Abstract
Germline mutations in the fumarate hydratase (FH) gene were recently shown to predispose to the dominantly inherited syndrome, hereditary leiomyomatosis and renal cell cancer (HLRCC). HLRCC is characterized by benign leiomyomas of the skin and the uterus, renal cell carcinoma, and uterine leiomyosarcoma. The aim of this study was to identify new families with FH mutations, and to further examine the tumor spectrum associated with FH mutations. FH germline mutations were screened from 89 patients with RCC, skin leiomyomas or ovarian tumors. Subsequently, 13 ovarian and 48 bladder carcinomas were analyzed for somatic FH mutations. Two patients diagnosed with ovarian mucinous cystadenoma (two out of 33, 6%) were found to be FH germline mutation carriers. One of the changes was a novel mutation (Ala231Thr) and the other one (435insAAA) was previously described in FH deficiency families. These results suggest that benign ovarian tumors may be associated with HLRCC.
Similar content being viewed by others
Introduction
Hereditary leiomyomatosis and renal cell cancer (HLRCC) is a recently described dominantly inherited cancer syndrome characterized by predisposition to cutaneous and uterine leiomyomas, renal cell cancer (RCC), and uterine leiomyosarcomas (ULMS) (HLRCC, OMIM 605839). HLRCC is caused by heterozygous germline mutations in fumarate hydratase (FH, fumarase).1, 2 Leiomyomas of the skin and the uterus are the most common feature of the syndrome with nearly complete penetrance.3 RCC and ULMS have been reported in a subset of mutation-positive families. In addition, other tumor types including breast carcinoma, prostate cancer, bladder carcinoma and skin leiomyosarcoma have been observed in HLRCC families.1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 Homozygous germline mutations in FH also cause a recessive syndrome called FH deficiency. FH deficiency is an inborn error characterized by severe neurological symptoms and developmental delay (OMIM 606812).12, 13
To identify new families with FH mutations and to further examine the tumor spectrum associated with FH mutations, we examined a series of Polish patients displaying papillary RCC, single-skin leiomyomas, ovarian cystadenomas or carcinomas for FH germline mutations. The rationale for including ovarian tumors in the present study was the recent report of an excess of uterine leiomyomas and ovarian cystadenomas in Polish families with hereditary ovarian cancer unassociated with BRCA1 mutations.14 Subsequently, a set of mucinous cystadenocarcinomas was studied for somatic FH mutations.
Although we have previously screened multiple different tumor types for somatic FH mutations, we took the opportunity to examine here also 48 bladder cancers; the rationale for this was recent detection of loss of the wild-type allele in a bladder cancer of a Finnish FH mutation carrier, and a report from UK on a bladder cancer case in an FH mutation-positive individual.3, 9 Although previous work on identification of somatic FH mutations has been largely though not completely negative, the example of frequent mutations of LKB1 in lung adenocarcinomas but few other tumor types emphasizes that thorough evaluation of a large variety of tumor types is warranted when a role of a hereditary cancer gene in somatic tumorigenesis is examined.15, 16
Materials and methods
Patient material
For germline mutation analysis, blood samples from 89 probands were collected. For analysis of somatic FH mutations, ovarian carcinomas and bladder carcinomas were collected. Details of the patient material are summarized in Table 1. Samples were collected following informed consent. DNA extractions from blood or tumor samples were performed using standard procedures.
Mutation analysis
Blood samples and ovarian tumors were analyzed for FH mutations by genomic sequencing. Polymerase chain reaction (PCR) conditions and the oligonucleotide primers used were published earlier by Kiuru et al.17 The PCR products were purified using ExoSAP-IT PCR purification kit (USB Corporation, Cleveland, Ohio, USA) and the sequencing reactions were performed using the Big Dye Terminator v.3.1 kit (Applied Biosystems, Foster City, CA, USA). Electrophoresis was performed on an ABI3730 Automatic DNA sequencer (Applied Biosystems). Bladder tumors were analyzed by denaturing high-performance liquid chromatography (DHPLC) as described earlier.18 Bladder tumor samples showing heteroduplex peaks in DHPLC analyses were reanalyzed by direct sequencing. One hundred and fifty healthy Polish individuals were used as controls.
