Abstract
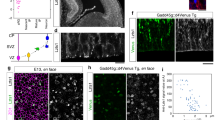
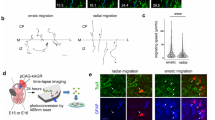
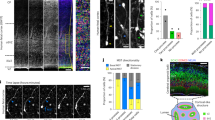
Midline astroglia in the cerebral cortex develop earlier than other astrocytes through mechanisms that are still unknown. We show that radial glia in dorsomedial cortex retract their apical endfeet at midneurogenesis and translocate to the overlaying pia, forming the indusium griseum. These cells require the fibroblast growth factor receptor 1 (Fgfr1) gene for their precocious somal translocation to the dorsal midline, as demonstrated by inactivating the Fgfr1 gene in radial glial cells and by RNAi knockdown of Fgfr1 in vivo. Dysfunctional astroglial migration underlies the callosal dysgenesis in conditional Fgfr1 knockout mice, suggesting that precise targeting of astroglia to the cortex has unexpected roles in axon guidance. FGF signaling is sufficient to induce somal translocation of radial glial cells throughout the cortex; furthermore, the targeting of astroglia to dorsolateral cortex requires FGFr2 signaling after neurogenesis. Hence, FGFs have an important role in the transition from radial glia to astrocytes by stimulating somal translocation of radial glial cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Schmechel, D.E. & Rakic, P. A Golgi study of radial glial cells in developing monkey telencephalon: morphogenesis and transformation into astrocytes. Anat. Embryol. (Berl.) 156, 115–152 (1979).
Pixley, S.K.R. & de Vellis, J. Transition between immature glia and mature astrocytes studied with a monoclonal antibody to vimentin. Dev. Brain Res. 15, 201–209 (1984).
Mission, J.P., Takahashi, T. & Caviness, V.S., Jr. Ontogeny of radial and other astroglial cells in murine cerebral cortex. Glia 4, 138–148 (1991).
Shu, T., Puche, A.C. & Richards, L.J. Development of midline glial populations at the corticoseptal boundary. J. Neurobiol. 57, 81–94 (2003).
Silver, J., Edwards, M.A. & Levitt, P. Immunocytochemical demonstration of early appearing astroglial structures that form boundaries and pathways along axon tracts in the fetal brain. J. Comp. Neurol. 328, 415–436 (1993).
Shu, T. & Richards, L.J. Cortical axon guidance by the glial wedge during the development of the corpus callosum. J. Neurosci. 21, 2749–2758 (2001).
Silver, J. & Ogawa, M.Y. Postnatally induced formation of the corpus callosum in acallosal mice on glia-coated cellulose bridges. Science 220, 1067–1069 (1983).
Shu, T., Butz, K.G., Plachez, C., Gronostajski, R.M. & Richards, L.J. Abnormal development of forebrain midline glia and commissural projections in Nfia knock-out mice. J. Neurosci. 23, 203–212 (2003).
Kriegstein, A.R. & Gotz, M. Radial glia diversity: a matter of cell fate. Glia 43, 37–43 (2003).
Qian, X., Davis, A.A., Goderie, S.K. & Temple, S. FGF2 concentration regulates the generation of neurons and glia from multipotent cortical stem cells. Neuron 18, 81–93 (1997).
Song, M.R. & Ghosh, A. FGF2-induced chromatin remodeling regulates CNTF-mediated gene expression and astrocyte differentiation. Nat. Neurosci. 7, 229–235 (2004).
Morrow, T., Song, M.R. & Ghosh, A. Sequential specification of neurons and glia by developmentally regulated extracellular factors. Development 128, 3585–3594 (2001).
Ornitz, D. & Itoh, N. Fibroblast growth factors. Genome Biol. 2, 3005–3012 (2001).
Itoh, N. & Ornitz, D.M. Evolution of the Fgf and Fgfr gene families. Trends Genet. 20, 563–569 (2004).
Storm, E.E., Rubenstein, J.L. & Martin, G.R. Dosage of FGF8 determines whether cell survival is positively or negatively regulated in the developing forebrain. Proc. Natl. Acad. Sci. USA 100, 1757–1762 (2003).
Vaccarino, F.M. et al. Changes in cerebral cortex size are governed by fibroblast growth factor during embryogenesis. Nat. Neurosci. 2, 246–253 (1999).
Raballo, R. et al. Basic fibroblast growth factor (Fgf2) is necessary for cell proliferation and neurogenesis in the developing cerebral cortex. J. Neurosci. 20, 5012–5023 (2000).
Garel, S., Huffman, K.J. & Rubenstein, J.L.R. Molecular regionalization of the neocortex is disrupted in Fgf8 hypomorphic mutants. Development 130, 1903–1914 (2003).
