Abstract
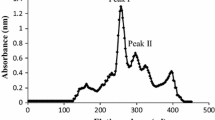
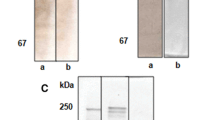
Three forms of female-specific plasma protein (FSPP 1-3) were purified from blood plasma of estrogen-treated white perch (Morone americana) by combining several types of ion-exchange chromatography including a novel, fast flow, strong anion exchanger (POROS media), followed by gel filtration. Native FSPP 1, FSPP 2 and FSPP 3 had molecular masses of 532 kDa, 532 kDa and 426 kDa, respectively. The apparent mass of purified FSPP 1 and FSPP 2 after SDS-PAGE under reducing conditions was ∼ 180 kDa, while FSPP 3 appeared as a major ∼ 148 kDa band. All of the FSPPs resembled one another with respect to amino acid composition but each appeared to be immunologically distinct. In double immunodiffusion using anti-total FSPP (antiserum raised against vitellogenic female plasma pre-absorbed by male plasma), each FSPP formed one precipitin line that crossed those produced by both others. A rabbit antiserum was raised against each FSPP and absorbed with combinations of the other two FSPPs to ensure specificity. Using the antisera, each FSPP was detected by immuno-electrophoresis in plasma from vitellogenic females or estrogen-treated male or immature fish, but no FSPP was detected in normal male plasma. Endoprotease (Asp-N) digests of the FSPPs were subjected to HPLC separation for N-terminal sequencing and mapping of isolated peptides to published vitellogenin (Vg) sequences. Results of these analyses indicate that white perch FSPP 1, FSPP 2, and FSPP 3 can be classified into three Vg groups identified in previous studies: VgA, VgB, and VgC-like protein, respectively. This is the first report, of which we are aware, on isolation of more than two Vg proteins from any species of vertebrate except the chicken.
Similar content being viewed by others
References
Altschul, S.F., Gish, W., Miller, W., Myers, E.W. and Lipman, D.J. 1990. Basic local alignment search tool. J. Mol. Biol. 215: 403–410.
Berlinsky, D.L., Jackson, L.F. and Sullivan, C.V. 1995. The annual reproductive cycle of the white bass, Morone chrysops. J. World Aquaculture Soc. 26: 252–260.
Buerano, C., Inaba, K., Natividad, F.F. and Morisawa, M. 1995. Vitellogenins of Oreochromis niloticus: identification, isolation, and biochemical and immunochemical characterization. J. Exp. Zool. 273: 59–69.
Byrne, B.M., Jong, H., Fouchier, R.A.M., Williams, D.L., Gruber, M. and AB, G. 1989. Rudimentary phosvitin domain in a minor chicken vitellogenin gene. Biochemistry 28: 2572–2577.
Denslow, N.D., Chow, M., Ming, M., Bonemelli, S., Folmar, L.C., Heppell, S.A. and Sullivan, C.V. 1997. Development of biomarkers for environmental contaminants affecting fish. In: Chemically Induced Alterations in Functional Development and Reproduction of Fishes. pp. 73–86. Edited by Rolland, R.M., Gilbertson, M. and Peterson, R.E. SEATAC Technical Publication Series, Society of Environmental Toxicology and Chemistry, Pensacola.
Ding, J.L., Hee, P.L. and Lam, T.J. 1989. Two forms of vitellogenin in the plasma and gonads of male Oreochromis aureus. Comp. Physiol. Biochem. 93B: 363–370.
Fujita, T., Shimizu, M., Hiramatsu, N., Fukada, H. and Hara, A. 2002. Purification of serum precursor proteins to vitelline emvelope (choriogenins) in masu salmon, Oncorhynchus masou. Comp. Biochem. Physiol. 132B: 599–610.
Fyhn, H.J., Finn, R.N., Reith, M. and Norberg, B. 1999. Yolk protein hydrolysis and oocyte free amino acids as key features in the adaptive evolution of teleost fishes to seawater. Sarsia 84: 451–456.
Greeley, M.S. Jr, Calder, D.R. and Wallace R.A. 1986. Changes in teleost yolk proteins during oocyte maturation: correlation of yolk proteolysis with oocyte hydration. Comp. Biochem. Physiol. 84B: 1–9.
Hashimoto, S., Bessho, H., Hara, A., Nakamura, M., Iguchi, T. and Fujita, K. 2000. Elevated serum vitellogenin levels and gonadal abnormalities in wild male flounder (Pleuronectes yokohamae) from Tokyo Bay, Japan. Marine Environ. Res. 49: 37–53.
Heppell, S.A., Denslow, N.D., Folmer, L.C. and Sullivan, C.V. 1995. Universal assay of vitellogenin as a biomarker for environmental estrogens. Environ. Health Perspect. 103: 9–15.
