Abstract
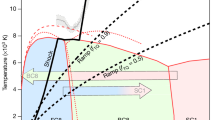
SYNTHESIS of diamond from graphite in the size range <0.1 to 1 mm requires an ultra-autoclave capable of operating at about 100,000 atm and 2000° K (ref. 1). A molten metal solvent-catalyst is also needed so that the conversion may be achieved in a relatively short time (minutes). Catalysts used include chromium, manganese and tantalum plus all elements of Group VIII of the periodic table, but nickel is used most frequently. The cross hatched region of Fig. 1 shows the combinations of pressure and temperature used for diamond synthesis when nickel is the catalyst.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bundy, F. P., Hall, H. T., Strong, H. M., and Wentorf, R. H., Nature, 176, 51 (1955).
Bovenkerk, H. P., Bundy, F. P., Hall, H. T., Strong, H. M., and Wentorf, R. H., Nature, 184, 1094 (1959).
Mayer, J. E., and Shaw, M. C., Lubric. Engng, 13, 21 (1957).
Marshall, E. R., and Shaw, M. C., Trans. ASME, 74, 51 (1952).
Shaw, M. C., Trans. ASME, 70, A-137 (1948).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KOMANDURI, R., SHAW, M. New method of nucleating diamonds. Nature 248, 582–584 (1974). https://doi.org/10.1038/248582a0
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1038/248582a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.