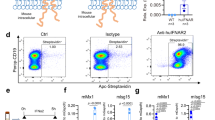
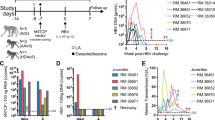
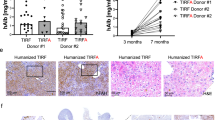
Abstract
Establishing a small animal model that accurately recapitulates hepatotropic pathogens, including hepatitis C virus (HCV) infection and immunopathogenesis, is essential for the study of hepatitis virus–induced liver disease and for therapeutics development. This protocol describes our recently developed humanized mouse model for studying HCV and other hepatotropic infections, human immune response and hepatitis and liver fibrosis. The first 5-h stage is the isolation of human liver progenitor and hematopoietic stem cells from fetal liver. Next, AFC8 immunodeficient mice are transplanted with the isolated progenitor/stem cells. This generally takes 2 h. The transplanted mice are then treated for a month with the mouse liver apoptosis–inducing AFC8 dimerizer and left for an additional 2-month period to permit human liver and immune cell growth as well as system reconstitution and development before inoculation with HCV clinical isolates. HCV infection, human immune response and liver disease are observed with high incidence from approximately 2 months after inoculation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mercer, D.F. et al. Hepatitis C virus replication in mice with chimeric human livers. Nat. Med. 7, 927–933 (2001).
Shultz, L.D., Ishikawa, F. & Greiner, D.L. Humanized mice in translational biomedical research. Nat. Rev. Immunol. 7, 118–130 (2007).
Willinger, T., Rongvaux, A., Strowig, T., Manz, M.G. & Flavell, R.A. Improving human hemato-lymphoid-system mice by cytokine knock-in gene replacement. Trends Immunol. 32, 321–327 (2011).
Meuleman, P. & Leroux-Roels, G. The human liver-uPA-SCID mouse: a model for the evaluation of antiviral compounds against HBV and HCV. Antiviral Res. 80, 231–238 (2008).
Meuleman, P. et al. Morphological and biochemical characterization of a human liver in a uPA-SCID mouse chimera. Hepatology 41, 847–856 (2005).
Azuma, H. et al. Robust expansion of human hepatocytes in Fah−/−/Rag2−/−/Il2rg−/− mice. Nat. Biotechnol. 25, 903–910 (2007).
Bissig, K.D., Le, T.T., Woods, N.B. & Verma, I.M. Repopulation of adult and neonatal mice with human hepatocytes: a chimeric animal model. Proc. Natl. Acad. Sci. USA 104, 20507–20511 (2007).
Bissig, K.D. et al. Human liver chimeric mice provide a model for hepatitis B and C virus infection and treatment. J. Clin. Invest. 120, 924–930 (2010).
Traggiai, E. et al. Development of a human adaptive immune system in cord blood cell-transplanted mice. Science 304, 104–107 (2004).
Pajvani, U.B. et al. Fat apoptosis through targeted activation of caspase 8: a new mouse model of inducible and reversible lipoatrophy. Nat. Med. 11, 797–803 (2005).
Washburn, M.L. et al. A humanized mouse model to study hepatitis C virus infection, immune response, and liver disease. Gastroenterology 140, 1334–1344 (2011).
Hayashi, H. & Sakai, T. Animal models for the study of liver fibrosis: new insights from knockout mouse models. Am. J. Physiol. Gastrointest. Liver Physiol. 300, G729–G738 (2011).
Merchante, N. et al. Survival and prognostic factors of HIV-infected patients with HCV-related end-stage liver disease. AIDS 20, 49–57 (2006).
Operskalski, E.A. & Kovacs, A. HIV/HCV co-infection: pathogenesis, clinical complications, treatment, and new therapeutic technologies. Curr. HIV/AIDS Rep. 8, 12–22 (2011).
de Jong, Y.P., Rice, C.M. & Ploss, A. New horizons for studying human hepatotropic infections. J. Clin. Invest. 120, 650–653 (2010).
Robinet, E. & Baumert, T.F. A first step towards a mouse model for hepatitis C virus infection containing a human immune system. J. Hepatol. 55, 718–720 (2011).
Haridass, D. et al. Repopulation efficiencies of adult hepatocytes, fetal liver progenitor cells, and embryonic stem cell-derived hepatic cells in albumin-promoter-enhancer urokinase-type plasminogen activator mice. Am. J. Pathol. 175, 1483–1492 (2009).
Mallet, V.O. et al. Conditional cell ablation by tight control of caspase-3 dimerization in transgenic mice. Nat. Biotechnol. 20, 1234–1239 (2002).
Zhang, L., Kovalev, G.I. & Su, L. HIV-1 infection and pathogenesis in a novel humanized mouse model. Blood 109, 2978–2981 (2007).
Lindenbach, B.D. et al. Cell culture-grown hepatitis C virus is infectious in vivo and can be recultured in vitro. Proc. Natl. Acad. Sci. USA 103, 3805–3809 (2006).
Acknowledgements
We thank the Su laboratory members for discussion and support. This work was supported in part by grants from a UNC University Cancer Research Fund innovation grant, from the US National Institutes of Health (AI076142, AA018009 to L.S.) and from a UNC Lineberger Comprehensive Cancer Center Postdoctoral Training grant (M.T.B).
Author information
Authors and Affiliations
Contributions
M.T.B. and L.S. organized and wrote the article; L.Z., M.L.W., T.A.C. and G.I.K. contributed to writing the article.
Corresponding authors
Ethics declarations
Competing interests
The University of North Carolina at Chapel Hill has applied for patents covering parts of the method, with L.Z., M.L.W. and L.S. as coinventors. The University of North Carolina at Chapel Hill manages the invention in accordance with its conflict of interest policies.
Rights and permissions
About this article
Cite this article
Bility, M., Zhang, L., Washburn, M. et al. Generation of a humanized mouse model with both human immune system and liver cells to model hepatitis C virus infection and liver immunopathogenesis. Nat Protoc 7, 1608–1617 (2012). https://doi.org/10.1038/nprot.2012.083
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2012.083
This article is cited by
-
Humanized Mice as Unique Tools for Human-Specific Studies
Archivum Immunologiae et Therapiae Experimentalis (2018)
-
Reconstitution of immune cell in liver and lymph node of adult- and newborn-engrafted humanized mice
BMC Immunology (2016)
-
Preclinical models for interrogating drug action in human cancers using Stable Isotope Resolved Metabolomics (SIRM)
Metabolomics (2016)
-
Experimental models of liver fibrosis
Archives of Toxicology (2016)
-
Mouse models for therapeutic vaccination against hepatitis B virus
Medical Microbiology and Immunology (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.