Abstract
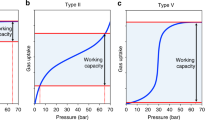
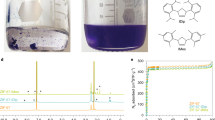
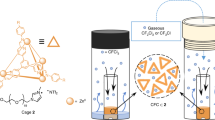
Porous solids such as zeolites1 and metal–organic frameworks2,3 are useful in molecular separation and in catalysis, but their solid nature can impose limitations. For example, liquid solvents, rather than porous solids, are the most mature technology for post-combustion capture of carbon dioxide because liquid circulation systems are more easily retrofitted to existing plants. Solid porous adsorbents offer major benefits, such as lower energy penalties in adsorption–desorption cycles4, but they are difficult to implement in conventional flow processes. Materials that combine the properties of fluidity and permanent porosity could therefore offer technological advantages, but permanent porosity is not associated with conventional liquids5. Here we report free-flowing liquids whose bulk properties are determined by their permanent porosity. To achieve this, we designed cage molecules6,7 that provide a well-defined pore space and that are highly soluble in solvents whose molecules are too large to enter the pores. The concentration of unoccupied cages can thus be around 500 times greater than in other molecular solutions that contain cavities8,9,10, resulting in a marked change in bulk properties, such as an eightfold increase in the solubility of methane gas. Our results provide the basis for development of a new class of functional porous materials for chemical processes, and we present a one-step, multigram scale-up route for highly soluble ‘scrambled’ porous cages prepared from a mixture of commercially available reagents. The unifying design principle for these materials is the avoidance of functional groups that can penetrate into the molecular cage cavities.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Wright, P. A. Microporous Framework Solids (Royal Society of Chemistry, 2007)
Cheetham, A. K., Férey, G. & Loiseau, T. Open-framework inorganic materials. Angew. Chem. Int. Edn 38, 3268–3292 (1999)
Kitagawa, S., Kitaura, R. & Noro, S. Functional porous coordination polymers. Angew. Chem. Int. Edn 43, 2334–2375 (2004)
D'Alessandro, D. M., Smit, B. & Long, J. R. Carbon dioxide capture: prospects for new materials. Angew. Chem. Int. Edn 49, 6058–6082 (2010)
O’Reilly, N., Giri, N. & James, S. L. Porous liquids. Chem. Eur. J. 13, 3020–3025 (2007)
Tozawa, T. et al. Porous organic cages. Nature Mater. 8, 973–978 (2009)
Jones, J. T. A. et al. Modular and predictable assembly of porous organic molecular crystals. Nature 474, 367–371 (2011)
Robbins, T. A., Knobler, C. B., Bellew, D. R. & Cram, D. J. A highly adaptive and strongly binding hemicarcerand. J. Am. Chem. Soc. 116, 111–122 (1994)
Chaffee, K. E., Fogarty, H. A., Brotin, T., Goodson, B. M. & Dutasta, J.-P. Encapsulation of small gas molecules by cryptophane-111 in organic solution. 1. Size- and shape-selective complexation of simple hydrocarbons. J. Phys. Chem. A 113, 13675–13684 (2009)
Little, M. A. et al. Synthesis and methane-binding properties of disulfide-linked cryptophane-0.0.0. Angew. Chem. Int. Edn 51, 764–766 (2012)
Pohorille, A. & Pratt, L. R. Cavities in molecular liquids and the theory of hydrophobic solubilities. J. Am. Chem. Soc. 112, 5066–5074 (1990)
Pierotti, R. A. The solubility of gases in liquids. J. Phys. Chem. 67, 1840–1845 (1963)
Pierotti, R. A. A scaled particle theory of aqueous and non-aqueous solutions. Chem. Rev. 76, 717–726 (1976)
Giri, N. et al. Alkylated organic cages: from porous crystals to neat liquids. Chem. Sci. 3, 2153–2157 (2012)
Melaugh, G., Giri, N., Davidson, C. E., James, S. L. & Del Pópolo, M. G. Designing and understanding permanent microporosity in liquids. Phys. Chem. Chem. Phys. 16, 9422–9431 (2014)
Mogensen, O. E. (ed.) Positron Annihilation in Chemistry (Springer Series in Chemical Physics 58, Springer, 1995)
Mahmood, T., Cheng, K. L. & Yean, Y. C. Microanalysis of open spaces in crown ethers by using a novel probe: positron annihilation spectroscopy. In Third International Workshop on Positron and Positronium Chemistry (ed. Jean, Y. C. ) 640 (World Scientific, 1990)
Lannung, A. & Gjaldbæk, J. C. The solubility of methane in hydrocarbons, alcohols, water and other solvents. Acta Chem. Scand. 14, 1124–1128 (1960)
Darwish, N. A., Gasem, K. A. M. & Robinson, R. L. Jr. Solubility of methane in benzene, naphthalene, phenanthrene and pyrene at temperatures from 323 to 433 K and pressures to 11.3 MPa. J. Chem. Eng. Data 39, 781–784 (1994)
Houndonougbo, Y. et al. A combined experimental–computational investigation of methane adsorption and selectivity in a series of isoreticular zeolitic imidazolate frameworks. J. Phys. Chem. C 117, 10326–10335 (2013)
Zhang, J. et al. Porous liquids: a promising class of media for gas separation. Angew. Chem. Int. Edn 54, 932–936 (2015)
Jiang, S. et al. Porous organic molecular solids by dynamic covalent scrambling. Nature Commun. 2, 207 (2011)