Fukuchi-Shimogori, T. & Grove, E.A. Neocortex patterning by the secreted signaling molecule FGF8. Science 294, 1071–1074 (2001).
Ohkubo, Y., Chiang, C. & Rubenstein, J.L. Coordinate regulation and synergistic actions of BMP4, SHH and FGF8 in the rostral prosencephalon regulate morphogenesis of the telencephalic and optic vesicles. Neuroscience 111, 1–17 (2002).
Gregg, C. & Weiss, S. Generation of functional radial glial cells by embryonic and adult forebrain neural stem cells. J. Neurosci. 23, 11587–11601 (2003).
Yoon, K. et al. Fibroblast growth factor receptor signaling promotes radial glial identity and interacts with Notch1 signaling in telencephalic progenitors. J. Neurosci. 24, 9497–9506 (2004).
Ohkubo, Y., Uchida, A.O., Shin, D., Partanen, J. & Vaccarino, F.M. Fibroblast growth factor receptor 1 is required for the proliferation of hippocampal progenitor cells and for hippocampal growth in mouse. J. Neurosci. 24, 6057–6069 (2004).
Dode, C. et al. Loss-of-function mutations in FGFR1 cause autosomal dominant Kallmann syndrome. Nat. Genet. 33, 463–465 (2003).
Pirvola, U. et al. FGFR1 is required for the development of the auditory sensory epithelium. Neuron 35, 671–680 (2002).
Malatesta, P. et al. Neuronal or glial progeny: regional differences in radial glia fate. Neuron 37, 751–764 (2003).
Graus-Porta, D. et al. Beta1-class integrins regulate the development of laminae and folia in the cerebral and cerebellar cortex. Neuron 31, 367–379 (2001).
Ozaki, H.S. & Wahlsten, D. Prenatal formation of the normal mouse corpus callosum: a quantitative study with carbocyanine dyes. J. Comp. Neurol. 323, 81–90 (1992).
Minowada, G. et al. Vertebrate Sprouty genes are induced by FGF signaling and can cause chondrodysplasia when overexpressed. Development 126, 4465–4475 (1999).
Maruoka, Y. et al. Comparison of the expression of three highly related genes, FgF8, Fgf17 and Fgf18, in the mouse embryo. Mech. Dev. 74, 175–177 (1998).
Crossley, P.H. & Martin, G.R. The mouse FGF8 gene encodes a family of polypeptides and is expressed in regions that direct outgrowth and patterning in the developing embyo. Development 121, 439–451 (1995).
Shu, T., Sundaresan, V., McCarthy, M.M. & Richards, L.J. Slit2 guides both precrossing and postcrossing callosal axons at the midline in vivo. J. Neurosci. 23, 8176–8184 (2003).
Zhu, Y. et al. Ablation of NF1 function in neurons induces abnormal development of cerebral cortex and reactive gliosis in the brain. Genes Dev. 15, 859–876 (2001).
Soriano, P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat. Genet. 21, 70–71 (1999).
Hasegawa, H. et al. Laminar patterning in the developing neocortex by temporally coordinated fibroblast growth factor signaling. J. Neurosci. 24, 8711–8719 (2004).
Yu, K., Herr, A.B., Waksman, G. & Ornitz, D.M. Loss of fibroblast growth factor receptor 2 ligand-binding specificity in Apert syndrome. Proc. Natl. Acad. Sci. USA 97, 14536–14541 (2000).
Meyers, E.N., Lewandoski, M. & Martin, G.R. An FGF8 mutant allelic series generated by Cre-and Flp-mediated recombination. Nat. Genet. 18, 136–141 (1998).
Huffman, K.J., Garel, S. & Rubenstein, J.L. FGF8 regulates the development of intra-neocortical projections. J. Neurosci. 24, 8917–8923 (2004).
Shanmugalingam, S. et al. Ace/FGF8 is required for forebrain commissure formation and patterning of the telencephalon. Development 127, 2549–2561 (2000).
Klambt, C., Glazer, L. & Shilo, B.Z. breathless, a Drosophila FGF receptor homolog, is essential for migration of tracheal and specific midline glial cells. Genes Dev. 6, 1668–1678 (1992).
Williams, E.J., Furness, J., Walsh, F.S. & Doherty, P. Activation of the FGF receptor underlies neurite outgrowth stimulated by L1, N-CAM, and N-Cadherin. Neuron 13, 583–594 (1994).
Webber, C.A., Hyakutake, M.T. & McFarlane, S. Fibroblast growth factors redirect retinal axons in vitro and in vivo. Dev. Biol. 263, 24–34 (2003).
