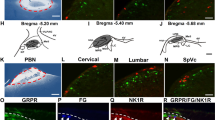
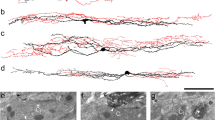
Abstract
The excitatory neurotransmitter glutamate coexists with the peptide known as substance P in primary afferents that respond to painful stimulation1. Because blockers of glutamate receptors reliably reduce pain behaviour2,3,4, it is assumed that ‘pain’ messages are mediated by glutamate action on dorsal horn neurons. The contribution of substance P, however, is still unclear. We have now disrupted the mouse preprotachykinin A gene (PPT-A), which encodes substance P and a related tachykinin, neurokinin A (ref. 5). We find that although the behavioural response to mildly painful stimuli is intact in these mice, the response to moderate to intense pain is significantly reduced. Neurogenic inflammation, which results from peripheral release of substance P and neurokinin A (ref. 6), is almost absent in the mutant mice. We conclude that the release of tachykinins from primary afferent pain-sensing receptors (nociceptors) is required to produce moderate to intense pain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
De Biasi, S. & Rustioni, A. Glutamate and substance P coexist in primary afferent terminals in the superficial laminae of spinal cord. Proc. Natl Acad. Sci. USA 85, 7820–7824 (1988).
Meller, S. T., Dykstra, C. L. & Gebhart, G. F. Acute mechanical hyperalgesia is produced by coactivation of AMPA and metabotropic glutamate receptors. Neuroreport 4, 879–892 (1993).
Wilcox, G. L. in Proceedings of the VIth World Congress on Pain(eds Bond, M. R., Charlton, J. E. & Woolf, C. J.) 97–111 (Elsevier Science, New York, (1991)).
Hunter, J. C. & Singh, L. Role of excitatory amino acid receptors in the mediation of the nociceptive response to formalin in the rat. Neurosci. Lett. 174, 217–221 (1994).
Nawa, H., Kotani, H. & Nakanishi, S. Tissue-specific generation of two preprotachykinin mRNAs from one gene by alternative RNA splicing. Nature 312, 729–734 (1984).
Amann, R., Schuligoi, R., Holzer, P. & Donnerer, J. The non-peptide NK1 receptor antagonist SR140333 produces long-lasting inhibition of neurogenic inflammation, but does not influence acute chemo- or thermonociception in rats. Naunyn Schmied. Arch. Pharmacol. 352, 201–205 (1995).
Rusin, K. I., Ryu, P. D. & Randic, M. Modulation of excitatory amino acid responses in rat dorsal horn neurons by tachykinins. J. Neurophysiol. 68, 265–286 (1992).
Marvizon, J. C. G., Martinez, V., Grady, E. F., Bunnett, N. W. & Mayer, E. A. Neurokinin 1 receptor internalization in spinal cord slices induced by dorsal root stimulation is mediated by NMDA receptors. J. Neurosci. 17, 8129–8136 (1997).
Duggan, A. W., Riley, R. C., Mark, M. A., MacMillan, S. J. A. & Schaible, H. G. Afferent volley patterns and the spinal release of immunoreactive substance P in the dorsal horn of the anaesthetized spinal cat. Neuroscience 65, 849–858 (1995).
Abbadie, C., Trafton, J., Liu, H., Mantyh, P. W. & Basbaum, A. I. Inflammation increases the distribution of dorsal horn neurons that internalize the neurokinin-1 receptor in response to noxious and non-noxious stimulation. J. Neurosci. 17, 8049–8060 (1997).
Mantyh, P. W. et al. Receptor endocytosis and dendrite reshaping in spinal neurons after somatosensory stimulation. Science 268, 1629–1632 (1995).
Maggi, C. A. The mammalian tachykinin receptors. Gen. Pharmacol. 26, 911–944 (1995).
Gyires, K. & Torma, Z. The use of the writhing test in mice for screening different types of analgesics. Arch. Int. Pharmacodyn. 267, 131–140 (1984).
Lawson, S. N., Crepps, B. A. & Perl, E. R. Relationship of substance P to afferent characteristics of dorsal root ganglion neurons in guinea-pig. J. Physiol. (Lond.) 505, 177–191 (1997).
