Abstract
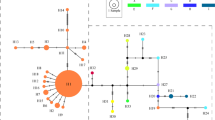
Hierarchical sampling from populations, incipient and recognized varieties within Phacelia dubia and P maculata has revealed high levels of intraspecific polymorphism in chloroplast DNA. Much of the variation is partitioned between populations as evidenced by population-specific variants at fixation in all three populations of P. dubia var. interior and in both populations of P. maculata. Nine of 16 populations were polymorphic for cpDNA haplotypes. A total of 16 haplotypes was found in a sample of 106 individuals; the most common occurred in eight of the 16 populations and in 31 per cent of the individuals in the entire sample. A phylogenetic analysis revealed four basic plastome types. The two major groups of plastomes were separated by four independent base-pair mutations which suggests an ancient split in the evolution of plastid genomes. Representatives from each major plastome division were found in each of five populations spanning two allopatric varieties of P. dubia. The geographical distribution of haplotypes and lack of evidence for recent admixture argue against migration as a source of the polymorphism. It is more likely that the current taxonomic varieties are descendants of a polymorphic common ancestor.
Similar content being viewed by others
Article PDF
References
Avise, J C, Arnold, J, Ball, R M, Bermingham, E, Lamb, T, Neigel, J E, Reeb, C A, and Saunders, N C. 1987. Intraspecific phylogeography: The mitochondrial DNA bridge between population genetics and systematics. Ann Rev Ecol Syst, 18, 489–522.
Avise, J C, Neigel, J E, and Arnold, J. 1984. Demographic influences on mitochondrial DNA lineage survivorship in animal populations. J Mol Evol, 20, 99–105.
Avise, J C, Shapira, J F, Daniel, S W, Aquadro, C F, and Lansman, R A. 1983. Mitochondrial DNA differentiation during the speciation process in Peromyscus. Mol Biol Evol, 1, 38–56.
Ballard, J W O, and Kreitman, M. 1994. Unraveling selection in the mitochondrial genome of Drosophila. Genetics, 138, 757–772.
Banks, J A, and Birky, C W, Jr, 1985. Chloroplast DNA diversity is low in a wild plant, Lupinus texensis. Proc Natl Acad Sci USA, 82, 6950–6954.
Bermingham, E, and Avise, J C. 1986. Molecular zoogeography of freshwater fishes in the southeastern United States. Genetics, 113, 939–965.
Byrne, M, and Moran, G F. 1994. Population divergence in the chloroplast genome of Eucalyptus nitens. Heredity, 73, 18–28.
Desalle, R, Giddings, L V, and Kaneshiro, L Y. 1986. Mitochondrial DNA variability in natural populations of Hawaiian Drosophila. II. Genetic and phylogenetic relationships of natural populations of D. silvestris and D. heteroneura. Heredity, 56, 87–92.
Doyle, J J. 1992. Gene trees and species trees: Molecular systematics as one-character taxonomy. Syst Bot, 17, 144–163.
Ennos, R A. 1994. Estimating the relative rates of pollen and seed migration among plant populations. Heredity, 74, 250–259.
Gillett, G W. 1964. Genetic barriers in the Cosmanthus Phacelias. Rhodora, 66, 359–366.
Hamrick, J L, Linhart, Y B, and Mitton, J B. 1979. Relationships between life history characteristics and electrophoretically detectable genetic variation in plants. Ann Rev Ecol Syst, 10, 173–200.
Harris, S A, and Ingram, R. 1991. Chloroplast DNA and biosystematics: the effects of intraspecific diversity and plastid transmission. Taxon, 40, 393–412.
Hong, Y-P, Hipkins, V D, and Strauss, S H. 1993. Chloroplast DNA diversity among trees, populations and species in the California closed-cone pines (Pinus radiata, Pinus muricata and Pinus attenuata). Genetics, 135, 1187–1196.
Hooglander, N, Lumaret, R, and Bos, M. 1993. Inter-intraspecific variation of chloroplast DNA of European Plantago spp. Heredity, 70, 322–334.
Kemble, R J. 1987. A rapid, single leaf, nucleic acid assay for determining the cytoplasmic organelle complement of rapeseed and related Brassica species. Theor Appl Genet, 73, 364–370.
Kochert, G, Halward, T, Branch, W D, and Simpson, C E. 1991. RFLP variability in peanut (Arachis hypogaea L.) cultivars and wild species. Theor Appl Genet, 81, 565–570.
Levy, F. 1991a. Morphological differentiation in Phacelia dubia and P maculata. Rhodora, 93, 11–25.
Levy, F. 1991b. A genetic analysis of reproductive barriers in Phacelia dubia. Heredity, 67, 331–345.
Mason, R J, Holsinger, K E, and Jansen, R K. 1994. Biparental inheritance of the chloroplast genome in Coreopsis (Asteraceae). J Hered, 85, 171–173.
Mayer, M S, and Soltis, P S. 1994. The evolution of serpentine endemics: A chloroplast DNA phylogeny of the Streptanthus glandulosus complex (Cruciferae). Syst Bot, 19, 557–574.
