Abstract
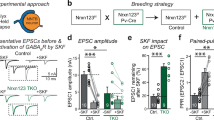
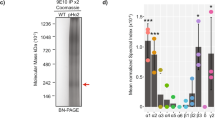
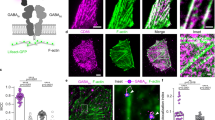
The ionotropic type-A and type-C receptors for the neurotransmitter γ-aminobutyric acid (GABAA and GABAC receptors) are the principal sites of fast synaptic inhibition in the central nervous system1,2,3, but it is not known how these receptors are localized at GABA-dependent synapses. GABAC receptors, which are composed of ρ-subunits3,4,5,6, are expressed almost exclusively inthe retina of adult vertebrates, where they are enriched on bipolar cell axon terminals7,8,9. Here we show that the microtubule-associated protein 1B (MAP-1B) specifically interacts with the GABAC ρ1 subunit but not with GABAA receptor subunits. Furthermore, GABAC receptors and MAP-1B co-localize at postsynaptic sites on bipolar cell axon terminals. Co-expression of MAP-1B and the ρ1 subunit in COS cells results in a dramatic redistribution of the ρ1 subunit. Our observations suggest a novel mechanism for localizing ionotropic GABA receptors to synaptic sites. This mechanism, which is specific for GABAC but not GABAA receptors, may allow these receptor subtypes, which have distinct physiological and pharmacological properties, to be differentially localized at inhibitory synapses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Macdonald, R. L. & Olsen, R. W. GABAAreceptor channels. Annu. Rev. Neurosci. 17, 569–602 (1994).
Rabow, L. E., Russek, S. J. & Farb, D. H. From ion currents to genomic analysis: recent advances in GABAAreceptor research. Synapse 21, 189–274 (1995).
Lukasiewicz, P. D. GABACreceptors in the vertebrae retina. Mol. Neurobiol. 12, 181–194 (1996).
Polenzani, L., Woodward, R. M. & Miledi, R. Expression of mammalian γ-aminobutyric acid receptors with distinct pharmacology in Xenopus. Proc. Natl Acad. Sci. USA 88, 4318–4322 (1991).
Cutting, G. R. et al. Cloning of the γ-aminobutyric acid (GABA) rho1 cDNA: aGABA receptor subunit highly expressed in the retina. Proc. Natl Acad. Sci. USA 88, 2673–2677 (1991).
Wang, T. L., Guggino, W. B. & Cutting, G. R. Anovel γ-aminobutyric acid receptor subunit (rho2) cloned from human retina forms bicuculline-insensitive homooligomeric receptors in Xenopus oocytes. J. Neurosci. 14, 6524–6531 (1994).
Enz, R., Brandstatter, J. H., Hartveit, E., Wassle, H. & Bormann, J. Expression of GABA receptor rho1 and rho2 subunits in the retina and brain of the rat. Eur. J. Neurosci. 7, 1495–1501 (1995).
Enz, R., Brandstatter, J. H., Hartveit, E., Wassle, H. & Bormann, J. Expression of GABA receptor rho1 and rho2 subunits in the retina and brain of the rat. Eur. J. Neurosci. 7, 1495–1501 (1995).
Koulen, P., Brandstatter, J. H., Enz, R. & Wassle, H. Synaptic clustering of GABACreceptor ρ-subunits in the rat retina. Eur. J. Neurosci. 10, 115–127 (1998).
Field, S. & Song, O. Anovel genetic system to detect protein–protein interactions. Nature 340, 245–246 (1989).
Hammarback, J. A. in Brain Microtubule-associated Proteins: Modifications in Diesease (eds Avila, J., Kosik, K. & Brandt, R.) 1–17 (Harwood, Amsterdam, (1997).
Ulloa, L., Avila, A. & Diaz-Nido, R. in Brain Microtubule-associated Proteins: Modifications in Diesease (eds Avila, J., Kosik, K. & Brandt, R.) 17–33 (Harwood, Amsterdam, (1997).
Lien, L., Feener, C. A., Fischbach, N. & Kunkel, L. M. Cloning of human microtubule-associated protein 1B and the identification of a related gene on chromosome 15. Genomics 22, 273–280 (1994).
