Abstract
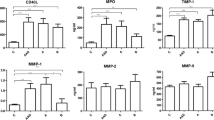
Background: Matrix metalloproteinases (MMPs) have been implicated in the pathogenesis of arterial aneurysms through increased proteolysis of extracellular matrix proteins. Increased proteolysis due to elevated matrix degrading enzyme activity in the arterial wall may act as a susceptibility factor for the development of coronary aneurysms. Plasmin strongly stimulates pro-MMP enzyme conversion to the active form. Plasmin hyperactivity due to decreased plasminogen activator inhibitor-1 (PAI-1) may cause MMP over activity and coronary aneurysms. The aim of this study was to investigate the association between PAI-1 and presence of coronary aneurysms.
Methods: Twenty-three patients with aneurysmal coronary artery disease and stable angina were enrolled into study (Group 1). Twenty-two patients without coronary aneurysm were selected as a control group (Group 2). PAI-1 was measured in peripheral venous blood.
Results: The plasma PAI-1 level was lower in the coronary artery aneurysmatic patients compared to the control group (8.41 ± 4.28 vs. 13.32 ± 10.05 ng/ml, p = 0.037). Serum C-reactive protein (CRP) values were not significantly different between groups (3.83 ± 1.08 vs. 4.01 ± 1.35 mg/l, p>0.05).
Conclusion: Increased matrix degrading enzyme activity can cause arterial wall destruction through increased proteolysis of extracellular matrix proteins. Unregulated plasmin hyperactivity due to decreased inhibition by PAI-1 may play an important role in coronary aneurysm formation.
Similar content being viewed by others
References
Dollery CM, McEwan JR, Henney AM. Matrix metalloproteinases and cardiovascular disease. Circ Res 1995;77:863–868.
Birkedal-Hansen H, Moore W, Bodden MK, et al. Matrix metalloproteinases: A review. Crit Rev Oral Biol Med 1993;4:197–250.
Fuster V. The Vulnerable Atherosclerotic Plaque: Understanding, Identification, and Modification. Inc. Armonk NY: Futura Publishing Company, 2001, Chap. 22: Regulation of Metalloproteinases and their Inhibitors in Atheroma.
Matrisian LM. Metalloproteinases and their inhibitors in matrix remodelling. Trends Genet 1990;6:121–125.
Loftus IM, Naylor AR, Bell PR, Thompson MM. Matrix metalloproteinases and atherosclerotic plaque instability. Br J Surg 2002;89:680–694.
Robinson FC. Aneurysms of the coronary arteries. Am Heart J 1985;109:129–135.
Syed M, Lesch M. Coronary artery aneurysm: A review. Progr Cardiovasc Dis 1997;40:77–84.
Demopoulus VP, Olympios CD, Fakiolas CN, et al. The natural history of aneurysmal coronary artery disease. Heart 1997;78:136–141.
Ercan E, Tengiz I, Yakut N, Gurbuz A. Large atheroscle-rotic left main coronary aneurysm: A case report and review of literature. Int J Cardiol 2003;88:95–98.
Gallin JI, Synderman R. Inflammation: Basic Principles and Clinical Correlates. Chapter 16. Fibroblasts and Matrix Proteins. 3rd ed. Philadelphia: Lippincott Williams & Wilkins, 1999; pp. 227–257.
Thompson RW, Parks WC. Role of matrix metalloproteinases in abdominal aortic aneurysms. Ann N Y Acad Sci 1996;800:157–174.
Annabi B, Shedid D, Ghosn P, et al. Differential regulation of matrix metalloproteinase activities in abdominal aortic aneurysms. J Vasc Surg 2002;35:539–546.
Kim SC, Singh M, Huang J, et al. Matrix metalloproteinase-9 in cerebral aneurysms. Neurosurgery 1997; 41:642–647.
Lesauskaite V, Tanganelli P. Morphogenesis of thoracic aorta aneurysms: Investigation of matrix metalloproteinases and their tissue inhibitors. Medicina 2002;38:31–35.
Knox JB, Sukhova GK, Whittemore AD, Libby P. Evidence for altered balance between matrix metalloproteinases and their inhibitors in human aortic diseases. Circulation 1997;95:205–212.
Petersen E, Gineitis A, Wagberg F, Angquist KA. Activity of matrix metalloproteinase-2 and-9 in abdominal aortic aneurysms. Relation to size and rupture. Eur J Vasc Endovasc Surg 2000;20:457–461.
Petersen E, Wagberg F, Angquist KA. Proteolysis of the abdominal aortic aneurysm wall and the association with rupture. Eur J Vasc Endovasc Surg 2002;23:153–157.
Lamblin N, Bauters C, Hermant X, Lablanche JM, Helbecque N, Amouyel P. Polymorphisms in the promoter regions of MMP-2, MMP-3, MMP-9 and MMP-12 genes as determinants of aneurysmal coronary artery disease. JAm Coll Cardiol 2002;3;40(1):43–48.
Senzaki H, Kobayashi T, Nagasaka H, et al. Plasminogen activator inhibitor-1 in patients with Kawasaki disease: Diagnostic value for the prediction of coronary artery lesion and implication for a new mode of therapy. Pediatr Res 2003;53:983–988.
Domanovits H, Schillinger M, Mullner M, et al. Acute phase reactants in patients with abdominal aortic aneurysm. Atherosclerosis 2002;163:297–302.
Sakai M, Asayama K, Otabe T, Kohri T, Shirahata A. Low tissue plasminogen activator relative to plasminogen activator inhibitor-1 as a marker of cardiac complication in children with Kawasaki disease. Clin Appl Thromb Hemost 2001;7:214–218.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ercan, E., Tengiz, I., Duman, C. et al. Decreased Plasminogen Activator Inhibitor-1 Levels in Coronary Artery Aneurysmatic Patients. J Thromb Thrombolysis 17, 207–211 (2004). https://doi.org/10.1023/B:THRO.0000040490.00796.30
Issue Date:
DOI: https://doi.org/10.1023/B:THRO.0000040490.00796.30