Abstract
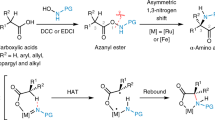
Chiral amines are prevalent in many bioactive molecules, including amino acids and pharmaceutical agents. tert-Butanesulfinamide (tBS) is a chiral amine reagent that has enabled the reliable asymmetric synthesis of a very broad range of different amine structures from simple, readily available starting materials. Three steps are commonly applied to the asymmetric synthesis of amines: (i) condensation of tBS with a carbonyl compound, (ii) nucleophile addition and (iii) tert-butanesulfinyl group cleavage. Here we demonstrate these steps with the preparation of a propargylic tertiary carbinamine, one of a class of amines that have been used for many different biological purposes, including click chemistry applications, diversity-oriented synthesis, the preparation of peptide isosteres and the development of protease inhibitors as drug candidates and imaging agents. The process described here can be performed in 3–4 d.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Nugent, T.C. Chiral Amine Synthesis: Methods, Developments and Applications 494 (Wiley, 2010).
Okamoto, Y. & Ikai, T. Chiral HPLC for efficient resolution of enantiomers. Chem. Soc. Rev. 37, 2593–2608 (2008).
Noyori, R. Asymmetric catalysis: science and opportunities (Nobel lecture). Angew. Chem Intl. Edn. 41, 2008–2022 (2002).
Savile, C.K. et al. Biocatalytic asymmetric synthesis of chiral amines from ketones applied to sitagliptin manufacture. Science 329, 305–309 (2010).
Kroutil, W. et al. Asymmetric preparation of prim-, sec-, and tert-amines employing selected biocatalysts. Org. Process Res. Dev. 17, 751–759 (2013).
Friestad, G.K. & Mathies, A.K. Recent developments in asymmetric catalytic addition to C=N bonds. Tetrahedron 63, 2541–2569 (2007).
Kobayashi, S., Mori, Y., Fossey, J.S. & Salter, M.M. Catalytic enantioselective formation of C-C bonds by addition to imines and hydrazones: a ten-year update. Chem. Rev. 111, 2626–2704 (2011).
Enders, D. & Reinhold, U. Asymmetric synthesis of amines by nucleophilic 1,2-addition of organometallic reagents to the CN-double bond. Tetrahedron Asymmetry 8, 1895–1946 (1997).
Robak, M.T., Herbage, M.A. & Ellman, J.A. Synthesis and applications of tert-butanesulfinamide. Chem. Rev. 110, 3600–3740 (2010).
Ferreira, F., Botuha, C., Chemla, F. & Perez-Luna, A. tert-Butanesulfinimines: structure, synthesis and synthetic applications. Chem. Soc. Rev. 38, 1162–1186 (2009).
Lin, G.-Q., Xu, M.-H., Zhong, Y.-W. & Sun, X.-W. An advance on exploring N-tert-butanesulfinyl imines in asymmetric synthesis of chiral amines. Acc. Chem. Res. 41, 831–840 (2008).
Morton, D. & Stockman, R.A. Chiral non-racemic sulfinimines: versatile reagents for asymmetric synthesis. Tetrahedron 62, 8869–8905 (2006).
Senanayake, C.H., Han, Z. & Krishnamurthy, D. Organosulfur chemistry in asymmetric synthesis. (Wiley, 2008).
Senanayake, C.H., Krishnamurthy, D., Lu, Z.-H., Han, Z. & Gallon, E. Enantiopure sulfoxides and sulfinamides: recent developments in their stereoselective synthesis and application to asymmetric synthesis. Aldrichim. Acta 38, 93–103 (2005).
Liu, G., Cogan, D.A., Owens, T.D., Tang, T.P. & Ellman, J.A. Synthesis of enantiomerically pure N-tert-butanesulfinyl imines (tert-butanesulfinimines) by the direct condensation of tert-butanesulfinamide with aldehydes and ketones. J. Org. Chem. 64, 1278–1284 (1999).
Datta, G.K. & Ellman, J.A. Racemization free protocol for the synthesis of N-tert-butanesulfinyl ketimines. J. Org. Chem. 75, 6283–6285 (2010).
Higashibayashi, S., Tohmiya, H., Mori, T., Hashimoto, K. & Nakata, M. Synthesis of sulfinimines by direct condensation of sulfinamides with aldehydes using Cs2CO3 as an activating and dehydrating reagent. Synlett 2004, 457–460 (2004).
