Abstract
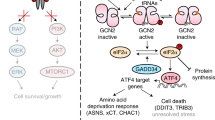
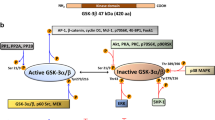
A fundamental challenge in treating disease is identifying molecular states that affect cellular responses to drugs. Here, we focus on glycogen synthase kinase 3 (GSK-3), a key regulator for many of the hallmark behaviors of cancer cells. We alter GSK-3 activity in colon epithelial cells to test its role in modulating drug response. We find that GSK-3 activity broadly affects the cellular sensitivities to a panel of oncology drugs and kinase inhibitors. Specifically, inhibition of GSK-3 activity can strongly desensitize or sensitize cells to kinase inhibitors (for example, mTOR or PLK1 inhibitors, respectively). Additionally, colorectal cancer cell lines, in which GSK-3 function is commonly suppressed, are resistant to mTOR inhibitors and yet highly sensitive to PLK1 inhibitors, and this is further exacerbated by additional GSK-3 inhibition. Finally, by conducting a kinome-wide RNAi screen, we find that GSK-3 modulates the cell proliferative phenotype of a large fraction (∼35%) of the kinome, which includes ∼50% of current, clinically relevant kinase-targeted drugs. Our results highlight an underappreciated interplay of GSK-3 with therapeutically important kinases and suggest strategies for identifying disease-specific molecular profiles that can guide optimal selection of drug treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Doble, B.W. & Woodgett, J.R. GSK-3: tricks of the trade for a multi-tasking kinase. J. Cell Sci. 116, 1175–1186 (2003).
Kim, L. & Kimmel, A.R. GSK3, a master switch regulating cell-fate specification and tumorigenesis. Curr. Opin. Genet. Dev. 10, 508–514 (2000).
Pap, M. & Cooper, G.M. Role of glycogen synthase kinase-3 in the phosphatidylinositol 3-kinase/Akt cell survival pathway. J. Biol. Chem. 273, 19929–19932 (1998).
Sutherland, C. What are the bona fide GSK3 substrates? Int. J. Alzheimers Dis. 2011, 505607 (2011).
Farago, M. et al. Kinase-inactive glycogen synthase kinase 3β promotes Wnt signaling and mammary tumorigenesis. Cancer Res. 65, 5792–5801 (2005).
Kang, T. et al. GSK-3β targets Cdc25A for ubiquitin-mediated proteolysis, and GSK-3β inactivation correlates with Cdc25A overproduction in human cancers. Cancer Cell 13, 36–47 (2008).
Leis, H., Segrelles, C., Ruiz, S., Santos, M. & Paramio, J.M. Expression, localization, and activity of glycogen synthase kinase 3β during mouse skin tumorigenesis. Mol. Carcinog. 35, 180–185 (2002).
Zhang, H.H., Lipovsky, A.I., Dibble, C.C., Sahin, M. & Manning, B.D. S6K1 regulates GSK3 under conditions of mTOR-dependent feedback inhibition of Akt. Mol. Cell 24, 185–197 (2006).
Zhou, B.P. et al. Dual regulation of Snail by GSK-3β–mediated phosphorylation in control of epithelial-mesenchymal transition. Nat. Cell Biol. 6, 931–940 (2004).
Cancer Genome Atlas, N. Comprehensive molecular characterization of human colon and rectal cancer. Nature 487, 330–337 (2012).
Knox, C. et al. DrugBank 3.0: a comprehensive resource for ′omics′ research on drugs. Nucleic Acids Res. 39, D1035–D1041 (2011).
Roig, A.I. et al. Immortalized epithelial cells derived from human colon biopsies express stem cell markers and differentiate in vitro. Gastroenterology 138, 1012-21.e1–1012-21.e5 (2010).
Doble, B.W., Patel, S., Wood, G.A., Kockeritz, L.K. & Woodgett, J.R. Functional redundancy of GSK-3α and GSK-3β in Wnt/β-catenin signaling shown by using an allelic series of embryonic stem cell lines. Dev. Cell 12, 957–971 (2007).
