Abstract
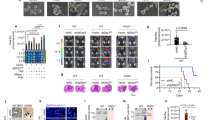
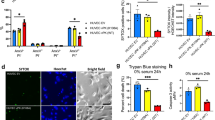
Pathophysiological hypoxia, which fosters the glioma stem-like cell (GSC) phenotype, is present in high-grade gliomas and has been linked to tumor development, invasiveness and resistance to chemotherapy and radiation. Oncolytic virotherapy with engineered herpes simplex virus-1 (HSV-1) is a promising therapy for glioblastoma; however, the efficacy of γ134.5-deleted HSVs, which have been used in clinical trials, was diminished in hypoxia. We investigated the ability of a chimeric human cytolomegalovirus (HCMV)/HSV-1 virus, which expresses the human CMV protein kinase R evasion gene IRS1 and is in preparation for clinical trials, to infect and kill adult and pediatric patient-derived glioblastoma xenografts in hypoxia and normoxia. Infectivity, cytotoxicity and viral recovery were significantly greater with the chimeric virus compared with the γ134.5-deleted virus, regardless of oxygen tension. The chimeric virus infected and killed CD133+ GSCs similarly to wild-type HSV-1. Increased activation of mitogen-activated protein kinase p38 and its substrate heat-shock protein 27 (Hsp27) was seen after viral infection in normoxia compared with hypoxia. Hsp27 knockdown or p38 inhibition reduced virus recovery, indicating that the p38 pathway has a role in the reduced efficacy of the γ134.5-deleted virus in hypoxia. Taken together, these findings demonstrate that chimeric HCMV/HSV-1 efficiently targets both CD133+ GSCs and glioma cells in hypoxia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
02 April 2015
This article has been corrected since advance online publication and an erratum is also printed in this issue
References
Ries L, Melbert D, Krapcho M, Mariotto A, Miller B, Feuer E et al SEER Cancer Statistics Review. National Cancer Institute: Bethesda, MD, 2006, pp 1975–2004.
Cohen KJ, Pollack IF, Zhou T, Buxton A, Holmes EJ, Burger PC et al. Temozolomide in the treatment of high-grade gliomas in children: a report from the Children’s Oncology Group. Neuro Oncol 2011; 13: 317–323.
Stupp R, Mason WP, Van Den Bent MJ, Weller M, Fisher B, Taphoorn MJ et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 2005; 352: 987–996.
Mineta T, Rabkin SD, Yazaki T, Hunter WD, Martuza RL . Attenuated multi-mutated herpes simplex virus-1 for the treatment of malignant gliomas. Nat Med 1995; 1: 938–943.
Cassady KA . Human cytomegalovirus TRS1 and IRS1 gene products block the double-stranded-RNA-activated host protein shutoff response induced by herpes simplex virus type 1 infection. J Virol 2000; 9: 8707–8715.
Shah AC, Parker JN, Gillespie GY, Lakeman FD, Meleth S, Markert JM et al. Enhanced antiglioma activity of chimeric HCMV/HSV-1 oncolytic viruses. Gene Therapy 2007; 14: 1045–1054.
Markert JM, Medlock MD, Rabkin S, Gillespie G, Todo T, Hunter W et al. Conditionally replicating herpes simplex virus mutant, G207 for the treatment of malignant glioma: results of a phase I trial. Gene Therapy 2000; 7: 867–874.
Rampling R, Cruickshank G, Papanastassiou V, Nicoll J, Hadley D, Brennan D et al. Toxicity evaluation of replication-competent herpes simplex virus (ICP 34.5 null mutant) in patients with recurrent malignant glioma. Gene Therapy 2000; 7: 859–866.
Friedman GK, Haas MC, Kelly VM, Markert JM, Gillespie GY, Cassady KA . Hypoxia moderates γ(1)34.5-deleted herpes simplex virus oncolytic activity in human glioma Xenoline primary culture. Transl Oncol 2012; 5: 200–207.
Evans SM, Judy KD, Dunphy I, Jenkins WT, Nelson PT, Collins R et al. Comparative measurements of hypoxia in human brain tumors using needle electrodes and EF5 binding. Cancer Res 2004; 64: 1886–1892.
Evans SM, Judy KD, Dunphy I, Jenkins WT, Hwang WT, Nelson PT et al. Hypoxia is important in the biology and aggression of human glial brain tumors. Clin Cancer Res 2004; 10: 8177–1884.
Jensen RL . Brain tumor hypoxia: tumorigenesis, angiogenesis, imaging, pseudoprogression, and as a therapeutic target. J Neurooncol 2009; 92: 317–335.
McCord AM, Jamal M, Shankavaram UT, Lang FF, Camphausen K, Tofilon PJ . Physiologic oxygen concentration enhances the stem-like properties of CD133+ human glioblastoma cells in vitro. Mol Cancer Res 2009; 7: 489–497.
Heddleston JM, Li Z, McLendon RE, Hjelmeland AB, Rich JN . The hypoxic microenvironment maintains glioblastoma stem cells and promotes reprogramming towards a cancer stem cell phenotype. Cell Cycle 2009; 8: 3274–3284.
Zachos G, Koffa M, Preston CM, Clements JB, Conner J . Herpes simplex virus type 1 blocks the apoptotic host cell defense mechanisms that target Bcl-2 and manipulates activation of p38 mitogen-activated protein kinase to improve viral replication. J Virol 2001; 75: 2710–2712.
Mathew SS, Della Selva MP, Burch AD . Modification and reorganization of the cytoprotective cellular chaperone Hsp27 during herpes simplex virus type 1 infection. J Virol 2009; 83: 9304–9312.