Huang, Z., Zhang, M., Wang, Y. & Qin, Y. KHSO4-mediated condensation reactions of tert-butanesulfinamide with aldehydes. Preparation of tert-butanesulfinyl aldimines. Synlett 2005, 1334–1336 (2005).
Collados, J.F., Toledano, E., Guijarro, D. & Yus, M. Microwave-assisted solvent-free synthesis of enantiomerically pure N-(tert-butylsulfinyl)imines. J. Org. Chem. 77, 5744–5750 (2012).
Guijarro, D., Pablo, Ó. & Yus, M. Ruthenium-catalysed asymmetric transfer hydrogenation of N-(tert-butanesulfinyl)imines. Tetrahedron Lett. 50, 5386–5388 (2009).
Weix, D.J., Shi, Y. & Ellman, J.A. Diastereoselective and enantioselective Rh(I)-catalyzed additions of arylboronic acids to N-tert-butanesulfinyl and N-diphenylphosphinoyl aldimines. J. Am. Chem. Soc. 127, 1092–1093 (2005).
Beenen, M.A., Weix, D.J. & Ellman, J.A. Asymmetric synthesis of protected arylglycines by rhodium-catalyzed addition of arylboronic acids to N-tert-butanesulfinyl imino esters. J. Am. Chem. Soc. 128, 6304–6305 (2006).
Dai, H. & Lu, X. Diastereoselective synthesis of arylglycine derivatives by cationic palladium(II)-catalyzed addition of arylboronic acids to N-tert-butanesulfinyl imino esters. Org. Lett. 9, 3077–3080 (2007).
Xiao, X. et al. Selective diethylzinc reduction of imines in the presence of ketones catalyzed by Ni(acac)2 . Org. Lett. 8, 139–142 (2005).
Bolshan, Y. & Batey, R.A. A room-temperature protocol for the Rhodium(I)-catalyzed addition of arylboron compounds to sulfinimines. Org. Lett. 7, 1481–1484 (2005).
Boebel, T.A. & Hartwig, J.F. Conversion of 1,3-disubstituted arenes to chiral α,α-diaryl methylammonium chlorides using arene borylation. Tetrahedron 64, 6824–6830 (2008).
Beenen, M.A., An, C. & Ellman, J.A. Asymmetric copper-catalyzed synthesis of α-amino boronate esters from N-tert-butanesulfinyl aldimines. J. Am. Chem. Soc. 130, 6910–6911 (2008).
Liu, G.C., Cogan, D.A. & Ellman, J.A. Catalytic asymmetric synthesis of tert-butanesulfinamide. Application to the asymmetric synthesis of amines. J. Am. Chem. Soc. 119, 9913–9914 (1997).
Patterson, A.W. & Ellman, J.A. Asymmetric synthesis of α,α-dibranched propargylamines by acetylide additions to N-tert-butanesulfinyl ketimines. J. Org. Chem. 71, 7110–7112 (2006).
Lo, V.K.-Y., Zhou, C.-Y., Wong, M.-K. & Che, C.-M. Silver(i)-mediated highly enantioselective synthesis of axially chiral allenes under thermal and microwave-assisted conditions. Chem. Commun. 46, 213–215 (2010).
Corbett, J.W. et al. Inhibition of clinically relevant mutant variants of HIV-1 by quinazolinone non-nucleoside reverse transcriptase inhibitors. J. Med. Chem. 43, 2019–2030 (2000).
Trost, B.M., Chung, C.K. & Pinkerton, A.B. Stereocontrolled total synthesis of (+)-streptazolin by a palladium-catalyzed reductive diyne cyclization. Angew. Chem. Intl. Edn. 43, 4327–4329 (2004).
Davidson, M.H. & McDonald, F.E. Stereoselective synthesis of D-desosamine and related glycals via tungsten-catalyzed alkynol cycloisomerization. Org. Lett. 6, 1601–1603 (2004).
Brennan, C.J., Pattenden, G. & Rescourio, G. Formal synthesis of (+)-lactacystin based on a novel radical cyclisation of an α-ethynyl substituted serine. Tetrahedron Lett. 44, 8757–8760 (2003).
Cantel, S. et al. Synthesis and conformational analysis of a cyclic peptide obtained via i to i+4 intramolecular side-chain to side-chain azide-alkyne 1,3-dipolar cycloaddition. J. Org. Chem. 73, 5663–5674 (2008).
