Abstract
Background:
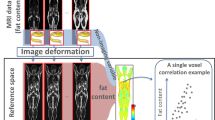
The ability to non-invasively measure body composition in mouse models of obesity and obesity-related disorders is essential for elucidating mechanisms of metabolic regulation and monitoring the effects of novel treatments. These studies aimed to develop a fully automated, high-throughput micro-computed tomography (micro-CT)-based image analysis technique for longitudinal quantitation of adipose, non-adipose and lean tissue as well as bone and demonstrate utility for assessing the effects of two distinct treatments.
Methods:
An initial validation study was performed in diet-induced obesity (DIO) and control mice on a vivaCT 75 micro-CT system. Subsequently, four groups of DIO mice were imaged pre- and post-treatment with an experimental agonistic antibody specific for anti-fibroblast growth factor receptor 1 (anti-FGFR1, R1MAb1), control immunoglobulin G antibody, a known anorectic antiobesity drug (rimonabant, SR141716), or solvent control. The body composition analysis technique was then ported to a faster micro-CT system (CT120) to markedly increase throughput as well as to evaluate the use of micro-CT image intensity for hepatic lipid content in DIO and control mice. Ex vivo chemical analysis and colorimetric analysis of the liver triglycerides were performed as the standard metrics for correlation with body composition and hepatic lipid status, respectively.
Results:
Micro-CT-based body composition measures correlate with ex vivo chemical analysis metrics and enable distinction between DIO and control mice. R1MAb1 and rimonabant have differing effects on body composition as assessed by micro-CT. High-throughput body composition imaging is possible using a modified CT120 system. Micro-CT also provides a non-invasive assessment of hepatic lipid content.
Conclusions:
This work describes, validates and demonstrates utility of a fully automated image analysis technique to quantify in vivo micro-CT-derived measures of adipose, non-adipose and lean tissue, as well as bone. These body composition metrics highly correlate with standard ex vivo chemical analysis and enable longitudinal evaluation of body composition and therapeutic efficacy monitoring.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ogden CL, Carroll MD, Kit BK, Flegal KM . Prevalence of childhood and adult obesity in the United States, 2011-2012. JAMA 2014; 311: 806–814.
Centers for Disease Control and Prevention National Diabetes Fact Sheet: National Estimates and General Information on Diabetes and Prediabetes in the United States. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention: Atlanta, GA, USA, 2011.
Collins S, Martin TL, Surwit RS, Robidoux J . Genetic vulnerability to diet-induced obesity in the C57BL/6 J mouse: physiological and molecular characteristics. Physiol Behav 2004; 81: 243–248.
Tinsley FC, Taicher GZ, Heiman ML . Evaluation of a quantitative magnetic resonance method for mouse whole body composition analysis. Obes Res 2004; 12: 150–160.
Xie D, Cheng H, Hamrick M, Zhong Q, Ding KH, Correa D et al. Glucose-dependent insulinotropic polypeptide receptor knockout mice have altered bone turnover. Bone 2005; 37: 759–769.
Metzinger MN, Miramontes B, Zhou P, Liu Y, Chapman S, Sun L et al. Correlation of X-ray computed tomography with quantitative nuclear magnetic resonance methods for pre-clinical measurement of adipose and lean tissues in living mice. Sensors (Basel) 2014; 14: 18526–18542.
So PW, Yu WS, Kuo YT, Wasserfall C, Goldstone AP, Bell JD et al. Impact of resistant starch on body fat patterning and central appetite regulation. PLoS One 2007; 2: e1309.
Changani KK, Nicholson A, White A, Latcham JK, Reid DG, Clapham JC . A longitudinal magnetic resonance imaging (MRI) study of differences in abdominal fat distribution between normal mice, and lean overexpressers of mitochondrial uncoupling protein-3 (UCP-3). Diabetes Obes Metab 2003; 5: 99–105.
