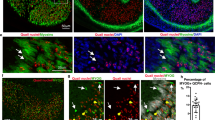
Abstract
Blastemal cells arise after the amputation of limbs or tails in urodele amphibians. These histologically undifferentiated mesenchymal cells divide and subsequently differentiate to regenerate a new appendage. Various studies (reviewed in ref. 1) indicate that blastemal cells arise from tissues near the site of amputation, including muscle, cartilage, nerve and dermis. The multinucleated myofibre, however, is a controversial source of blastemal cells2. The suggestion that myofibres can dedifferentiate is based on their histological appearance during the early stages of limb regeneration3–5. This is contrary to the widely accepted view of muscle regeneration in higher vertebrates which attributes it to satellite cells6,7. One prediction of the dedifferentiation hypothesis is that a population with properties of both myofibres and blastema cells should be present during the early stages of regeneration. Here we describe the isolation of two monoclonal antibodies, one that recognizes an antigen found only in myofibres and another that recognizes an antigen restricted to blastemal cells. By using these antibodies as cell markers, we can detect a small population of cells in the regenerating limbs of adult newts that bear both the myofibre and blastemal cell antigens. The time and location of these double-labelled cells supports the idea that blastemal cells originate, in part, by dedifferentiation of myofibres.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Wallace, H. Vertebrate Limb Regeneration (Wiley, Chichester, 1981).
Hay, E. D. in Regeneration of Striated Muscle and Myogenesis (eds Mauro, A., Shafiq, S. & Milhorat, A. T.) 3–24 (Excerpta Medica, Amsterdam, 1970).
Thornton, C. S. J. Morph. 62, 17 (1938).
Hay, E. D. Devl Biol. 1, 555 (1959).
Lentz, T. L. Am. J. Anat. 124, 447 (1969).
Bischoff, R. in Muscle Regeneration (ed. Mauro, A.) 13–29 (Raven, New York, 1979).
Konigsberg, U. R., Lipton, B. H. & Konigsberg, I. R. Devl Biol. 45, 260 (1975).
Iten, L. E. & Byrant, S. V. Wilhelm Roux Arch. EntwMech. Org. 173, 263 (1973).
Hay, E. D. & Doyle, C. M. Anat. Rec. 175, 339 (1973).
Popiela, H. J. exp. Zool. 198, 57 (1976).
Moore, H-P.H., Fritz, L. C., Raftery, M. A. & Brockes, J. P. Proc. natn. Acad. Sci. U.S.A. 79, 1673 (1982).
McLean, I. W. & Nakane, P. K. J. Histochem. Cytochem. 22, 1077 (1974).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kintner, C., Brockes, J. Monoclonal antibodies identify blastemal cells derived from dedifferentiating muscle in newt limb regeneration. Nature 308, 67–69 (1984). https://doi.org/10.1038/308067a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/308067a0
This article is cited by
-
Mesodermal origin of median fin mesenchyme and tail muscle in amphibian larvae
Scientific Reports (2015)
-
Notochord-derived hedgehog is essential for tail regeneration in Xenopus tadpole
BMC Developmental Biology (2014)
-
Genome-wide analysis of gene expression during Xenopus tropicalis tadpole tail regeneration
BMC Developmental Biology (2011)
-
Xenopus Tbx6 mediates posterior patterning via activation of Wnt and FGF signalling
Cell Research (2006)
-
brinker is a target of Dpp in Drosophila that negatively regulates Dpp-dependent genes
Nature (1999)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.