Hylden, J. L., Noguchi, K. & Ruda, M. A. Neonatal capsaicin treatment attenuates spinal Fos activation and dynorphin gene expression following peripheral tissue inflammation and hyperalgesia. J.Neurosci. 12, 1716–1725 (1992).
Malmberg, A. B., Chen, C., Tonegawa, S. & Basbaum, A. I. Preserved acute pain and reduced neuropathic pain in mice lacking PKCγ. Science 278, 279–283 (1997).
Safieh-Garabedian, B., Poole, S., Allchorne, A., Winter, J. & Woolf, C. J. Contribution of interleukin-1 beta to the inflammation-induced increase in nerve growth factor levels and inflammatory hyperalgesia. Br. J. Pharmacol. 115, 1265–1275 (1995).
Rupniak, N. M. J., Carlson, E., Boyce, S., Webb, J. K. & Hill, R. G. Enantioselective inhibition of the formalin paw late phase by the NK1 receptor antagonist L-733,060 in gerbils. Pain 67, 189–195 (1996).
Laneuville, O., Dorais, J. & Couture, R. Characterization of the effects produced by neurokinins and three agonists selective for neurokinin receptor subtypes in a spinal nociceptive reflex of the rat. Life Sci. 42, 1295–1305 (1988).
Munro, F. E., Fleetwood-Walker, S. M., Parker, R. M. & Mitchell, R. The effects of neurokinin receptor antagonists on mustard oil-evoked activation of rat dorsal horn neurons. Neuropeptides 25, 299–305 (1993).
Herrero, J. F. et al. Lack of “wind-up” of somatic nociceptive reflexes and persistence of visceral nociception in a NK1 receptor knock-out mouse. Neurosci. Abstr. 23, 2354 (1997).
De Felipe, C. et al. Characterization of the NK-1 receptor knockout mouse: alterations in nociceptive behavior. Neurosci. Abstr. 23, 2354 (1997).
Kako, K., Munekata, E., Hosaka, M., Murakami, K. & Nakayama, K. Cloning and sequence analysis of mouse cDNAs encoding preprotachykinin A and B. Biomed. Res. 14, 253–259 (1993).
Li, Y. et al. Dilated cardiomyopathy and neonatal lethality in muatant mice lacking manganese superoxide dismutase. Nature Genet. 11, 376–381 (1995).
Vigna, S. R. et al. Characterization of antibodies to the rat substance P (NK-1) receptor and to a chimeric substance P receptor expressed in mammalian cells. J. Neurosci. 14, 834–845 (1994).
Mantyh, P. W., Gates, T., Mantyh, C. R. & Maggio, J. E. Autoradiographic localization and characterization of tachykinin receptor binding sites in the rat brain and peripheral tissues. J.Neurosci. 9, 258–279 (1989).
Hargreaves, K., Dubner, R., Brown, F., Flores, C. & Joris, J. Anew and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 32, 77–88 (1988).
Chaplan, S. R., Bach, F. W., Pogrel, J. W., Chung, J. M. & Yaksh, T. L. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Meth. 53, 55–63 (1994).
Zimmer, A. et al. Hypoalgesia in mice with a targeted deletion of the tachyknin 1 gene. Proc. Natl Acad. Sci. (USA) 95, 2630–2635 (1998).
Acknowledgements
We thank A. Doupe, D. Julius, J. Levine, S. Lisberger, A. Malmberg and W. Martin for critical comments on the manuscript. R. Montenson for the pNTK vector and S. Vigna for antisera against the NK-1 receptor. This work was supported by the NINDS, NIDR, NIDK of the NIH and an RRP from HHMI.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cao, Y., Mantyh, P., Carlson, E. et al. Primary afferent tachykinins are required to experience moderate to intense pain. Nature 392, 390–394 (1998). https://doi.org/10.1038/32897
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/32897
This article is cited by
-
Cracking the Code: The Role of Peripheral Nervous System Signaling in Fracture Repair
Current Osteoporosis Reports (2024)
-
Immune sensing of food allergens promotes avoidance behaviour
Nature (2023)
-
Hallmarks of peripheral nerve function in bone regeneration
Bone Research (2023)
-
C. difficile intoxicates neurons and pericytes to drive neurogenic inflammation
Nature (2023)
-
The Circadian Clocks, Oscillations of Pain-Related Mediators, and Pain
Cellular and Molecular Neurobiology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.