Granton PV, Norley CJ, Umoh J, Turley EA, Frier BC, Noble EG et al. Rapid in vivo whole body composition of rats using cone beam muCT. J Appl Physiol (1985) 2010; 109: 1162–1169.
Luu YK, Lublinsky S, Ozcivici E, Capilla E, Pessin JE, Rubin CT et al. in vivo quantification of subcutaneous and visceral adiposity by micro-computed tomography in a small animal model. Med Eng Phys 2009; 31: 34–41.
Liu X, Harada N, Yamane S, Kitajima L, Uchida S, Hamasaki A et al. Effects of long-term dipeptidyl peptidase-IV inhibition on body composition and glucose tolerance in high fat diet-fed mice. Life Sci 2009; 84: 876–881.
Rubin CT, Capilla E, Luu YK, Busa B, Crawford H, Nolan DJ et al. Adipogenesis is inhibited by brief, daily exposure to high-frequency, extremely low-magnitude mechanical signals. Proc Natl Acad Sci USA 2007; 104: 17879–17884.
Bastie CC, Zong H, Xu J, Busa B, Judex S, Kurland IJ et al. Integrative metabolic regulation of peripheral tissue fatty acid oxidation by the SRC kinase family member Fyn. Cell Metab 2007; 5: 371–381.
Beaucage KL, Xiao A, Pollmann SI, Grol MW, Beach RJ, Holdsworth DW et al. Loss of P2X7 nucleotide receptor function leads to abnormal fat distribution in mice. Purinergic Signal 2014; 10: 291–304.
Fan W, Yanase T, Nomura M, Okabe T, Goto K, Sato T et al. Androgen receptor null male mice develop late-onset obesity caused by decreased energy expenditure and lipolytic activity but show normal insulin sensitivity with high adiponectin secretion. Diabetes 2005; 54: 1000–1008.
Chen Z, Holland W, Shelton JM, Ali A, Zhan X, Won S et al. Mutation of mouse Samd4 causes leanness, myopathy, uncoupled mitochondrial respiration, and dysregulated mTORC1 signaling. Proc Natl Acad Sci USA 2014; 111: 7367–7372.
Ravinet Trillou C, Arnone M, Delgorge C, Gonalons N, Keane P, Maffrand JP et al. Anti-obesity effect of SR141716, a CB1 receptor antagonist, in diet-induced obese mice. Am J Physiol Regul Integr Comp Physiol 2003; 284: R345–R353.
Wu AL, Feng B, Chen MZ, Kolumam G, Zavala-Solorio J, Wyatt SK et al. Antibody-mediated activation of FGFR1 induces FGF23 production and hypophosphatemia. PLoS One 2013; 8: e57322.
Wu AL, Kolumam G, Stawicki S, Chen Y, Li J, Zavala-Solorio J et al. Amelioration of type 2 diabetes by antibody-mediated activation of fibroblast growth factor receptor 1. Sci Transl Med 2011; 3: 113ra126.
Jain A . Fundamentals of Digital Image Processing. Prentice-Hall: Englewood Cliffs, NJ, USA, 1989.
Russ JC . Processing Binary Images. In: The Image Processing Handbook, 2nd edn, CRC Press, Inc.: Boca Raton, FL, USA, 1995. pp 433–439.
Ross R, Leger L, Guardo R, De Guise J, Pike BG . Adipose tissue volume measured by magnetic resonance imaging and computerized tomography in rats. J Appl Physiol (1985) 1991; 70: 2164–2172.
AOAC International. AOAC Official Method 923.03 (Modified). In: Horwitz W (ed). Official Methods of Analysis of AOAC International, 17th edn. AOAC International: Gaithersburg, MD, USA, 2000.
AOAC International. AOAC Official Method 968.06, 992.15 (Modified). In: Horwitz W (ed). Official Methods of Analysis of AOAC International, 17th edn. AOAC International: Gaithersburg, MD, USA, 2000.
