Abstract
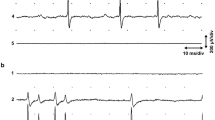
A nerve process grows by inserting new membrane material at its advancing tip, the growth cone1. In embryonic cell culture and in embryos of Xenopus laevis, many growth cones establish functional synaptic transmission within minutes after contact with muscle cells2,3. The rapidity of synapse formation suggests that the growth cone may have already acquired the appropriate neurotransmitter and the machinery for transmitter release before encountering the target cell. Here, we have used a patch of outside-out embryonic muscle membrane formed with gigaohm seal at the tip of a micropipette as an extracellular probe for the presence of channel-activating substances near the growth cones of the isolated Xenopus embryonic neurones in culture. We report that single-channel activity resembling that of muscle acetylcholine receptor channels was induced when the probe was positioned near the growth cones of 50% of the neurones, suggesting the spontaneous release of acetylcholine (ACh) from these growth cones. The release of material from growth cones may occur as a consequence of the incorporation of new membrane during neurite extension; it may also have a role in the interaction between the growth cone and its immediate environment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bray, D. Nature 244, 93–95 (1973).
Kullberg, R. W., Lentz, T. L. & Cohen, M. W. Devl. Biol. 60, 101–129 (1977).
Kidokoro, Y. & Yeh, E. Proc. natn. Acad. Sci. U.S.A. 79, 6727–6731 (1982).
Horn, R. & Patlak, J. Proc. natn. Acad. Sci. U.S.A. 77, 6930–6934 (1980).
Colquhoun, D. & Hawkes, A. G. Proc. R. Soc. B211, 205–235 (1981).
Brehm, P., Steinbach, J. H. & Kidokoro, Y. Devl. Biol. 91, 93–102 (1982).
Weldon, P. R. & Cohen, M. W. J. Neurocytol. 8, 239–259 (1979).
Nakajima, Y., Kidokoro, Y. & Klier, F. G. Devl Biol. 77, 52–72 (1980).
Landis, S. C. J. Cell Biol. 78, R8–R14 (1978).
Marchbanks, R. M. Biochem. J. 106, 87–95 (1968).
Hume, R. I., Role, L. W. & Fischbach, G. D. Nature 305, 632–634 (1983).
Landis, S. C. A. Rev. Physiol. 45, 567–580 (1983).
Poo, M.-M., Poo, W.-J. H. & Lam, J. W. J. Cell Biol. 76, 483–501 (1978).
Young, S. H. & Poo, M.-M. Nature 304, 161–163 (1983).
Hamill, O., Marty, A., Neher, E., Sakmann, B. & Sigworth, F. J. Pflügers Arch. ges. Physiol. 391, 85–100 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Young, S., Poo, Mm. Spontaneous release of transmitter from growth cones of embryonic neurones. Nature 305, 634–637 (1983). https://doi.org/10.1038/305634a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/305634a0
This article is cited by
-
Cerebellar sub-divisions differ in exercise-induced plasticity of noradrenergic axons and in their association with resilience to activity-based anorexia
Brain Structure and Function (2017)
-
Molecular control of neuromuscular junction development
Journal of Cachexia, Sarcopenia and Muscle (2012)
-
Acute knockdown of AMPA receptors reveals a trans-synaptic signal for presynaptic maturation
The EMBO Journal (2011)
-
An in vivo biosensor for neurotransmitter release and in situ receptor activity
Nature Neuroscience (2010)
-
Vesicular release of glutamate from unmyelinated axons in white matter
Nature Neuroscience (2007)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.