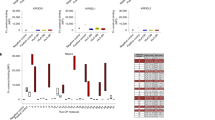
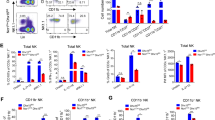
Abstract
Natural killer (NK) cells are crucial in the control of cytomegalovirus infections in mice and humans. Here we show that the viral UL141 gene product has an immunomodulatory function that is associated with low-passage strains of human cytomegalovirus. UL141 mediated efficient protection of cells against killing by a wide range of human NK cell populations, including interferon-α-stimulated bulk cultures, polyclonal NK cell lines and most NK cell clones tested. Evasion of NK cell killing was mediated by UL141 blocking surface expression of CD155, which was previously identified as a ligand for NK cell-activating receptors CD226 (DNAM-1) and CD96 (TACTILE). The breadth of the UL141-mediated effect indicates that CD155 has a key role in regulating NK cell function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Biron, C.A., Byron, K.S. & Sullivan, J.L. Severe herpesvirus infections in an adolescent without natural killer cells. N. Engl. J. Med. 320, 1731–1735 (1989).
Karre, K. How to recognize a foreign submarine. Immunol. Rev. 155, 5–9 (1997).
Carr, W.H., Little, A.M., Mocarski, E. & Parham, P. NK cell-mediated lysis of autologous HCMV-infected skin fibroblasts is highly variable among NK cell clones and polyclonal NK cell lines. Clin. Immunol. 105, 126–140 (2002).
Tomasec, P. et al. Surface expression of HLA-E, an inhibitor of natural killer cells, enhanced by human cytomegalovirus gpUL40. Science 287, 1031–1033 (2000).
Wang, E.C. et al. UL40-mediated NK evasion during productive infection with human cytomegalovirus. Proc. Natl. Acad. Sci. USA 99, 7570–7575 (2002).
Cosman, D. et al. ULBPs, novel MHC class I-related molecules, bind to CMV glycoprotein UL16 and stimulate NK cytotoxicity through the NKG2D receptor. Immunity 14, 123–133 (2001).
Kubin, M. et al. ULBP1, 2, 3: novel MHC class I-related molecules that bind to human cytomegalovirus glycoprotein UL16, activate NK cells. Eur. J. Immunol. 31, 1428–1437 (2001).
Welte, S.A. et al. Selective intracellular retention of virally induced NKG2D ligands by the human cytomegalovirus UL16 glycoprotein. Eur. J. Immunol. 33, 194–203 (2003).
Leong, C.C. et al. Modulation of natural killer cell cytotoxicity in human cytomegalovirus infection: the role of endogenous class I major histocompatibility complex and a viral class I homolog. J. Exp. Med. 187, 1681–1687 (1998).
Dunn, W. et al. Functional profiling of a human cytomegalovirus genome. Proc. Natl. Acad. Sci. USA 100, 14223–14228 (2003).
Cha, T.A. et al. Human cytomegalovirus clinical isolates carry at least 19 genes not found in laboratory strains. J. Virol. 70, 78–83 (1996).
Berstein, D.L. & Plotkin, S.A. Cytomegalovirus vaccines. in New Generation Vaccines (eds. Levine, M.M., Kaper, J.B., Rappuloi, R., Liu, M.A. & Good, M.F.) 649–659 (Marcel Dekker, New York, 2004).
Cerboni, C. et al. Human cytomegalovirus strain-dependent changes in NK cell recognition of infected fibroblasts. J. Immunol. 164, 4775–4782 (2000).
Davison, A.J. et al. The human cytomegalovirus genome revisited: comparison with the chimpanzee cytomegalovirus genome. J. Gen. Virol. 84, 17–28 (2003).
Penfold, M.E. et al. Cytomegalovirus encodes a potent α chemokine. Proc. Natl. Acad. Sci. USA 96, 9839–9844 (1999).
Benedict, C.A. et al. Cutting edge: a novel viral TNF receptor superfamily member in virulent strains of human cytomegalovirus. J. Immunol. 162, 6967–6970 (1999).
Mocarski, E.S., Kemble, G.W., Lyle, J.M. & Greaves, R.F. A deletion mutant in the human cytomegalovirus gene encoding IE1(491aa) is replication defective due to a failure in autoregulation. Proc. Natl. Acad. Sci. USA 93, 11321–11326 (1996).
Dolan, A. et al. Genetic content of wild-type human cytomegalovirus. J. Gen. Virol. 85, 1301–1312 (2004).
Novotny, J., Rigoutsos, I., Coleman, D. & Shenk, T. In silico structural and functional analysis of the human cytomegalovirus (HHV5) genome. J. Mol. Biol. 310, 1151–1166 (2001).
Bottino, C. et al. Identification of PVR (CD155) and Nectin-2 (CD112) as cell surface ligands for the human DNAM-1 (CD226) activating molecule. J. Exp. Med. 198, 557–567 (2003).
Fuchs, A., Cella, M., Giurisato, E., Shaw, A.S. & Colonna, M. Cutting edge: CD96 (tactile) promotes NK cell-target cell adhesion by interacting with the poliovirus receptor (CD155). J. Immunol. 172, 3994–3998 (2004).
Bernhardt, G., Bibb, J.A., Bradley, J. & Wimmer, E. Molecular characterization of the cellular receptor for poliovirus. Virology 199, 105–113 (1994).
Zibert, A. & Wimmer, E. N glycosylation of the virus binding domain is not essential for function of the human poliovirus receptor. J. Virol. 66, 7368–7373 (1992).
Aoki, J., Koike, S., Ise, I., Sato-Yoshida, Y. & Nomoto, A. Amino acid residues on human poliovirus receptor involved in interaction with poliovirus. J. Biol. Chem. 269, 8431–8438 (1994).