Brak, K., Doyle, P.S., McKerrow, J.H. & Ellman, J.A. Identification of a new class of nonpeptidic inhibitors of cruzain. J. Am. Chem. Soc. 130, 6404–6410 (2008).
Brak, K. et al. Nonpeptidic tetrafluorophenoxymethyl ketone cruzain inhibitors as promising new leads for Chagas disease chemotherapy. J. Med. Chem. 53, 1763–1773 (2010).
Verdoes, M. et al. A nonpeptidic cathepsin S activity-based probe for noninvasive optical imaging of tumor-associated macrophages. Chem. Biol. 19, 619–628 (2012).
Leyva, M.J. et al. Identification and evaluation of small molecule pan-caspase inhibitors in Huntington's disease models. Chem. Biol. 17, 1189–1200 (2010).
Deu, E. et al. Functional studies of Plasmodium falciparum dipeptidyl aminopeptidase I using small molecule inhibitors and active site probes. Chem. Biol. 17, 808–819 (2010).
Wood, W.J., Patterson, A.W., Tsuruoka, H., Jain, R.K. & Ellman, J.A. Substrate activity screening: a fragment-based method for the rapid identification of nonpeptidic protease inhibitors. J. Am. Chem. Soc. 127, 15521–15527 (2005).
Patterson, A.W. et al. Identification of selective, nonpeptidic nitrile inhibitors of cathepsin S using the substrate activity screening method. J. Med. Chem. 49, 6298–6307 (2006).
Inagaki, H. et al. Characterization and optimization of selective, nonpeptidic inhibitors of cathepsin S with an unprecedented binding mode. J. Med. Chem. 50, 2693–2699 (2007).
Moss, N. et al. Exploration of cathepsin S inhibitors characterized by a triazole P1-P2 amide replacement. Bioorg. Med. Chem. Lett. 22, 7189–7193 (2012).
Moura-Letts, G., Diblasi, C.M., Bauer, R.A. & Tan, D.S. Solid-phase synthesis and chemical space analysis of a 190-membered alkaloid/terpenoid-like library. Proc. Natl. Acad. Sci. USA 108, 6745–6750 (2011).
Cogan, D.A., Liu, G. & Ellman, J. Asymmetric synthesis of chiral amines by highly diastereoselective 1,2-additions of organometallic reagents to N-tert-butanesulfinyl imines. Tetrahedron 55, 8883–8904 (1999).
Shaw, A.W. & deSolms, S.J. Asymmetric synthesis of α,α-diaryl and α-aryl-α-heteroaryl alkylamines by organometallic additions to N-tert-butanesulfinyl ketimines. Tetrahedron Lett. 42, 7173–7176 (2001).
Chen, B.-L., Wang, B. & Lin, G.-Q. Highly diastereoselective addition of alkynylmagnesium chlorides to N-tert-butanesulfinyl aldimines: a practical and general access to chiral α-branched amines. J. Org. Chem. 75, 941–944 (2009).
Ding, C.-H., Chen, D.-D., Luo, Z.-B., Dai, L.-X. & Hou, X.-L. Highly diastereoselective synthesis of N-tert-butylsulfinylpropargylamines through direct addition of alkynes to N-tert-butanesulfinimines. Synlett 2006, 1272–1274 (2006).
Acknowledgements
This work was supported by the US National Science Foundation (CHE-1049571).
Author information
Authors and Affiliations
Contributions
H.-C.X. and S.C. carried out the experiments; J.A.E. designed the protocol and supervised the project; and H.-C.X. and J.A.E. assembled the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Data
1H NMR and 13C NMR spectra of products 16–19 (PDF 515 kb)
Rights and permissions
About this article
Cite this article
Xu, HC., Chowdhury, S. & Ellman, J. Asymmetric synthesis of amines using tert-butanesulfinamide. Nat Protoc 8, 2271–2280 (2013). https://doi.org/10.1038/nprot.2013.134
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2013.134
This article is cited by
-
Tunable chiroptical application by encapsulating achiral lanthanide complexes into chiral MOF thin films
Nano Research (2022)
-
A novel strategy for the asymmetric synthesis of (S)-ketamine using (S)-tert-butanesulfinamide and 1,2-cyclohexanedione
Journal of the Iranian Chemical Society (2018)
-
Study on the noncoincidence effect phenomenon using matrix isolated Raman spectra and the proposed structural organization model of acetone in condense phase
Scientific Reports (2017)
-
A reductive aminase from Aspergillus oryzae
Nature Chemistry (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.