Bennett, C.N. et al. Regulation of Wnt signaling during adipogenesis. J. Biol. Chem. 277, 30998–31004 (2002).
US National Cancer Institutes–National Institutes of Health. Synthetics and Pure Natural Products. Developmental Therapeutics Program NCI/NIH, http://dtp.nci.nih.gov/branches/dscb/oncology_drugset_explanation.html (2014).
Yost, C. et al. The axis-inducing activity, stability, and subcellular distribution of β-catenin is regulated in Xenopus embryos by glycogen synthase kinase 3. Genes Dev. 10, 1443–1454 (1996).
Liu, C. et al. Control of β-catenin phosphorylation/degradation by a dual-kinase mechanism. Cell 108, 837–847 (2002).
Jacinto, E. et al. Mammalian TOR complex 2 controls the actin cytoskeleton and is rapamycin insensitive. Nat. Cell Biol. 6, 1122–1128 (2004).
Sarbassov, D.D. et al. Rictor, a novel binding partner of mTOR, defines a rapamycin-insensitive and raptor-independent pathway that regulates the cytoskeleton. Curr. Biol. 14, 1296–1302 (2004).
Efeyan, A. & Sabatini, D.M. mTOR and cancer: many loops in one pathway. Curr. Opin. Cell Biol. 22, 169–176 (2010).
Sabatini, D.M. mTOR and cancer: insights into a complex relationship. Nat. Rev. Cancer 6, 729–734 (2006).
Thoreen, C.C. & Sabatini, D.M. Rapamycin inhibits mTORC1, but not completely. Autophagy 5, 725–726 (2009).
O′Reilly, K.E. et al. mTOR inhibition induces upstream receptor tyrosine kinase signaling and activates Akt. Cancer Res. 66, 1500–1508 (2006).
Chen, C.H. et al. ER stress inhibits mTORC2 and Akt signaling through GSK-3beta-mediated phosphorylation of rictor. Sci. Signal. 4, ra10 (2011).
Huang, J. & Manning, B.D. A complex interplay between Akt, TSC2 and the two mTOR complexes. Biochem. Soc. Trans. 37, 217–222 (2009).
Thoreen, C.C. et al. An ATP-competitive mammalian target of rapamycin inhibitor reveals rapamycin-resistant functions of mTORC1. J. Biol. Chem. 284, 8023–8032 (2009).
Feldman, M.E. et al. Active-site inhibitors of mTOR target rapamycin-resistant outputs of mTORC1 and mTORC2. PLoS Biol. 7, e38 (2009).
Petronczki, M., Lenart, P. & Peters, J.M. Polo on the rise–from mitotic entry to cytokinesis with Plk1. Dev. Cell 14, 646–659 (2008).
Rudolph, D. et al. BI 6727, a Polo-like kinase inhibitor with improved pharmacokinetic profile and broad antitumor activity. Clin. Cancer Res. 15, 3094–3102 (2009).
Olmos, D. et al. Phase I study of GSK461364, a specific and competitive Polo-like kinase 1 inhibitor, in patients with advanced solid malignancies. Clin. Cancer Res. 17, 3420–3430 (2011).
Schöffski, P. Polo-like kinase (PLK) inhibitors in preclinical and early clinical development in oncology. Oncologist 14, 559–570 (2009).
Zheng, H., Saito, H., Masuda, S., Yang, X. & Takano, Y. Phosphorylated GSK3β-Ser9 and EGFR are good prognostic factors for lung carcinomas. Anticancer Res. 27, 3561–3569 (2007).
Fiol, C.J. et al. Phosphoserine as a recognition determinant for glycogen synthase kinase-3: phosphorylation of a synthetic peptide based on the G-component of protein phosphatase-1. Arch. Biochem. Biophys. 267, 797–802 (1988).
Frame, S., Cohen, P. & Biondi, R.M. A common phosphate binding site explains the unique substrate specificity of GSK3 and its inactivation by phosphorylation. Mol. Cell 7, 1321–1327 (2001).
