Abstract
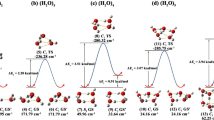
Small hydrogen-bonded water clusters have received much attention and are the subject of both theoretical and chemical research. The crystalline molecular host provides an environment for the stabilization of various topologies of water clusters. The present article deals with (H2O) n clusters, identified/observed crystallographically in respective crystalline hydrates. The structural details of water decamers, octamers, hexamers, tetramers, and dimers are described systematically.
Similar content being viewed by others
References
(a) R. Ludwig (2001). Angew. Chem. Int. Ed. 40, 1808.
(b) P. Ball, H 2 O: A Biography of Water (Weidenfeld and Nicolson, 1999).
(a) C. J. Tsai and K. D. Jordan (1993). J. Chem. Phys. 99, 6957.
(b) C. J. Tsai and K. D. Jordan (1993). J. Phys. Chem. 97, 5208.
(c) J. O. Jensen, P. N. Krishnan, and L. A. Burke (1995). Chem. Phys. Lett. 246, 13.
(d) M. J. Pedulla, F. Vila, and K. D. Jordan (1996). J. Chem. Phys. 105, 11091.
(e) J. O. Jung and R. B. Gerber (1996). J. Chem. Phys. 105, 10332.
(f) K. S. Kim, M. Dupuis, G. C. Lie, and E. Clementi (1986). Chem. Phys. Lett. 131, 451.
(g) B. R. Eggen, A. J. Marks, J. N. Murrell, and S. C. Farantos (1994). Chem. Phys. Lett. 219, 247.
(h) P. N. Krishnan, J. O. Jensen, and L. A. Burke (1994). Chem. Phys. Lett. 217, 311.
(i) J. Kim, B. J. Mhin, S. J. Lee, and K. S. Kim (1994). Chem. Phys. Lett. 219, 243.
(j) L. A. Burke, J. O. Jensen, J. L. Jensen, and P. N. Krishnan (1993). Chem. Phys. Lett. 206, 293.
(k) C. J. Tsai and K. D. Jordan (1991). J. Chem. Phys. 95, 3850.
(a) C. J. Tsai and K. D. Jordan (1993). Chem. Phys. Lett. 213, 181.
(b) F. H. Stillinger and C. W. David (1980). J. Chem. Phys. 73, 3384.
(c) S. S. Xantheas (1994). J. Chem. Phys. 100, 7523.
(d) R. Knochenmuss and S. Leutwyler (1992). J. Chem. Phys. 96, 5233.
(e) S. Y. Fredericks, K. D. Jordan, and T. S. Zwier (1996). J. Phys. Chem. 100, 7810.
(f) C. J. Gruenloh, J. R. Carney, C. A. Arrington, T. S. Zwier, S. Y. Fredericks, and K. D. Jordan (1997). Science 276, 1678.
(g) F. D. Coker, R. E. Miller, and R. O. Watts (1985). J. Chem. Phys. 82, 3554.
(h) R. J. Saykally and G. A. Blake (1993). Science 259, 1570.
(i) K. Liu, M. G. Brown, and R. J. Saykally (1997). J. Phys. Chem. A. 48, 8995.
(k) M. R. Viant, J. D. Cruzan, D. D. Lucas, M. G. Brown, K. Liu, and R. J. Saykally (1997). J. Phys. Chem. A. 48, 9032.
(l) J. D. Cruzan, M. R. Viant, M. G. Brown, and R. J. Saykally (1997). J. Phys. Chem. A 48, 9022.
(m) K. Liu, M. G. Brown, J. D. Cruzan, and R. J. Saykally (1997). J. Phys. Chem. A 48, 9011.
(n) S. S. Xantheas (1995). J. Chem. Phys. 102, 4505.
(o) K. Kim, K. D. Jordan, and T. S. Zwier (1994). J. Am. Chem. Soc. 116, 11568.
(a) L. J. Barbour, G. W. Orr, and J. L. Atwood (1998). Nature 393, 671.
(b) L. J. Barbour, G. W. Orr, and J. L. Atwood (2000). Chem. Commun., 859.
W. B. Blanton, S. W. Gordon-Wylie, G. R. Clark, K. D. Jordon, J. T. Wood, U. Geiser, and T. J. Collins (1999). J. Am. Chem. Soc. 121, 3551.