Results and discussion
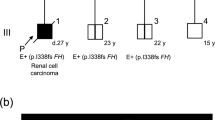
In the FH germline mutation, analysis of 89 probands with P-RCC, skin leiomyomas or ovarian tumors, two mutations were found. Neither of the mutations was found in control samples. Both mutation-positive patients were diagnosed with ovarian mucinous cystadenoma (two out of 33, 6%) (Table 1 and Figure 1). One of the changes was a novel missense substitution 691G>A (Ala231Thr) and the other was a previously reported three-base pair insertion at codon 435 (435insAAA). Unfortunately, ovarian tumor tissues of mutation carriers were not available, and thus, possible biallelic inactivation of FH gene in the tumors could not be examined.
The patient with a missense mutation Ala231Thr was diagnosed with mucinous cystadenoma of the left ovary and cysta endometrialis of the right ovary at the age of 25 years. The patient had no features of HLRCC. The skin was evaluated for cutaneous leiomyomas by a dermatologist, and abdominal ultrasound showed normal kidneys. There was no evidence of family history of HLRCC, but one case of breast cancer and one case of leukemia were observed in her second-degree relatives. The absence of HLRCC phenotype might be due to the patient's relatively young age. The risk of skin and uterine leiomyomas in FH mutation carriers have been proposed to be 28 and 9% at the age of 25 years, respectively. Moreover, the mean age at diagnosis of uterine leiomyomas has been estimated to be 31 years (median, 30 years; range, 19–47 years).3
The 435insAAA carrier was diagnosed with mucinous cystadenoma at the age of 49 years. The patient was also diagnosed with small multiple uterine myomas, but no skin lesions, evaluated by a dermatologist, were observed. Abdominal ultrasound revealed no kidney lesions. Her father was also found to be a mutation carrier but without HLRCC phenotype. A family history of cancer was also negative in this family. However, her FH mutation negative mother and her first-degree relative were diagnosed with ovarian cancer. 435insAAA mutation has been previously reported in four different FH deficiency families.12, 13 Three other missense mutations, K187R, R190C and R190H, have been previously detected in both tumor predisposition and FH deficiency families, and also, phenotypic similarities have been observed between heterozygous parents of FH deficiency patients and HLRCC patients.2
After detecting germline mutations in patients with ovarian mucinous cystadenoma, we investigated the role of FH somatic mutations in a set of ovarian mucinous cystadenocarcinomas. No mutations were found in 13 samples. In addition, as bladder carcinoma has recently been linked to FH mutation-positive families and this tumor type has not been previously analyzed, we studied 48 bladder carcinomas for FH mutations with negative results.3, 9
Taken together, the present study revealed for the first time that germline FH mutations might predispose to benign ovarian lesions. Recently, also other benign tumors including atypical uterine leiomyomas, kidney cysts, and adrenal gland adenomas have been reported in mutation carriers.9 Further studies and increased clinical awareness should clarify which tumor types are indeed causally associated with FH germline mutations.
References
Launonen V, Vierimaa O, Kiuru M et al: Inherited susceptibility to uterine leiomyomas and renal cell cancer. Proc Natl Acad Sci USA 2001; 98: 3387–3392.
Tomlinson IP, Alam NA, Rowan AJ et al: Germline mutations in FH predispose to dominantly inherited uterine fibroids, skin leiomyomata and papillary renal cell cancer. Nat Genet 2002; 30: 406–410.
Alam NA, Barclay E, Rowan AJ et al: Clinical features of multiple cutaneous and uterine leiomyomatosis: an underdiagnosed tumor syndrome. Arch Dermatol 2005; 141: 199–206.
Toro JR, Nickerson ML, Wei MH et al: Mutations in the fumarate hydratase gene cause hereditary leiomyomatosis and renal cell cancer in families in North America. Am J Hum Genet 2003; 73: 95–106.
Martinez-Mir A, Glaser B, Chuang GS et al: Germline fumarate hydratase mutations in families with multiple cutaneous and uterine leiomyomata. J Invest Dermatol 2003; 121: 741–744.
