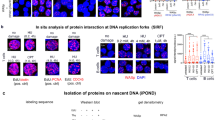
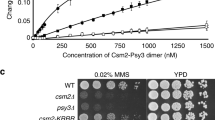
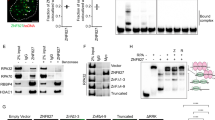
Abstract
Single-strand DNA (ssDNA)-binding proteins (SSBs) are ubiquitous and essential for a wide variety of DNA metabolic processes, including DNA replication, recombination, DNA damage detection and repair1. SSBs have multiple roles in binding and sequestering ssDNA, detecting DNA damage, stimulating nucleases, helicases and strand-exchange proteins, activating transcription and mediating protein–protein interactions. In eukaryotes, the major SSB, replication protein A (RPA), is a heterotrimer1. Here we describe a second human SSB (hSSB1), with a domain organization closer to the archaeal SSB than to RPA. Ataxia telangiectasia mutated (ATM) kinase phosphorylates hSSB1 in response to DNA double-strand breaks (DSBs). This phosphorylation event is required for DNA damage-induced stabilization of hSSB1. Upon induction of DNA damage, hSSB1 accumulates in the nucleus and forms distinct foci independent of cell-cycle phase. These foci co-localize with other known repair proteins. In contrast to RPA, hSSB1 does not localize to replication foci in S-phase cells and hSSB1 deficiency does not influence S-phase progression. Depletion of hSSB1 abrogates the cellular response to DSBs, including activation of ATM and phosphorylation of ATM targets after ionizing radiation. Cells deficient in hSSB1 exhibit increased radiosensitivity, defective checkpoint activation and enhanced genomic instability coupled with a diminished capacity for DNA repair. These findings establish that hSSB1 influences diverse endpoints in the cellular DNA damage response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Primary accessions
Gene Expression Omnibus
Data deposits
The genes hSSB1 and hSSB2 are deposited in NCBI under accession numbers NM_024068 and NM_001031716, respectively.
References
Wold, M. S. Replication protein A: a heterotrimeric single-stranded DNA-binding protein required for eukaryotic DNA metabolism. Annu. Rev. Biochem. 66, 3050–3059 (1997)
West, S. C. Molecular views of recombination proteins and their control. Nature Rev. Mol. Cell Biol. 4, 435–445 (2003)
Khanna, K. K. & Jackson, S. P. DNA double-strand breaks: signaling, repair and the cancer connection. Nature Genet. 27, 247–254 (2001)
Khanna, K. K. et al. ATM associates with and phosphorylates p53: mapping the region of interaction. Nature Genet. 20, 398–400 (1998)
Gatei, M. et al. Role for ATM in DNA damage-induced phosphorylation of BRCA1. Cancer Res. 60, 3299–3304 (2000)
Bekker-Jensen, S. et al. Spatial organization of the mammalian genome surveillance machinery in response to DNA strand breaks. J. Cell Biol. 173, 195–206 (2006)
Pierce, A. J., Johnson, R. D., Thompson, L. H. & Jasin, M. XRCC3 promotes homology-directed repair of DNA damage in mammalian cells. Genes Dev. 13, 2633–2638 (1999)
Rodrigue, A. et al. Interplay between human DNA repair proteins at a unique double-strand break in vivo. EMBO J. 25, 222–231 (2006)
Jazayeri, A. et al. ATM- and cell cycle-dependent regulation of ATR in response to DNA double-strand breaks. Nature Cell Biol. 8, 37–45 (2006)
Soutoglou, E. et al. Positional stability of single double-strand breaks in mammalian cells. Nature Cell Biol. 9, 675–682 (2007)
Lisby, M., Barlow, J. H., Burgess, R. C. & Rothstein, R. Choreography of the DNA damage response: spatiotemporal relationships among checkpoint and repair proteins. Cell 16, 328–334 (2004)
Fabbro, M. et al. BRCA1-BARD1 complexes are required for p53Ser-15 phosphorylation and a G1/S arrest following ionising radiation-induced DNA damage. J. Biol. Chem. 279, 31251–31258 (2004)
Xu, B., Kim, S. T. & Kastan, M. B. Involvement of Brca1 in S-phase and G(2)-phase checkpoints after ionising irradiation. Mol. Cell. Biol. 21, 3445–3450 (2001)
