Abstract
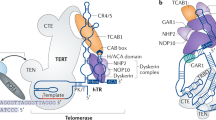
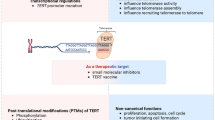
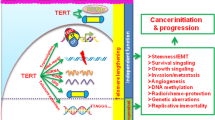
Telomerase is a specialized type of reverse transcriptase which catalyzes the synthesis and extension of telomeric DNA (for review, see ref.1). This enzyme is highly active in most cancer cells, but is inactive in most somatic cells2. This striking observation led to the suggestion that telomerase might be important for the continued growth3 or progression4 of cancer cells. However, little is known about the molecular mechanism of telomerase activation in cancer cells. Human telomerase reverse transcriptase (hTRT) has recently been identified as a putative human telomerase catalytic subunit5,6. We transfected the gene encoding hTRT into telomerase-negative human normal fibroblast cells and demonstrated that expression of wild-type hTRT induces telomerase activity, whereas hTRT mutants containing mutations in regions conserved among other reverse transcriptases did not. Hepatocellular carcinoma (2O samples) and non-cancerous liver tissues (19 samples) were examined for telomerase activity and expression of hTRT, the human telomerase RNA component (hTR; encoded by TER)7 and the human telomerase-associated protein (HTLP1; encoded by 7EP7)8,9. A significant correlation between hTRT expression and telomerase activity was observed. These results indicate that the hTRT protein is the catalytic subunit of human telomerase, and that it plays a key role in the activation of telomerase in cancer cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Blackburn, E.H. Telomerase. Annu. Rev. Biochem. 61, 113–129 (1992)
Kirn, N.W. et al. Specific association of human telomerase activity with immortal cells and cancer Science 266, 2011–2015 (1994)
Harley, C.B. & Villeponteau, B. Telomeres and telomerase in aging and cancer Curr. Opin. Genet. Dev. 5, 249–255 (1995)
Ishikawa, F. Telomere crisis, the driving force in cancer cell evolution. Biochem. Biophys. Res. Commun. 230, 1–6 (1997)
Meyerson, M. et al. hE5T2, the putative human telomerase catalytic subunit gene, is up-regulated in tumor cells and during immortalization. Cell 90, 785–795 (1997)
Nakamura, T.M. et al. Telomerase catalytic subunit homologs from fission yeast and human. Science 277, 955–959 (1997)
Feng, J. et al. The RNA Component of Human Telomerase. Science 269, 1236–1241 (1995)
Nakayama, J., Saito, M., Nakamura, H., Matsuura, A. & Ishikawa, f. TLP1: a gene encoding a protein component of mammalian telomerase is a novel member of WD repeats family. Cell 88, 875–884 (1997)
Harrington, L et al.A mammalian telomerase-associated protein Science 275, 973–977 (1997)
Lingner, J. et al. Reverse transcriptase motifs in the catalytic subunit of telomerase. Science 276, 561–567 (1997)
Counter, CM., Meyerson, M., Eaton, E.N. & Weinberg, R.A. The catalytic subunit of yeast telomerase. Proc. Natl. Acad. Sci. USA 94, 9202–9207 (1997)
Graham, F.L., Smiley, J., Russell, W.C. & Nairn, R. Characteristics of a human cell line transformed by DNA from human adenovirus type 5. J. Gen. Virol. 36, 59–74 (1977)
Matsuo, M., Kaji, K., Utakoji, T. & Hosoda, K. Ploidy of human embryonic fibroblasts during in vitro aging. Gerontol. 37, 33–37 (1982)
Jacobs, J.P., Jones, C.M. & Bailie, J.P. Nature 227, 168–170 (1970)
Tatematsu, K. et al. A novel quantitative “stretch PCR assay”, that detects a dramatic increase in telomerase activity during the progression of myeloid leukemias. Oncogene 13, 2265–2274 (1996)
Steitz, T.A., Smerdon, S.J ., Jäger, J. & Joyce, C.M. A unified polymerase mechanism for nonhomologous DNA and RNA polymerases. Science 266, 2022–2025 (1994)
Nakashio, R. et al. Significance of telomerase activity in the diagnosis of small differentiated hepatocellular carcinoma. Int. J. Cancer 74, 141–147 (1997)
Tahara, H. et al. Telomerase activity in human liver tissues: comparison between chronic liver disease and hepatocellular carcinomas. Cancer Res. 55, 2734–2736 (1995)
Wright, W.E., Shay, J.W. & Piatyszek, M.A. Modifications of a telomeric repeat amplification protocol (TRAP) result in increased reliability, linearity and sensitivity.Nucleic Acids Res. 23, 3794–3795 (1995)
Xiong, Y. & Eickbush, T.H. Origin and evolution of retroelements based upon their reverse transcriptase sequences. EMBO J. 9, 3353–3362 (1990)
Eickbush, T.H. in The evolutionary biology of viruses (ed. Morse, S.S.) 121–157 (Raven Press, New York, 1994)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakayama, Ji., Tahara, H., Tahara, E. et al. Telomerase activation by hTRT in human normal fibroblasts and hepatocellular carcinomas . Nat Genet 18, 65–68 (1998). https://doi.org/10.1038/ng0198-65
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0198-65
This article is cited by
-
A CRISPR base editing approach for the functional assessment of telomere biology disorder-related genes in human health and aging
Biogerontology (2024)
-
A pan-cancer analysis of Dyskeratosis congenita 1 (DKC1) as a prognostic biomarker
Hereditas (2023)
-
The role of telomeres and telomerase in cirrhosis and liver cancer
Nature Reviews Gastroenterology & Hepatology (2019)
-
Correlation of human telomerase reverse transcriptase single nucleotide polymorphisms with in vitro fertilisation outcomes
Journal of Assisted Reproduction and Genetics (2019)
-
The protein subunit of telomerase displays patterns of dynamic evolution and conservation across different metazoan taxa
BMC Evolutionary Biology (2017)