J. L. Atwood, L. J. Barbour, T. J. Ness, C. L. Raston, and P. L. Raston (2001). J. Am. Chem. Soc. 123, 7192.
R.J.Doedens,E.Yohannes,and M.I.Khan (2002).Chem.Commun .,62.
N. Moorthy, R. Natarajan, and P. Venugopalan (2002). Angew. Chem. Int. Ed. 41, 3417.
R. Custelcean, C. Afloroaei, M. Vlassa, and M. Polverejan (2000). Angew. Chem. Int. Ed. 39, 3094.
K. M. Park, R. Kuroda, and T. Iwamoto (1993). Angew. Chem. Int. Ed. 32, 884.
C. Foces-Foces, F. H. Cano, M. Martinez-Ripoll, R. Faure, C. Roussel, R. M. Claramunt, C. Lopez, D. Sanz, and J. Elguero (1990). Tetrahedron: Asymmetry 1, 65.
J. Xu, E. Radkov, M. Ziegler, and K. N. Raymond (2000). Inorg. Chem. 39, 4156.
S. Supriya, S. Manikumari, P. Raghavaiah, and S. K. Das (2003). New. J. Chem. 2, 218.
K. K. Chacko and W. Saenger (1981). J. Am. Chem. Soc. 103, 1708.
V. Zabel, W. Saenger, and S. A. Mason (1986). J. Am. Chem. Soc. 108, 3664.
D. K. Chand and P. K. Bharadwaj (1998). Inorg. Chem. 37, 5050.
G. R. Newkome, F. R. Fronczek, and D. K. Kohli (1981). Acta Cryst. B37, 2114.
P. C. Manor and W. Saenger (1974). J. Am. Chem. Soc. 96, 3630.
S. Manikumari, V. Shivaiah, and S. K. Das (2002). Inorg. Chem. 41, 6953.
H. König (1944). Z. Kristallogr. 105, 279.
U. Buck, I. Ettischer, M. Melzer, V. Buch, and J. Sadlej (1998). Phys. Rev. Lett. 80, 2578.
R. Ludwig (2000). Chem. Phys. Chem. 1, 53.
K. Nauta and R. E. Miller (2000). Science 287, 293.
B. Kamb (1964). Acta Cryst. 17, 1437.
(a) D. Eisenberg and W. Kauzmann, The Structure and Properties of Water (Oxford University Press, Oxford, 1969).
(b) N. H. Fletcher, The Chemical Physics of Ice (Cambridge University Press, Cambridge, 1970).
G. A. Jeffrey, An Introduction to Hydrogen Bonding (Oxford University Press, Oxford, 1997).
S. W. Peterson and H. A. Levy (1957). Acta Cryst. 10, 70.
(a) F. A. Cotton, Advanced Inorganic Chemistry (Wiley, New York, 1980), pp 154-155.
(b) R. D. Cannon and R. P. White (1988). Prog. Inorg. Chem. 36, 195.
(a) T. R. Dyke, K. M. Mack, and J. S. Muenter (1977). J. Chem. Phys. 66, 498.
(b) J. A. Odutola and T. R. Dyke (1980). J. Chem. Phys. 72, 5062.
(a) M. F. Vernon, D. J. Krajnovich, H. S. Kwok, J. M. Lisy, Y.-R. Shen, and Y. T. Lee (1982). J. Chem. Phys. 77, 47.
(b) R. H. Page, J. G. Frey, Y.-R. Shen, and Y. T. Lee (1984). Chem. Phys. Lett. 106, 373.
(c) D. F. Coker, R. E. Miller, and R. O. Watts (1985). J. Chem. Phys. 82, 3354.
(a) D. K. Chand, K. G. Ragunathan, T. C. W. Mak, and P. K. Bharadwaj (1996). J. Org. Chem. 61, 1169.
(b) J.-M. Lehn (1978). Acc. Chem. Res. 11, 49.
S. Pal, N. B. Sankaran, and A. Samanta (2003). Angew. Chem. Int. Ed. 42, 1741.
A. Müller, E. Krickemeyer, H. Bögge, M. Schmidtmann, B. Botar, and M. O. Talismanova (2003). Angew. Chem. Int. Ed. 42, 2085.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Supriya, S., Das, S.K. Small Water Clusters in Crystalline Hydrates. Journal of Cluster Science 14, 337–366 (2003). https://doi.org/10.1023/B:JOCL.0000005068.77622.a4
Issue Date:
DOI: https://doi.org/10.1023/B:JOCL.0000005068.77622.a4