Chuang GS, Martinez-Mir A, Geyer A et al: Germline fumarate hydratase mutations and evidence for a founder mutation underlying multiple cutaneous and uterine leiomyomata. J Am Acad Dermatol 2005; 52: 410–416.
Chan I, Wong T, Martinez-Mir A et al: Familial multiple cutaneous and uterine leiomyomas associated with papillary renal cell cancer. Clin Exp Dermatol 2005; 30: 75–78.
Wei MH, Toure O, Glenn G et al: Novel mutations in FH and expansion of the spectrum of phenotypes expressed in families with hereditary leiomyomatosis and renal cell cancer. J Med Genet 2006; 43: 18–27.
Lehtonen HJ, Kiuru M, Ylisaukko-Oja SK et al: Increased risk of cancer in patients with fumarate hydratase germline mutation. J Med Genet 2006, [E-pub ahead of print].
Chuang GS, Martinez-Mir A, Engler DE, Gmyrek RF, Zlotogorski A, Christiano AM : Multiple cutaneous and uterine leiomyomata resulting from missense mutations in the fumarate hydratase gene. Clin Exp Dermatol 2006; 31: 118–121.
Alam NA, Olpin S, Rowan A et al: Missense mutations in fumarate hydratase in multiple cutaneous and uterine leiomyomatosis and renal cell cancer. J Mol Diagn 2005; 7: 437–443.
Coughlin EM, Christensen E, Kunz PL et al: Molecular analysis and prenatal diagnosis of human fumarase deficiency. Mol Genet Metab 1998; 63: 254–262.
Loeffen J, Smeets R, Voit T, Hoffmann G, Smeitink J : Fumarase deficiency presenting with periventricular cysts. J Inherit Metab Dis 2005; 28: 799–800.
Menkiszak J, Brzosko M, Gorski B et al: Ovarian cystadenoma as a characteristic feature of families with hereditary ovarian cancers unassociated with BRCA1 and BRCA2 mutations. J Appl Genet 2004; 45: 255–263.
Avizienyte E, Loukola A, Roth S et al: LKB1 somatic mutations in sporadic tumors. Am J Pathol 1999; 154: 677–681.
Sanchez-Cespedes M, Parrella P, Esteller M et al: Inactivation of LKB1/STK11 is a common event in adenocarcinomas of the lung. Cancer Res 2002; 62: 3659–3662.
Kiuru M, Lehtonen R, Arola J et al: Few FH mutations in sporadic counterparts of tumor types observed in hereditary leiomyomatosis and renal cell cancer families. Cancer Res 2002; 62: 4554–4557.
Lehtonen R, Kiuru M, Rokman A et al: No fumarate hydratase (FH) mutations in hereditary prostate cancer. J Med Genet 2003; 40: e19.
Acknowledgements
We thank Gynel Arifdshan for excellent technical assistance. The study was supported by grants from the Academy of Finland (44870/Finnish Center of Excellence Program 2000-2005, 76227, 77547, 213183, 214268), the Sigrid Juselius Foundation, the Cancer Society of Finland, and Helsinki University Central Hospital.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ylisaukko-oja, S., Cybulski, C., Lehtonen, R. et al. Germline fumarate hydratase mutations in patients with ovarian mucinous cystadenoma. Eur J Hum Genet 14, 880–883 (2006). https://doi.org/10.1038/sj.ejhg.5201630
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejhg.5201630
Keywords
This article is cited by
-
Class-II dihydroorotate dehydrogenases from three phylogenetically distant fungi support anaerobic pyrimidine biosynthesis
Fungal Biology and Biotechnology (2021)
-
The emerging role and targetability of the TCA cycle in cancer metabolism
Protein & Cell (2018)
-
Leiomyoma Cutis: A Focused Review on Presentation, Management, and Association with Malignancy
American Journal of Clinical Dermatology (2015)
-
Metabolic alteration in tumorigenesis
Science China Life Sciences (2013)
-
Structural basis of fumarate hydratase deficiency
Journal of Inherited Metabolic Disease (2011)