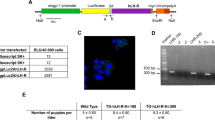
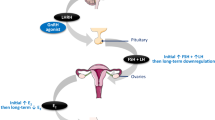
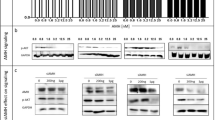
Abstract
About one-third of human breast cancers require hormones for their continued growth1, and endocrine ablation or anti-hormone therapy can cause regression of these tumours. As a consequence, ovariectomy in premenopausal women or administration of an anti-oestrogen (tamoxifen) in postmenopausal women represent major options for treatment of metastatic breast cancer. Alternatively, chronic administration of agonistic analogues of luteinizing hormone-releasing hormone (LHRH)2 causes regression of mammary tumours in experimental animals3–6, and such treatment has shown promise in a small series of premenopausal women with advanced breast cancer7. It has been assumed that these results were achieved by suppressing the pituitary–ovarian axis, as the treatment causes a reduction in circulating levels of gonadal steroids similar to that produced by castration6–8. However, LHRH agonists can exert major effects on tissues other than the pituitary in animals9,10 and in the human11–15. Such findings, coupled with reports of LHRH in human breast milk16 and immunohistochemical evidence for the presence of LHRH-like activity in some human breast tumours17, prompted us to test whether LHRH agonists could have direct antitumour effects. We now report major direct effects of LHRH and its agonists on the growth of breast tumour cells in culture.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Henderson, I. C. & Canellos, G. P. New Engl. J. Med. 302, 17–30; 78–90 (1980).
Fraser, H. M. Nature 296, 391–392 (1982).
Corbin, A., Rosanoft, E. & Bex, F. J. in Hormones and Cancer Vol. 14 (eds lacobelli, S., King, R. J. B., Lindner, H. R. & Lippman, M. E.) 533–540 (Raven, New York, 1980).
Desombre, E. R., Johnson, E. S. & White, W. F. Cancer Res. 36, 3830–3833 (1976).
Danguy, Z. et al. Eur. J. Cancer 13, 1089–1094 (1977).
Nicholson, R. I. & Maynard, P. V. Br. J. Cancer 39, 268–273 (1979).
Klijn, J. G. M. & De Jong, F. H. Lancet i, 1213–1216 (1982).
Robin, D. & McNeil, L. W. J. clin. Endocr. Metab. 51, 873–876 (1980).
Hsueh, A. J. W. & Jones, P. B. C. Endocr. Rev. 2, 437–461 (1981).
Sharpe, R. M. J. Reprod. Fert. 64, 517–527 (1982).
Tureck, R. W., Mastroianni, L. Jr, Blasco, L. & Strauss, J. F. J. clin. Endocr. Metab. 54, 1078–1080 (1982).
Lamberts, S. W. J. et al. J. clin. Endocr. Metab. 54, 450–453 (1982).
Popkin, R., Bramley, T. A., Currie, A. J., Sharpe, R. M. & Fraser, H. M. in LHRH and its Analogues—Fertility and Antifertility Aspects (ed. Schmidt-Gollwitzer, M.) (de Gruyter, Berlin, in the press).
Tan, L. & Rousseau, P. Biochem. biophys. Res. Commun. 109, 1061–1071 (1983).
Khodr, G. & Siler-Khodr, T. M. Fert. Steril. 30, 301–304 (1978).
Amarant, T., Fridkin, M. & Koch, Y. Eur. J. Biochem. 127, 647–650 (1982).
Seppala, M. & Wahlstrom, T. Int. J. Cancer 26, 267–268 (1980).
Lippman, M. E. & Bolan, G. Nature 256, 592–593 (1975).
Lippman, M., Bolan, G. & Huff, K. Cancer Res. 36, 4595–4601, 4610–4618 (1976).
Lippman, M. E., Monaco, M. E. & Bolan, G. Cancer Res. 37, 1901–1907 (1977).
Sharpe, R. M. & Fraser, H. M. Biochem. biophys. Res. Commun. 95, 256–262 (1980).
Currie, A. J., Fraser, H. M. & Sharpe, R. M. Biochem. biophys. Res. Commun. 99, 332–338 (1981).
Popkin, R. et al. Biochem. biophys. Res. Commun. 14, 750–756 (1983).
Belisle, S., Guevin, J.-F., Bellabarba, D. & Lehoux, J.-G. J. clin. Endocr. Metab. 59, 119–126 (1984).
Butzow, R. Int. J. Cancer. 29, 9–11 (1982).
Siler-Khodr, T. M., Khodr, G. S., Vickery, B. H. & Nestor, J. J. Life Sci. 32, 2741–2745 (1983).
Sundaram, K. et al. Life Sci. 28, 83–91 (1981).
Pedroza, E., Vilchez-Martinez, J. A., Coy, D. H., Arimura, A. & Schally, A. V. Biochem. biophys. Res. Commun. 95, 1056–1062 (1980).
Benz, C. et al. Cancer Res. 43, 5298–5303 (1983).
Corbin, A. Yale J. biol. Med. 55, 27–47 (1982).
Vickery, B. H., Nestor, J. J. Jr & Hafez, E. S. E. (eds) LHRH and its Analogs, Contraceptive and Clinical Applications (MTP, Lancester, 1984).
Harvey, H. A., Lipton, A. & Max, D. T. in LHRH and its Analogs, Contraceptive and Clinical Applications (eds Vickery, B. H., Nestor, J. J. Jr & Hafez, E. S. E.) 329–335 (MTP, Lancaster, 1984).
Kent, J. S. et al. Contracept. Deliv. Syst. 3, 58–68 (1982).
Furr, B. J. A. & Hutchinson, F. G. in LHRH and its Analogues (ed. Labrie, F.) (Excerpta Medica, Amsterdam, in the press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Miller, W., Scott, W., Morris, R. et al. Growth of human breast cancer cells inhibited by a luteinizing hormone-releasing hormone agonist. Nature 313, 231–233 (1985). https://doi.org/10.1038/313231a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/313231a0
This article is cited by
-
GNRH family genes contributed to gender-specific disparity of bladder cancer prognosis through exerting opposite regulatory roles between males and females
Journal of Cancer Research and Clinical Oncology (2023)
-
Development of a 68Ga-peptide tracer for PET GnRH1-imaging
Annals of Nuclear Medicine (2016)
-
Gonadotropin-releasing hormone receptor activates GTPase RhoA and inhibits cell invasion in the breast cancer cell line MDA-MB-231
BMC Cancer (2012)
-
Preparation and biodistribution of [67Ga]-DTPA-gonadorelin in normal rats
Journal of Radioanalytical and Nuclear Chemistry (2008)
-
GnRH and LHRgene variants predict adverse outcome in premenopausal breast cancer patients
Breast Cancer Research (2007)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.