Noble, M., Lewis, S. A. & Cowan, N. The microtubule binding domain of microtubule-associated protein MAP-1B contains a repeated sequence motif unrelated to that of MAP2 and tau. J. Cell. Biol. 109, 3367–3376 (1989).
Smith, D. B. & Johnson, K. S. Single-step purification of polypeptides expressed in E. coli as fusions with glutathione S-transferase. Gene 67, 31–40 (1988).
Johnstone, M., Goold, R. G., Fischer, I. & Gordon-Weeks, P. R. The neurofilament antibody RT97 recognises a developmentally regulated phosphorylation epitope on microtubule-associated protein 1B. J. Anat. 191, 229–244 (1997).
Gunther, U. et al. Benzodiazepine-insensitive mice generated by targeted disruption of the γ2 subunit of GABAAreceptors. Proc. Natl Acad. Sci. USA 92, 7749–7753 (1995).
Fujii, T., Watanabe, M., Ogoma, Y., Kondo, Y. & Arai, T. Microtubule-associated proteins, MAP 1A and MAP 1B, interact with F-actin in vitro. J. Biochem. 6, 827–829 (1993).
Albrecht, B. E. & Darlison, M. G. Localisation of the ρ1 and ρ2 subunit messenger RNAs in chick retina by in situ hybridization predicts the existence of γ-aminobutyric acid type C receptor subtypes. Neurosci. Letts 189, 155–158 (1995).
Wyszynski, M. et al. Differential regional expression and ultrastructural localization of α-actinin-2, aputative NMDA-receptor-anchoring protein, in rat brain. J. Neurosci. 18, 1383–1392 (1998).
Prior, P. et al. Primary structure and alternative splice variants of gephyrin, a putative glycine receptor-tubulin linker protein. Neuron 8, 1161–1170 (1992).
Tachibana, M. & Kaneko, T. γ-Aminobutyric acid exerts a local inhibitory action on the axon terminal of bipolar cells: evidence for negative feedback from amacrine cells. Proc. Natl Acad. Sci. USA 85, 3501–3505 (1987).
Kirsch, J., Kuhse, J. & Betz, H. Targeting of glycine receptor subunits to gephyrin-rich domains in transfected human embryonic kidney cells. Mol. Cell. Neurosci. 6, 450–461 (1995).
Kirsch, J., Wolters, I., Triller, A. & Betz, H. Gephyrin antisense oligonucleotides prevent glycine-receptor clustering in spinal neurons. Nature 366, 745–748 (1993).
Connolly, C. N., Wooltorton, J. A., Smart, T. G. & Moss, S. J. Subcellular localization of GABAAreceptors is determined by receptor β subunits. Proc. Natl Acad. Sci. USA 93, 9899–9904 (1996).
Connolly, C. N., Krishek, B., McDonald, B. M., Smart, T. G. & Moss, S. J. Assembly and cell surface expression of heteromeric and homomeric γ-aminobutyric acid type A receptors. J. Biol. Chem. 271, 89–96 (1996).
Li, M., Jan, Y. & Jan, L. Specification of subunit assembly by the hydrophilic amino-terminal domain of the Shaker potassium channel. Science 257, 1225–1229 (1992).
Acknowledgements
We thank C.-H. Sung for the retinal cDNA library, D. Attwell for help with retinal cultures, and W. Wisden and T. Smart for their comments on the manuscript. This work was supported by the MRC, the Wellcome Trust and the Royal Society.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hanley, J., Koulen, P., Bedford, F. et al. The protein MAP-1B links GABAC receptors to the cytoskeleton at retinal synapses. Nature 397, 66–69 (1999). https://doi.org/10.1038/16258
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/16258
This article is cited by
-
γ-Aminobutyric acid (GABA) homeostasis regulates pollen germination and polarized growth in Picea wilsonii
Planta (2013)
-
Changes in the Subcellular Distribution of Microtubule-Associated Protein 1B During Synaptogenesis of Cultured Rat Cortical Neurons
Cellular and Molecular Neurobiology (2007)
-
The MAP1 family of microtubule-associated proteins
Genome Biology (2006)
-
Constructing inhibitory synapses
Nature Reviews Neuroscience (2001)
-
GABAA receptor cell surface number and subunit stability are regulated by the ubiquitin-like protein Plic-1
Nature Neuroscience (2001)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.