Dodson, G. E., Shi, Y. & Tibbetts, R. S. DNA replication defects, spontaneous DNA damage, and ATM-dependent checkpoint activation in replication protein A-deficient cells. J. Biol. Chem. 279, 34010–34014 (2004)
Mailand, N. et al. Rapid destruction of human Cdc25A in response to DNA damage. Science 288, 1425–1429 (2000)
Lee, J. H. & Paull, T. T. ATM activation by DNA double-strand breaks through the Mre11-Rad50-Nbs1 complex. Science 308, 551–554 (2005)
Wang, Y. et al. Mutation in Rpa1 results in defective DNA double-strand break repair, chromosomal instability and cancer in mice. Nature Genet. 37, 750–755 (2002)
Zhang, J., Ma, Z., Treszezamsky, A. & Powell, S. N. MDC1 interacts with Rad51 and facilitates homologous recombination. Nature Struct. Mol. Biol. 12, 902–909 (2005)
McIlwraith, M. J. et al. Reconstitution of the strand invasion step of double-strand break repair using human RAD51, RAD52 and RPA proteins. J. Mol. Biol. 304, 151–164 (2000)
Song, B. W. & Sung, P. Functional interactions among yeast Rad51 recombinase, Rad52 mediator, and replication protein A in DNA strand exchange. J. Biol. Chem. 275, 15895–15904 (2000)
New, J. H. & Kowalczykowski, S. C. Rad52 protein has a second stimulatory role in DNA strand exchange that complements replication protein-A function. J. Biol. Chem. 277, 26171–26176 (2002)
Sugiyama, T. & Kowalczykowski, S. C. Rad52 protein associates with replication protein A (RPA)-single-stranded DNA to accelerate Rad51-mediated displacement of RPA and presynaptic complex formation. J. Biol. Chem. 277, 31663–31672 (2002)
Wadsworth, R. I. & White, M. F. Identification and properties of the crenarchaeal single-stranded DNA binding protein from Sulfolobus solfataricus. Nucleic Acids Res. 29, 914–920 (2001)
Bartkova, J. et al. DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434, 864–870 (2005)
Aparicio, O. et al. in Current Protocols in Molecular Biology (eds Ausubel, F. A. et al.) 21.3.1–21.3.17 (John Wiley, New York, 2005)
Slavotinek, A., McMillan, T. J. & Steel, C. M. Measurement of radiation survival using the MTT assay. Eur. J. Cancer 30, 1376–1382 (1994)
Pandita, R. K. et al. Mammalian Rad9 plays a role in telomere stability, S- and G2-phase-specific cell survival, and homologous recombinational repair. Mol. Cell. Biol. 26, 1850–1864 (2006)
Acknowledgements
This study was supported by grants from the National Health and Medical Research Council of Australia (to K.K.K.), Cancer Research UK (to S.C.W.), the Biotechnology and Biological Sciences Research Council of the UK (to M.F.W), the National Institutes of Health (CA92245 to J.G.; CA10445 and CA123232 to T.K.P.) and the American Cancer Society (RGS-04-173-01-CCG to T.T.P.).
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary information
The file contains Supplementary Figures S1-S22 with Legends. (PDF 2446 kb)
Rights and permissions
About this article
Cite this article
Richard, D., Bolderson, E., Cubeddu, L. et al. Single-stranded DNA-binding protein hSSB1 is critical for genomic stability. Nature 453, 677–681 (2008). https://doi.org/10.1038/nature06883
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06883
This article is cited by
-
R-loop-dependent promoter-proximal termination ensures genome stability
Nature (2023)
-
SOSSB1 and SOSSB2 mutually regulate protein stability through competitive binding of SOSSA
Cell Death Discovery (2023)
-
Genomic regulation of transcription and RNA processing by the multitasking Integrator complex
Nature Reviews Molecular Cell Biology (2023)
-
Crystal structure of the INTS3/INTS6 complex reveals the functional importance of INTS3 dimerization in DSB repair
Cell Discovery (2021)
-
hSSB2 (NABP1) is required for the recruitment of RPA during the cellular response to DNA UV damage
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.