Friedman GK, Langford C, Coleman J, Cassady KA, Parker JN, Markert JM et al. Engineered herpes simplex virus efficiently infects and kills CD133+ glioma cells that express CD111. J Neurooncol 2009; 95: 199–209.
Krummenacher C, Baribaud F, Ponce de Leon M, Baribaud I, Whitbeck JC, Xu R et al. Comparative usage of herpesvirus entry mediator A and nectin-1 by laboratory strains and clinical isolates of herpes simplex virus. Virology 2004; 322: 286–299.
Cowan KJ, Storey KB . Mitogen-activated protein kinases: new signaling pathways functioning in cellular responses to environmental stress. J Exp Biol 2003; 206: 1107–1115.
Wada T, Penninger JM . Mitogen-activated protein kinases in apoptosis regulation. Oncogene 2004; 23: 2838–2849.
Haddad JJ, Hanbali LB . Hypoxia upregulates MAPK(p38)/MAPK(ERK) phosphorylation in vitro: neuroimmunological differential time-dependent expression of MAPKs. Protein Pept Lett 2014; 21: 444–451.
Yoshino Y, Aoyagi M, Tamaki M, Duan L, Morimoto T, Ohno K . Activation of p38 MAPK and/or JNK contributes to increased levels of VEGF secretion in human malignant glioma cells. Int J Oncol 2006; 29: 981–987.
Levin VA, Panchabhai S, Shen L, Baggerly KA . Protein and phosphoprotein levels in glioma and adenocarcinoma cell lines grown in normoxia and hypoxia in monolayer and three-dimensional cultures. Proteome Sci 2012; 10: 5.
Gillis PA, Okagaki LH, Rice SA . Herpes simplex virus type 1 ICP27 induces p38 mitogen-activated protein kinase signaling and apoptosis in HeLa cells. J Virol 2009; 83: 1767–1777.
Zachos G, Clements B, Conner J . Herpes simplex virus type 1 infection stimulates p38/c-Jun N-terminal mitogen-activated protein kinase pathways and activates transcription factor AP-1. J Biol Chem 1999; 274: 5097–5103.
Zachos G, Koffa M, Preston CM, Clements JB, Conner J . Herpes simplex virus type 1 blocks the apoptotic host cell defense mechanisms that target Bcl-2 and manipulates activation of p38 mitogen-activated protein kinase to improve viral replication. J Virol 2001; 75: 2710–2728.
Karaca G, Hargett D, McLean TI, Aguilar JS, Ghazal P, Wagner EK et al. Inhibition of the stress-activated kinase, p38, does not affect the virus transcriptional program of herpes simplex virus type 1. Virology 2004; 329: 142–156.
Mezhir JJ, Advani SJ, Smith KD, Darga TE, Poon AP, Schmidt H et al. Ionizing radiation activates late herpes simplex virus 1 promoters via the p38 pathway in tumors treated with oncolytic viruses. Cancer Res 2005; 65: 9479–9484.
Mohr I, Sternberg D, Ward S, Leib D, Mulvey M, Gluzman Y . A herpes simplex virus type 1 gamma34.5 second-site suppressor mutant that exhibits enhanced growth in cultured glioblastoma cells is severely attenuated in animals. J Virol 2001; 75: 5189–5196.
Parker JN, Gillespie GY, Love CE, Randall S, Whitley RJ, Markert JM . Engineered herpes simplex virus expressing IL-12 in the treatment of experimental murine brain tumors. Proc Natl Acad Sci USA 2000; 97: 2208–2213.
Acknowledgements
Funding was provided by the St Baldrick’s Foundation, The Rally Foundation, Hyundai Hope on Wheels and Kaul Pediatric Research Institute to GKF, as well as the National Institutes of Health (NIH) (CA071933, CA097247 and CA151129). We thank Enid Keyser and the Analytic and Preparative Core Facility (supported by P30 AR048311 and P30 AI027767) for assistance with FACS analysis and Drs Darell Bigner, Jann Sarkaria and C David James for sharing the human xenograft lines with us for these studies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Markert and Dr Gillespie are founders of and own stock and stock options (<7% interest) in Catherex Inc. and in Aettis Inc., biotechnology companies that are developing other oncolytic HSV that are not the subject of this current investigation. They serve as consultants for Catherex Inc. as well. Dr Gillespie currently serves as one of five unpaid members of the Board of Directors for Catherex Inc. The virus used in this study is not licensed by either company or the subject of any company plans for clinical use. Dr Gillespie has served as a paid advisor to the Program Project at the Ohio State University that seeks to find improved methods for the application of oncolytic HSV to treat localized and metastatic cancers. This is generally, but not specifically, related to the subject matter of this investigation.
Rights and permissions
About this article
Cite this article
Friedman, G., Nan, L., Haas, M. et al. γ134.5-deleted HSV-1-expressing human cytomegalovirus IRS1 gene kills human glioblastoma cells as efficiently as wild-type HSV-1 in normoxia or hypoxia. Gene Ther 22, 348–355 (2015). https://doi.org/10.1038/gt.2014.107
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2014.107
This article is cited by
-
Antitumor effects of IL-12 and GM-CSF co-expressed in an engineered oncolytic HSV-1
Gene Therapy (2021)
-
Improving antitumor efficacy via combinatorial regimens of oncolytic virotherapy
Molecular Cancer (2020)
-
Oncolytic herpes simplex virus immunotherapy for brain tumors: current pitfalls and emerging strategies to overcome therapeutic resistance
Oncogene (2019)
-
Pediatric cancer gone viral. Part I: strategies for utilizing oncolytic herpes simplex virus-1 in children
Molecular Therapy - Oncolytics (2015)
-
Pediatric cancer gone viral. Part II: potential clinical application of oncolytic herpes simplex virus-1 in children
Molecular Therapy - Oncolytics (2015)