AOAC International. AOAC Official Method 950.46 (Modified). In: Horwitz W (ed). Official Methods of Analysis of AOAC International, 17th edn. AOAC International: Gaithersburg, MD, USA, 2000.
AOAC International. AOAC Official Methods 948.22 and 960.39 (Modified). In: Horwitz W (ed): Official Methods of Analysis of AOAC International, 17th edn. AOAC International: Gaithersburg, MD, USA, 2000.
Nagy TR, Clair AL . Precision and accuracy of dual-energy X-ray absorptiometry for determining in vivo body composition of mice. Obes Res 2000; 8: 392–398.
Kodama Y, Ng CS, Wu TT, Ayers GD, Curley SA, Abdalla EK et al. Comparison of CT methods for determining the fat content of the liver. AJR Am J Roentgenol 2007; 188: 1307–1312.
Lubura M, Hesse D, Neumann N, Scherneck S, Wiedmer P, Schurmann A . Non-invasive quantification of white and brown adipose tissues and liver fat content by computed tomography in mice. PLoS One 2012; 7: e37026.
Judex S, Luu YK, Ozcivici E, Adler B, Lublinsky S, Rubin CT . Quantification of adiposity in small rodents using micro-CT. Methods 2010; 50: 14–19.
Lublinsky S, Luu YK, Rubin CT, Judex S . Automated separation of visceral and subcutaneous adiposity in in vivo microcomputed tomographies of mice. J Digit Imaging 2009; 22: 222–231.
Fong TM, Heymsfield SB . Cannabinoid-1 receptor inverse agonists: current understanding of mechanism of action and unanswered questions. Int J Obes (Lond) 2009; 33: 947–955.
Coskun T, Bina HA, Schneider MA, Dunbar JD, Hu CC, Chen Y et al. Fibroblast growth factor 21 corrects obesity in mice. Endocrinology 2008; 149: 6018–6027.
Xu J, Lloyd DJ, Hale C, Stanislaus S, Chen M, Sivits G et al. Fibroblast growth factor 21 reverses hepatic steatosis, increases energy expenditure, and improves insulin sensitivity in diet-induced obese mice. Diabetes 2009; 58: 250–259.
Wei W, Dutchak PA, Wang X, Ding X, Wang X, Bookout AL et al. Fibroblast growth factor 21 promotes bone loss by potentiating the effects of peroxisome proliferator-activated receptor gamma. Proc Natl Acad Sci USA 2012; 109: 3143–3148.
Ogawa Y, Kurosu H, Yamamoto M, Nandi A, Rosenblatt KP, Goetz R et al. BetaKlotho is required for metabolic activity of fibroblast growth factor 21. Proc Natl Acad Sci USA 2007; 104: 7432–7437.
Shimada T, Mizutani S, Muto T, Yoneya T, Hino R, Takeda S et al. Cloning and characterization of FGF23 as a causative factor of tumor-induced osteomalacia. Proc Natl Acad Sci USA 2001; 98: 6500–6505.
Acknowledgements
The authors would like to acknowledge Genentech’s Departments of Antibody Engineering and Bioinformatics and Necropsy Group for their assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
All authors are/were employees of Genentech, a member of the Roche Group, and have/had financial holdings in Genentech/Roche during this work. Genentech/Roche owns all patent rights.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Wyatt, S., Barck, K., Kates, L. et al. Fully-automated, high-throughput micro-computed tomography analysis of body composition enables therapeutic efficacy monitoring in preclinical models. Int J Obes 39, 1630–1637 (2015). https://doi.org/10.1038/ijo.2015.113
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2015.113
This article is cited by
-
Diet-induced obesity in animal models: points to consider and influence on metabolic markers
Diabetology & Metabolic Syndrome (2021)
-
Radiological characterization of gilthead seabream (Sparus aurata) fat by X-ray micro-computed tomography
Scientific Reports (2020)
-
Automated computerized image analysis for the user-independent evaluation of disease severity in preclinical models of NAFLD/NASH
Laboratory Investigation (2020)