Abstract
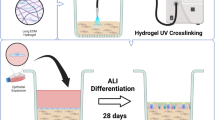
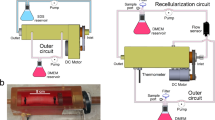
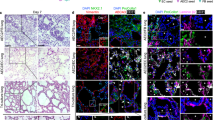
This protocol describes the setup, maintenance and characteristics of a tissue-engineered model of the human bronchial mucosa that can be used for basic physiology and pathophysiology studies. The model includes a well-differentiated epithelium with functional cilia, mucus secretion and subepithelial fibroblasts within type I collagen. The tissue is created within porous polymeric wells to prevent gel contraction and allow culture at the air–liquid interface. It requires at least 2 wk to be established and can be maintained thereafter for over 4 wk, with tissue differentiation moving towards a more physiologically relevant phenotype with increasing time in culture. Over time, the extracellular matrix also remodels, depositing proteins such as types III and IV collagen and fibronectin. Because it recapitulates many key anatomical and functional features of the airway wall, this model is well suited for a wide range of studies, including those on airway remodeling, transepithelial transport and inflammatory cell interactions with the mucosa. The entire protocol takes 4–6 wk, including cell expansion, depending on the extent of ciliogenesis desired.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cukierman, E., Pankov, R. & Yamada, K.M. Cell interactions with three-dimensional matrices. Curr. Opin. Cell Biol. 14, 633–639 (2002).
Pedersen, J.A. & Swartz, M.A. Mechanobiology in the third dimension. Ann. Biomed. Eng. 33, 1469–1490 (2005).
Griffith, L.G. & Swartz, M.A. Capturing complex 3D tissue physiology in vitro. Nat. Rev. Mol. Cell Biol. 7, 211–224 (2006).
Chakir, J. et al. Bronchial mucosa produced by tissue engineering: a new tool to study cellular interactions in asthma. J. Allergy Clin. Immunol. 107, 36–40 (2001).
Choe, M.M., Sporn, P.H. & Swartz, M.A. An in vitro airway wall model of remodeling. Am. J. Physiol. Lung Cell Mol. Physiol. 285, L427–L433 (2003).
Kojima, K. & Vacanti, C.A. Generation of a tissue-engineered tracheal equivalent. Biotechnol. Appl. Biochem. 39, 257–262 (2004).
Le Visage, C., Dunham, B., Flint, P. & Leong, K.W. Coculture of mesenchymal stem cells and respiratory epithelial cells to engineer a human composite respiratory mucosa. Tissue Eng. 10, 1426–1435 (2004).
Choe, M.M., Sporn, P.H. & Swartz, M.A. Extracellular matrix remodeling by dynamic strain in a 3D airway wall model. Am. J. Resp. Cell Mol. Biol. published online April 6 2006 (doi:10.1165/rcmb.2005-0443OC).
James, A. Airway remodeling in asthma. Curr. Opin. Pulm. Med. 11, 1–6 (2005).
Agarwal, A.R., Mih, J. & George, S.C. Expression of matrix proteins in an in vitro model of airway remodeling in asthma. Allergy Asthma Proc. 24, 35–42 (2003).
Paquette, J.S. et al. Tissue-engineered human asthmatic bronchial equivalents. Eur. Cell Mater. 7, 1–11 (2004).
Look, D.C. et al. Effects of paramyxoviral infection on airway epithelial cell Foxj1 expression, ciliogenesis, and mucociliary function. Am. J. Pathol. 159, 2055–2069 (2001).
Coraux, C. et al. Embryonic stem cells generate airway epithelial tissue. Am. J. Respir. Cell Mol. Biol. 32, 87–92 (2005).
Matsui, H., Randell, S.H., Peretti, S.W., Davis, C.W. & Boucher, R.C. Coordinated clearance of periciliary liquid and mucus from airway surfaces. J. Clin. Invest. 102, 1125–1131 (1998).
Sachs, L.A. et al. Effects of media on differentiation of cultured human tracheal epithelium. In Vitro Cell Dev. Biol. Anim. 39, 56–62 (2003).
Gray, T.E., Guzman, K., Davis, C.W., Abdullah, L.H. & Nettesheim, P. Mucociliary differentiation of serially passaged normal human tracheobronchial epithelial cells. Am. J. Respir. Cell Mol. Biol. 14, 104–112 (1996).
Bell, E., Ivarsson, B. & Merrill, C. Production of a tissue-like structure by contraction of collagen lattices by human fibroblasts of different proliferative potential in vitro. Proc. Natl. Acad. Sci. USA 76, 1274–1278 (1979).
LeDizet, M., Beck, J.C. & Finkbeiner, W.E. Differential regulation of centrin genes during ciliogenesis in human tracheal epithelial cells. Am. J. Physiol. 275, L1145–L1156 (1998).
de Jong, P.M. et al. Ciliogenesis in human bronchial epithelial cells cultured at the air–liquid interface. Am. J. Respir. Cell. Mol. Biol. 10, 271–277 (1994).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Choe, M., Tomei, A. & Swartz, M. Physiological 3D tissue model of the airway wall and mucosa. Nat Protoc 1, 357–362 (2006). https://doi.org/10.1038/nprot.2006.54
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.54
This article is cited by
-
Xenobiotica-metabolizing enzymes in the lung of experimental animals, man and in human lung models
Archives of Toxicology (2019)
-
Human Airway Primary Epithelial Cells Show Distinct Architectures on Membrane Supports Under Different Culture Conditions
Cell Biochemistry and Biophysics (2016)
-
Novel in vitro respiratory models to study lung development, physiology, pathology and toxicology
Stem Cell Research & Therapy (2013)
-
Three-dimensional tissue cultures: current trends and beyond
Cell and Tissue Research (2013)
-
Engineering a stem cell house into a home
Stem Cell Research & Therapy (2011)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.