McCauley, D E. 1994. Contrasting the distribution of chloroplast DNA and allozyme polymorphism among local populations of Silene alba: Implications for studies of gene flow in plants. Proc Natl Acad Sci USA, 91, 8127–8131.
Milligan, B G. 1991. Chloroplast DNA diversity within and among populations of Trifolium pratense. Curr Genet, 19, 411–416.
Murdy, W H. 1966. The systeMatics of Phacelia maculata and Phacelia dubia var. georgiana, both endemic to granite outcrop communities. Am J Bot, 53, 1028–1036.
Neale, D B, Saghai-Maroof, M A, Allard, R W, Zhang, Q, and Jorgensen, R A. 1988. Chlloroplast DNA diversity in populations of wild and cultivated barley. Genetics, 120, 1105–1110.
Nei, M. 1986. Stochastic errors in DNA evolution and molecular phylogeny. In: Gershowitz, H., Rucknagel, D. L. and Tashian, R. E. (eds) Evolutionary Perspectives and the New Genetics, pp. 133–147. Alan Liss, New York.
Neigel, J E, and Avise, J C. 1986. Phylogenetic relationships of mitochondrial DNA under various demographic models of speciation. In: Nevo, E. and Karlin, S. (eds) Evolutionary Processes and Theory, pp. 515–534. Academic Press, New York.
Palmer, J D. 1982. Physical and gene mapping of chloroplast DNA from Atriplex triangularis and Cucumis sativa. Nucl Acids Res, 10, 1593–1605.
Palmer, J D. 1985. Comparative arrangement of chloroplast genomes. Ann Rev Genet, 19, 325–354.
Palmer, J D. 1987. Chloroplast DNA evolution and biosystematic uses of chloroplast DNA variation. Am Nat, 130, S6–S29.
Palmer, J D, and Zamir, D. 1982. Chloroplast DNA evolution and phylogenetic relationships in Lycopersicon. Proc Natl Acad Sci USA, 79, 5006–5010.
Palmer, J D, Jansen, R K, Michaels, H J, Chase, M W, and Manhart, J R. 1988. Chloroplast DNA variation and plant phylogeny. Ann Mo Bot Gard, 75, 1180–1206.
Pamilo, P, and Nei, M. 1988. Relationships between gene trees and species trees. Mol Biol Evol, 5, 568–583.
Petit, R J, Kremer, A, and Wagner, D B. 1994. Geographic structure of chloroplast DNA polymorphisms in European oaks. Theor Appl Genet, 87, 122–128.
Rand, D M, Dorfsman, M, and Kann, L M. 1994. Neutral and non-neutral evolution of Drosophila mitochondrial DNA. Genetics, 138, 741–756.
Scowcraft, W R. 1979. Nucleotide polymorphism in chloroplast DNA of Nicotiana debneyi. Theor Appl Genet, 55, 133–137.
Slatkin, M. 1985. Rare alleles as indicators of gene flow. Evolution, 39, 53–65.
Soltis, D E, Soltis, P S, Kuzoff, R K, and Tucker, T L. 1992. Geographic structuring of chloroplast DNA genotypes in Tiarella trifoliata (Saxifragaceae). Pl Syst Evol, 181, 203–216.
Swofford, D L. 1991. PAUP: Phylogenetic Analysis Using Parsimony, version 3.0. Computer program distributed by the Illinois Natural History Survey.
Tajima, F. 1983. Evolutionary relationship of DNA sequences in finite populations. Genetics, 105, 437–460.
Terauchi, R, Terauchi, T, and Tsunewaki, K. 1991. Intraspecific variation of chloroplast DNA in Dioscorea bulbifera L. Theor Appl Genet, 81, 461–470.
Weir, B S. 1990. Genetic Data Analysis. Sinauer, Sunderland, MA.
Wolfe, K H, Li, W-H, and Sharp, P M. 1987. Reates of nucleotide substitution vary greatly among plant mitochondrial, chloroplast, and nuclear DNAs. Proc Natl Acad Sci USA, 84, 9054–9058.
Wolff, K, and Schaal, B. 1992. Chloroplast DNA variation within and among five Plantago species. J Evol Biol, 5, 325–344.
Acknowledgements
We thank B. Anderson, B. Burkhardt, L. Harris, J. P. Hosier, P. Liu, K. MacElwaine and D. Robertson for helpful advice and technical assistance. B. Weir kindly supplied a computer program to estimate coancestry. The manuscript benefited from M. Uyenoyama's insightful comments and the suggestions of two anonymous reviewers. This research was supported in part by NIH grant no. 5 T32 GM07754-09, NSF grant no. 333-0861 and a Grant-in-aid-of-Research from Sigma Xi.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Levy, F., Antonovics, J., Boynton, J. et al. A population genetic analysis of chloroplast DNA in Phacelia. Heredity 76, 143–155 (1996). https://doi.org/10.1038/hdy.1996.22
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1996.22
Keywords
This article is cited by
-
Phylogenetic relationships among Japanese flowering cherries (Prunus subgenus Cerasus) based on nucleotide sequences of chloroplast DNA
Plant Systematics and Evolution (2007)
-
Spatial and temporal genetic structure in chloroplast and allozyme markers in Phacelia dubia implicate genetic drift
Heredity (1999)