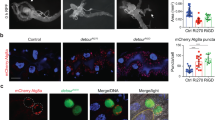
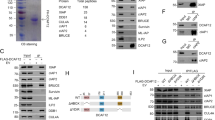
Abstract
In Drosophila melanogaster, apoptosis is controlled by the integrated actions of the Grim-Reaper (Grim-Rpr) and Drosophila Inhibitor of Apoptosis (DIAP) proteins (reviewed in refs 1–4). The anti-apoptotic DIAPs bind to caspases and inhibit their proteolytic activities. DIAPs also bind to Grim-Rpr proteins, an interaction that promotes caspase activity and the initiation of apoptosis. Using a genetic modifier screen, we identified four enhancers of grim-reaper-induced apoptosis that all regulate ubiquitination processes: uba-1, skpA, fat facets (faf), and morgue. Strikingly, morgue encodes a unique protein that contains both an F box and a ubiquitin E2 conjugase domain that lacks the active site Cys required for ubiquitin linkage. A reduction of morgue activity suppressed grim-reaper-induced cell death in Drosophila. In cultured cells, Morgue induced apoptosis that was suppressed by DIAP1. Targeted morgue expression downregulated DIAP1 levels in Drosophila tissue, and Morgue and Rpr together downregulated DIAP1 levels in cultured cells. Consistent with potential substrate binding functions in an SCF ubiquitin E3 ligase complex, Morgue exhibited F box-dependent association with SkpA and F box-independent association with DIAP1. Morgue may thus have a key function in apoptosis by targeting DIAP1 for ubiquitination and turnover.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Abrams, J. M. Trends Cell Biol. 9, 435–440 (1999).
Bangs, P. & White, K. Dev. Dyn. 218, 68–79 (2000).
Tittel, J. N. & Steller, H. Genome Biol. 1, 0003.1–0003.6 (2000).
Vernooy, S. Y. et al. J. Cell Biol. 150, F69–F76 (2000).
Wing, J. P., Schwartz, L. M. & Nambu, J. R. Mech. Dev. 102, 193–203 (2001).
Rørth, P. et al. Development 125, 1049–1057 (1998).
Tyers, M. & Jorgensen, P. Curr. Opin. Genet. Dev. 10, 54–64 (2000).
Kipreos, E. T. & Pagano, M. Genome Biol. 1, 3002.1–3002.7 (2000).
Cooley, L., Thompson, D. & Spradling, A. C. Proc. Natl Acad. Sci. USA 87, 3170–3173 (1990).
Sancho, E. et al. Mol. Cell. Biol. 18, 576–589 (1998).
Xiao, W., Lin, S. L., Broomfield, S., Chow, B. L. & Wei, Y. F. Nucleic Acids Res. 26, 3908–3914 (1998).
Hofmann, R. M. & Pickart, C. M. Cell 96, 645–653 (1999).
Zheng, N., Wang, P., Jeffrey, P. D. & Pavletich, N. P. Cell 102, 533–539 (2000).
Hay, B. A. Cell Death Differ. 7, 1045–1056 (2000).
Yang, Y., Fang, S., Jensen, J. P., Weissman, A. M. & Ashwell, J. D. Science 288, 874–877 (2000).
Orlowski, R. Z. Cell Death Differ. 6, 303–313 (1999).
Wojcik, C. Cell Mol. Life Sci. 56, 908–917 (1999).
Jesenberger, V. & Jentsch, S. Nature Rev. Mol. Cell Biol. 3, 112–121 (2002).
Haas, A. L., Baboshina, O., Williams, B. & Schwartz, L. M. J. Biol Chem. 270, 9407–9412 (1995).
Grimm, L. M., Goldberg, A. L., Poirier, G. G., Schwartz, L. M. & Osborne, B. A. EMBO J. 15, 3835–3844 (1996).
Sadoul, R. et al. EMBO J. 15, 3845–3852 (1996).
Suzuki, Y., Nakabayashi, Y. & Takahashi, R. Proc. Natl Acad. Sci. USA 98, 8662–8667 (2001).
Huang, H. et al. J. Biol. Chem. 275, 26661–26664 (2000).
Zhou, L. et al. Proc. Natl Acad. Sci. USA 94, 5131–5136 (1997).
Ma, Y. et al. J. Neurosci. 20, 4596–4605 (2000).
Lisi, S., Mazzon, I. & White, K. Genetics 154, 669–678 (2000).
Wing, J. P. et al. Curr. Biol. 12, 131–135 (2002).
Bunch, T. A., Grinblat, Y. & Goldstein, L. S. Nucleic Acids Res. 16, 1043–1061 (1988).
Holley, C.L., Olson, M.R., Colon-Ramos, D.A. & Kornbluth S. Nature Cell Biol.DOI: 10.1038/ncb798.
Ryoo, H.D., Bergmann, A., Gonen, H., Ciechanover, A. & Steller, H. Nature Cell Biol. DOI: 10.1038/ncb795.
Hays, R., Wickline, L. & Cagan R. Nature Cell Biol. DOI: 10.1038/ncb794.
Yoo et al. Nature Cell Biol. DOI: 10.1038/ncb793.
Wilson et al. Nature Cell Biol. DOI: 10.1038/ncb799.
Acknowledgements
The authors are grateful to S. Boulton for providing a SkpA expression plasmid and purified GST–SkpA protein. We also thank the Bloomington Drosophila Stock Center, G. Reuter, J. Szidonya, the Berkeley Drosophila Genome Project and the Developmental Studies Hybridoma Bank for providing fly strains, DNAs and antibodies. We are grateful to R. Cagan for discussions on morgue gene analyses before publication. K.W. is supported by National Institutes of Health grant GM55568 and a grant from the Shiseido Company of Japan to the Massachusetts General Hospital/Harvard Medical School. L.M.S. and J.R.N. were supported by NIH grant AG55118.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary figure and table
Figure S1. morgue expression induces apoptosis in cultured Spodoptera Sf-9 cells. (PDF 67 kb)
Table 1. Modification of R/Grim-induced Eye Cell Death by EP Insertions
Rights and permissions
About this article
Cite this article
Wing, J., Schreader, B., Yokokura, T. et al. Drosophila Morgue is an F box/ubiquitin conjugase domain protein important for grim-reaper mediated apoptosis. Nat Cell Biol 4, 451–456 (2002). https://doi.org/10.1038/ncb800
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb800
This article is cited by
-
Genetic characterization of two gain-of-function alleles of the effector caspase DrICE in Drosophila
Cell Death & Differentiation (2016)
-
La FAM fatale: USP9X in development and disease
Cellular and Molecular Life Sciences (2015)
-
The drosophila Bcl-2 family protein Debcl is targeted to the proteasome by the β-TrCP homologue slimb
Apoptosis (2014)
-
The cullin protein family
Genome Biology (2011)
-
The predator becomes the prey: regulating the ubiquitin system by ubiquitylation and degradation
Nature Reviews Molecular Cell Biology (2011)