Thomas, G.M. et al. A GSK3-binding peptide from FRAT1 selectively inhibits the GSK3-catalysed phosphorylation of axin and β-catenin. FEBS Lett. 458, 247–251 (1999).
Tighe, A., Ray-Sinha, A., Staples, O.D. & Taylor, S.S. GSK-3 inhibitors induce chromosome instability. BMC Cell Biol. 8, 34 (2007).
Hertz, N.T. et al. A neo-substrate that amplifies catalytic activity of Parkinson′s-disease–related kinase PINK1. Cell 154, 737–747 (2013).
Simpson, G.L., Hughes, J.A., Washio, Y. & Bertrand, S.M. Direct small-molecule kinase activation: novel approaches for a new era of drug discovery. Curr. Opin. Drug Discov. Devel. 12, 585–596 (2009).
Thorne, C.A. et al. Small-molecule inhibition of Wnt signaling through activation of casein kinase 1α. Nat. Chem. Biol. 6, 829–836 (2010).
Coster, A.D., Wichaidit, C., Rajaram, S., Altschuler, S.J. & Wu, L.F. A simple image correction method for high-throughput microscopy. Nat. Methods 11, 602 (2014).
Acknowledgements
We thank M. Cobb and members of the Altschuler and Wu laboratories for critical reading of the manuscript; J. Shay (University of Texas Southwestern) for providing the HCEC cell line and guidance on culture techniques, M. Ramirez for experimental support and J. Life for help with high-throughput screening experiments. This research was supported by a Cancer Biology Training Grant T32 CA124334 (C.A.T.), an American Cancer Society–Lakeshore Division Postdoctoral Fellowship (C.A.T.), the National Institute of Health grants K99 DK103126-01 (C.A.T.), CA133253 (S.J.A.), CA184984 (L.F.W.), R01 GM081549 (L.F.W.), CPRIT RP10900 (L.F.W.), and the Welch Foundation I-1619 (S.J.A.) and I-1644 (L.F.W.).
Author information
Authors and Affiliations
Contributions
C.A.T. designed, performed and analyzed experiments. C.W. designed experiments and performed analysis of data. A.D.C. aided in analysis of data. B.A.P. designed HTS experiments and provided critical reagents. C.A.T., C.W., L.F.W. and S.J.A. wrote the manuscript with advice from all authors. L.F.W. and S.J.A. guided all aspects of this study.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Figures 1–10, Supplementary Table 1 and Supplementary Notes 1–4 (PDF 2419 kb)
Supplementary Data Set 1
DrugBank gene in kinome list (XLSX 118 kb)
Supplementary Data Set 2
Oncology set (XLSX 27 kb)
Supplementary Data Set 3
PKIS (XLS 234 kb)
Supplementary Data Set 4
RNAi (XLSX 26 kb)
Rights and permissions
About this article
Cite this article
Thorne, C., Wichaidit, C., Coster, A. et al. GSK-3 modulates cellular responses to a broad spectrum of kinase inhibitors. Nat Chem Biol 11, 58–63 (2015). https://doi.org/10.1038/nchembio.1690
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1690
This article is cited by
-
Blocking methionine catabolism induces senescence and confers vulnerability to GSK3 inhibition in liver cancer
Nature Cancer (2024)
-
Targeting a splicing-mediated drug resistance mechanism in prostate cancer by inhibiting transcriptional regulation by PKCβ1
Oncogene (2022)
-
Glycogen synthase kinase 3β inhibition synergizes with PARP inhibitors through the induction of homologous recombination deficiency in colorectal cancer
Cell Death & Disease (2021)
-
Functional patient-derived cellular models for neuropsychiatric drug discovery
Translational Psychiatry (2021)
-
Chemical compound cinobufotalin potently induces FOXO1-stimulated cisplatin sensitivity by antagonizing its binding partner MYH9
Signal Transduction and Targeted Therapy (2019)