon binding, and cytoplasmic sequestration of heat shock factor 1. Mol Cell Biol 2003; 23: 6013–6026.
Fujita N, Jaye DL, Kajita M, Geigerman C, Moreno CS, Wade PA . MTA3, a Mi-2/NuRD complex subunit, regulates an invasive growth pathway in breast cancer. Cell 2003; 113: 207–219.
Li YM, Pan Y, Wei Y, Cheng X, Zhou BP, Tan M et al. Upregulation of CXCR4 is essential for HER2-mediated tumor metastasis. Cancer Cell 2004; 6: 459–469.
Lo H-W, Hsu S-C, Ali-Seyed M, Gunduz M, Xia W, Wei Y et al. Nuclear Interaction of EGFR and STAT3 in the Activation of iNOS/NO Pathway. Cancer Cell 2005; 7: 575–589.
Lo HW, Zhu H, Cao X, Aldrich A, Ali-Osman F . A novel splice variant of GLI1 that promotes glioblastoma cell migration and invasion. Cancer Res 2009; 69: 6790–6798.
Lo HW, Cao X, Zhu H, Ali-Osman F . Cyclooxygenase-2 is a novel transcriptional target of the nuclear EGFR-STAT3 and EGFRvIII-STAT3 signaling axes. Mol Cancer Res 2010; 8: 232–245.
Zhu H, Cao X, Ali-Osman F, Keir S, Lo HW . EGFR and EGFRvIII interact with PUMA to inhibit mitochondrial translocalization of PUMA and PUMA-mediated apoptosis independent of EGFR kinase activity. Cancer Lett 2010; 294: 101–110.
Acknowledgements
We are thankful to Dr Mong-Hong Lee at MD Anderson Cancer Center who provided CA-Akt and DN-Akt constructs. This study was supported by the NIH grant K01-CA118423, and W81XWH-11-1-0600 from the US Department of Defense, the Beez Foundation and the Intramural Division of Surgical Sciences grants, Dani P. Bolognesi PhD Award and Clarence Gardner PhD Award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Carpenter, R., Paw, I., Dewhirst, M. et al. Akt phosphorylates and activates HSF-1 independent of heat shock, leading to Slug overexpression and epithelial–mesenchymal transition (EMT) of HER2-overexpressing breast cancer cells. Oncogene 34, 546–557 (2015). https://doi.org/10.1038/onc.2013.582
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.582
Keywords
This article is cited by
-
K-Ras(V12) differentially affects the three Akt isoforms in lung and pancreatic carcinoma cells and upregulates E-cadherin and NCAM via Akt3
Cell Communication and Signaling (2024)
-
Combining lapatinib and palbociclib inhibits cell proliferation and invasion via AKT signaling pathway in endocrine-resistant breast cancer cells
Medical Oncology (2024)
-
USP13 promotes breast cancer metastasis through FBXL14-induced Twist1 ubiquitination
Cellular Oncology (2023)
-
ErbB2 promotes breast cancer metastatic potential via HSF1/LDHA axis-mediated glycolysis
Medical Oncology (2022)
-
Epithelial-to-Mesenchymal Transition Signaling Pathways Responsible for Breast Cancer Metastasis
Cellular and Molecular Bioengineering (2022)