Hasell, T. et al. Controlling the crystallization of porous organic cages: molecular analogs of isoreticular frameworks using shape-specific directing solvents. J. Am. Chem. Soc. 136, 1438–1448 (2014)
Chen, L. et al. Separation of rare gases and chiral molecules by selective binding in porous organic cages. Nature Mater. 13, 954–960 (2014)
Jorgensen, W. L., Maxwell, D. S. & Tirado-Rives, J. Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J. Am. Chem. Soc. 118, 11225 (1996)
Roux, B. The calculation of the potential of mean force using computer simulations. Comput. Phys. Commun. 91, 275 (1995)
Kumar, S., Rosenberg, J. M., Bouzida, D., Swendsen,R. H. & Kollman, P. A. Multidimensional free-energy calculations using the weighted histogram analysis method. J. Comput. Chem. 13, 1011 (1992)
Hess, B., Kutzner, C., van der Spoel, D. & Lindahl, E. GROMACS 4: algorithms for highly efficient, load-balanced, and scalable molecular simulation. J. Chem. Theory Comput. 4, 435 (2008)
Nosé, S. A molecular-dynamics methods for simulations in the canonical ensemble. Mol. Phys. 52, 255 (1984)
Hoover, W. G. Canonical dynamics: equilibrium phase-space distributions. Phys. Rev. A 31, 1695 (1985)
Parrinello, M. & Rahman, A. Polymorphic transitions in single crystals: a new molecular dynamics method. J. Appl. Phys. 52, 7182 (1981)
Essmann, U. et al. A smooth particle mesh Ewald method. J. Chem. Phys. 103, 8577 (1995)
Harms, S. et al. Free volume of interphases in model nanocomposites studied by positron annihilation lifetime spectroscopy. Macromolecules 43, 10505 (2010)
Jacquemin, J., Costa Gomes, M. F., Husson, P. & Majer, V. Solubility of carbon dioxide, ethane, methane, oxygen, nitrogen, hydrogen, argon, and carbon monoxide in 1-butyl-3-methylimidazolium tetrafluoroborate between temperatures 283 K and 343 K and at pressures close to atmospheric. J. Chem. Thermodyn. 38, 490 (2006)
Acknowledgements
This work was funded by the Leverhulme Trust (F/00 203/T) and by EPSRC (EP/C511794/1). M.G.D.P. acknowledges financial support from ANPCyT (PICT-2011-2128) and from the EC-H2020, MSCRISE-2014 programme, through project 643998 ENACT. L.P. and M.C.G. acknowledge support from the Contrat d’Objectifs Partagés (CNRS, Blaise Pascal University, and the Auvergne Regional Government, France). A.I.C. acknowledges the European Research Council under the European Union’s Seventh Framework Programme/ERC Grant Agreement no. 321156 for financial support. We thank M. E. Briggs for assistance with the cage syntheses.
Author information
Authors and Affiliations
Contributions
N.G. and R.L.G. synthesized the porous crown cage. M.D.P. and G.M. carried out the molecular simulations. K.R. and T.K. performed the PALS measurements. M.C.G. and L.P. measured the methane gas solubilities for the crown cage porous liquid. R.L.G. and A.I.C. conceived the synthesis of the scrambled porous imine cages. R.L.G synthesized and characterized the scrambled cage porous liquid and measured its gas solubilities. S.L.J. led the project overall and conceived the design of the porous liquid based on the crown-ether cage together with N.G. S.L.J. and A.I.C. led the writing of the manuscript with contributions from all co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Synthetic and analytical methods, with full synthesis details and characterisation. Molecular Simulations, positron annihilation lifetime spectroscopy (PALS), gas solubility and guest selectivities. (PDF 4891 kb)
Guest selectivity in ‘scrambled’ porous liquids
Two batches of porous liquid prepared in vials (200 mg scrambled cage dissolved in 1 mL hexachloropropene) and both were saturated with xenon (5 mins bubbling at 50-60 mL/min), followed by the addition of a stirrer bar. To one sample was added chloroform (16 μL, 1.0 mol. eq. based on cage) and to the other was added 1-t-butyl-3,5-dimethylbenzene (36 μL, 1.0 mol. eq.), in both cases being careful not to mix the solvents. Stirring was started to mix the solvent layers – as can be observed in the video, chloroform displaces the xenon gas whereas the large, bulky solvent does not. (MP4 29264 kb)
Source data
Rights and permissions
About this article
Cite this article
Giri, N., Del Pópolo, M., Melaugh, G. et al. Liquids with permanent porosity. Nature 527, 216–220 (2015). https://doi.org/10.1038/nature16072
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature16072
This article is cited by
-
A porous metal-organic cage liquid for sustainable CO2 conversion reactions
Nature Communications (2023)
-
Enhanced activity for the oxygen reduction reaction in microporous water
Nature Catalysis (2023)
-
Breathing porous liquids based on responsive metal-organic framework particles
Nature Communications (2023)
-
Porous Liquids
Resonance (2023)
-
The synthesis of bi(poly)- and macrocyclic derivatives of trans-diaminocyclohexane (microreview)
Chemistry of Heterocyclic Compounds (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.