Tahara-Hanaoka, S. et al. Functional characterization of DNAM-1 (CD226) interaction with its ligands PVR (CD155) and nectin-2 (PRR-2/CD112). Int. Immunol. 16, 533–538 (2004).
Leis, M., Marschall, M. & Stamminger, T. Downregulation of the cellular adhesion molecule Thy-1 (CD90) by cytomegalovirus infection of human fibroblasts. J. Gen. Virol. 85, 1995–2000 (2004).
Scholz, M. et al. Cytomegalovirus- and interferon-related effects on human endothelial cells. Cytomegalovirus infection reduces upregulation of HLA class II antigen expression after treatment with interferon-gamma. Hum. Immunol. 35, 230–238 (1992).
Kojima, H. et al. CD226 mediates platelet and megakaryocytic cell adhesion to vascular endothelial cells. J. Biol. Chem. 278, 36748–36753 (2003).
Shibuya, K. et al. CD226 (DNAM-1) is involved in lymphocyte function-associated antigen 1 costimulatory signal for naive T cell differentiation and proliferation. J. Exp. Med. 198, 1829–1839 (2003).
Ohka, S. et al. Receptor (CD155)-dependent endocytosis of poliovirus and retrograde axonal transport of the endosome. J. Virol. 78, 7186–7198 (2004).
Mueller, S., Cao, X., Welker, R. & Wimmer, E. Interaction of the poliovirus receptor CD155 with the dynein light chain Tctex-1 and its implication for poliovirus pathogenesis. J. Biol. Chem. 277, 7897–7904 (2002).
Takai, Y., Irie, K., Shimizu, K., Sakisaka, T. & Ikeda, W. Nectins and nectin-like molecules: roles in cell adhesion, migration, and polarization. Cancer Sci. 94, 655–667 (2003).
Oda, T., Ohka, S. & Nomoto, A. Ligand stimulation of CD155α inhibits cell adhesion and enhances cell migration in fibroblasts. Biochem. Biophys. Res. Commun. 319, 1253–1264 (2004).
Reymond, N. et al. DNAM-1 and PVR regulate monocyte migration through endothelial junctions. J. Exp. Med. 199, 1331–1341 (2004).
McSharry, B.P., Jones, C.J., Skinner, J.W., Kipling, D. & Wilkinson, G.W. Human telomerase reverse transcriptase-immortalized MRC-5 and HCA2 human fibroblasts are fully permissive for human cytomegalovirus. J. Gen. Virol. 82, 855–863 (2001).
Robertson, M.J. et al. Characterization of a cell line, NKL, derived from an aggressive human natural killer cell leukemia. Exp. Hematol. 24, 406–415 (1996).
McSharry, B.P., Tomasec, P., Neale, M.L. & Wilkinson, G.W. The most abundantly transcribed human cytomegalovirus gene (β 2.7) is non-essential for growth in vitro. J. Gen. Virol. 84, 2511–2516 (2003).
He, T.C. et al. A simplified system for generating recombinant adenoviruses. Proc. Natl. Acad. Sci. USA 95, 2509–2514 (1998).
Acknowledgements
We thank C. Jones and D. Kipling for their cooperation with the telomerase immortalization of fibroblasts; V. Groh for MIC mAbs; O. Mandelboim for NKp30-Fc and NKp46-Fc proteins; and E. Mocarski, M. Wills and J. Sathish for discussions. Supported by the Wellcome Trust, Biotechnology and Biological Sciences Research Council and Medical Research Council.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Amino acid sequence alignment of UL141 from 23 HCMV strains allowing a high degree of conservation. (PDF 69 kb)
Supplementary Fig. 2
Ig-like b sandwich domains encoded by HCMV UL141 (residues 15-114 in the mature protein), its orthologues in chimpanzee and rhesus cytomegalovirus (CCMV and RhCMV UL141), and the related UL14 gene in HCMV and CCMV. (PDF 54 kb)
Supplementary Fig. 3
Expression of a Secreted form of gpUL141. (PDF 138 kb)
Supplementary Fig. 4
GpUL141 is expressed with early-late phase kinetics. (PDF 47 kb)
Supplementary Fig. 5
Analysis of intracellular localization of gpUL141. (PDF 231 kb)
Supplementary Fig. 6
NK killing of autologous strain TB40E-Bart-infected fibroblasts is inhibited by treatment with the anti-CD155 mAb D171. (PDF 53 kb)
Supplementary Fig. 7
UL141, but not UL144 or UL145, inhibited expression of CD155 on stably transfected HEK-293 cells. (PDF 46 kb)
Supplementary Fig. 8
Pre-treatment with soluble UL141 blocks binding of the CD155 mAb D171. (PDF 59 kb)
Supplementary Fig. 9
Adhesion between NK cells and targets expression UL141. (PDF 99 kb)
Rights and permissions
About this article
Cite this article
Tomasec, P., Wang, E., Davison, A. et al. Downregulation of natural killer cell–activating ligand CD155 by human cytomegalovirus UL141. Nat Immunol 6, 181–188 (2005). https://doi.org/10.1038/ni1156
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1156
This article is cited by
-
Natural killer cells and their exosomes in viral infections and related therapeutic approaches: where are we?
Cell Communication and Signaling (2023)
-
Genome sequences of human cytomegalovirus strain TB40/E variants propagated in fibroblasts and epithelial cells
Virology Journal (2021)
-
Pathogenesis of human cytomegalovirus in the immunocompromised host
Nature Reviews Microbiology (2021)
-
Generation of hypoimmunogenic T cells from genetically engineered allogeneic human induced pluripotent stem cells
Nature Biomedical Engineering (2021)
-
Modulation of innate and adaptive immunity by cytomegaloviruses
Nature Reviews Immunology (2020)