Rash, B.G. & Richards, L.J. A role for cingulate pioneering axons in the development of the corpus callosum. J. Comp. Neurol. 434, 147–157 (2001).
Shin, D.M. et al. Loss of glutamatergic pyramidal neurons in frontal and temporal cortex resulting from attenuation of FGFR1 signaling is associated with spontaneous hyperactivity in mice. J. Neurosci. 24, 2247–2258 (2004).
Cheng, Y., Black, I.B. & DiCicco-Bloom, E. Hippocampal granule neuron production and population size are regulated by levels of bFGF. Eur. J. Neurosci. 15, 3–12 (2002).
Faux, C.H., Turnley, A.M., Epa, R., Cappai, R. & Bartlett, P.F. Interactions between fibroblast growth factors and Notch regulate neuronal differentiation. J. Neurosci. 21, 5587–5596 (2001).
Hebert, J.M., Lin, M., Partanen, J., Rossant, J. & McConnell, S.K. FGF signaling through FGFR1 is required for olfactory bulb morphogenesis. Development 15, 1101–1111 (2003).
Zhuo, L. et al. hGFAP-cre transgenic mice for manipulation of glial and neuronal function in vivo. Genesis 31, 85–94 (2001).
Chen, J.G., Rasin, M.R., Kwan, K.Y. & Sestan, N. Zfp312 is required for subcortical axonal projections and dendritic morphology of deep-layer pyramidal neurons of the cerebral cortex. Proc. Natl. Acad. Sci. USA 102, 17792–17797 (2005).
Schmid, R.S., Yokota, Y. & Anton, E.S. Generation and characterization of brain lipid-binding protein promoter-based transgenic mouse models for the study of radial glia. Glia 53, 345–351 (2006).
Acknowledgements
We thank A. Uchida for explant preparation and valuable discussions; S.L. Ellis and J. Silbereis for technical assistance; R. Slack (University of Ottawa, Ottawa, Ontario) for the Nestin-Cre mice; J. Marth (University of California San Diego) for the SynI-Cre mice; J. Partanen (Institute of Biotechnology, University of Helsinki, Helsinki, Finland) for the FGFr1f/f mice; D. Ornitz (Washington University Medical School, Saint Louis) for the FGFr2f/f mice; K. Kwan (Yale University, New Haven) for generating pCGLH; E. Anton (University of North Carolina, Chapel Hill, North Carolina) for providing pBLBP-EGFP; and J. Miyazaki (Osaka University, Osaka, Japan) for providing pCAGGS. We are grateful to M. Tessier-Lavigne (Genentech, South San Francisco) for probes and J. Rubenstein (University of California San Francisco) for sharing data. This work was supported by the US National Institutes of Health (grants MH067715, NS35476, 5T32-MH18268, NS054273 and HD045481) and the National Alliance for Research on Schizophrenia and Depression Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Lack of dorsal telencephalic commissures in Fgfr1 mutants correlates with absence of the IG. (PDF 1564 kb)
Supplementary Fig. 2
Fgfr1 mutants have specific deficits in the formation of dorsal telencephalic commissures. (PDF 706 kb)
Supplementary Fig. 3
Fgfr1 mutation does not alter the expression of neuronal factors implicated in midline axon guidance. (PDF 487 kb)
Supplementary Fig. 4
In situ hybridization and RT-PCR of axon guidance molecules in control and hGfap/Cre Fgfr1 mutant embryos. (PDF 497 kb)
Supplementary Fig. 5
Decreased expression of Fgfr1 protein after shRNA knockdown and Fgfr2 mRNA in Fgfr2f/f;hGfapCre mice. (PDF 573 kb)
Rights and permissions
About this article
Cite this article
Smith, K., Ohkubo, Y., Maragnoli, M. et al. Midline radial glia translocation and corpus callosum formation require FGF signaling. Nat Neurosci 9, 787–797 (2006). https://doi.org/10.1038/nn1705
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1705
This article is cited by
-
Ca2+ homeostasis maintained by TMCO1 underlies corpus callosum development via ERK signaling
Cell Death & Disease (2022)
-
Enhanced FGFR3 activity in postmitotic principal neurons during brain development results in cortical dysplasia and axonal tract abnormality
Scientific Reports (2020)
-
Mutations in SMARCB1 and in other Coffin–Siris syndrome genes lead to various brain midline defects
Nature Communications (2019)
-
Actionable FGFR1 and BRAF mutations in adult circumscribed gliomas
Journal of Neuro-Oncology (2019)
-
Nkx2.1 regulates the generation of telencephalic astrocytes during embryonic development
Scientific Reports (2017)