Heppell, S.A., Jackson, L.F., Weber, G.M. and Sullivan, C.V. 1999. Enzyme-linked immunosorbent assay (ELISA) of vitellogenin in temperate basses (genus Morone): plasma and in vitro analyses. Trans. Am. Fish. Soc. 128: 532–541.
Hiramatsu, N. and Hara, A. 1996. Relationship between vitellogenin and its related egg yolk proteins in Sakhalin taimen (Hucho perryi). Comp. Biochem. Physiol. 115A: 243–251.
Hiramatsu, N., Hara, A., Hiramatsu, K., Fukada, H., Weber, G.M., Denslow, N.D. and Sullivan, C.V. 2002b. Vitellogenin-derived yolk proteins of white perch, Morone americana: Purification, characterization and vitellogenin-receptor binding. Biol. Reprod. 67: 655–667.
Hiramatsu, N., Hiramatsu, K., Hirano, K. and Hara, A. 2002c. Vitellogenin-derived yolk proteins in a hybrid sturgeon, bester (Huso huso x Acipencer ruthenus): Identification, characterization and course of proteolysis during embryogenesis. Comp. Biochem. Physiol. 131A: 429–441.
Hiramatsu, N., Matsubara, T., Weber, G.M., Sullivan, C.V. and Hara, A. 2002a. Vitellogenesis in aquatic animals. Fish. Sci. 68. Suppl. I: 694–699.
Jackson, L.F. and Sullivan, C.V. 1995. Reproduction of white perch (Morone americana): the annual gametogenic cycle. Trans. Amer. Fish. Soc. 124: 563–577.
King, V.W., Gosh, S., Thomas, P. and Sullivan, C.V. 1997. A receptor for the oocyte maturation-inducing hormone 17β, 20β,21–trihydroxy-4–pregnen-3–one (20β-S) on ovarian membranes of striped bass. Biol. Reprod. 56: 266–271.
Kishida, M., Anderson, T.R. and Specker, J.L. 1992. Induction by β-estradiol of vitellogenin in striped bass (Morone saxatilis): Characterization and quantification in plasma and mucus. Gen. Comp. Endocrinol. 88: 29–39.
Kishida, M., Specker, J.L. 1993. Vitellogenin in tilapia (Oreochromis mossambicus): Induction of two forms by estradiol, quantification in plasma and characterization in oocyte extract. Fish. Physiol. Biochem. 12: 171–182.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
LaFleur, G.J., Byrne, J.B.M., Haux, C., Greenberg, R. and Wallace, R.A. 1995a. Liver-derived cDNAs: Vitellogenins and vitelline envelope protein precursors (choriogenins). In: Reproductive Physiology of Fish. pp. 336–338. Edited by Goetz, F. and Thomas, P. The University of Texas at Austin, Texas.
LaFleur, G.J., Byrne, J.B.M., Kanungo, J., Nelson, L.D., Greenburg, R.M. and Wallace, R.A. 1995b. Fundulus heteroclitus vitellogenin: The deduced primary structure of a piscine precursor to noncrystalline, liquid-phase yolk protein. J. Mol. Evol. 41: 505–521.
Lee, K.B.H., Lim,E.H., Lam, T.J. and Ding, J.L. 1992. Vitellogenin diversity in the perciformes. J. Exp. Zool. 264: 100–106.
Lund, E.D., Sullivan, C.V. and Place, A.R. 2000. Annual cycle of plasma lipids in captive reared striped bass: effects of environmental conditions and reproductive cycle. Fish Physiol. Biochem. 22: 263–275.
Matsubara, T., Ohkubo, N., Andoh, T., Sullivan, C.V. and Hara, A. 1999. Two forms of vitellogenin, yielding two distinct lipovitellins, play different roles during oocyte maturation and early development of barfin flounder, Verasper moseri, a marine teleost that spawns pelagic eggs. Dev. Biol. 213: 18–32.
Mommsen, T.P. and Walsh, P.L. 1988. Vitellogenesis and oocyte assembly. In: Fish Physiology Vol. XI A. pp. 347–406. Edited by Hoar, W.S. and Randall, D.J. Academic Press, New York.
Murakami, M., Iuchi, I. and Yamagami, K. 1990. Yolk phosphoprotein metabolism during early development of the fish, Oryzias latipes. Dev. Growth Differ. 32: 619–627.
Ohkubo, N. and Matsubara, T. 2002. Sequential utilization of free amino acids, yolk proteins and lipids in developing eggs and yolk-sac larvae of barfin flounder, Verasper moseri. Marine Biol. 140: 187–196.
Parks, L.G., Cheek, A.O., Denslow, N.D., Heppell, S.A., McLachlan, J.A., LeBlanc, G.A. and Sullivan, C.V. 1999. Fathead minnow (Pimephales promelas) vitellogenin: Purification, characterization and quantitative immunoassay for the detection of estrogenic compounds. Comp. Biochem. Physiol. 123C: 113–125.
Patiño, R. and Sullivan, C.V. 2002. Ovarian follicle growth, maturation, and ovulation in teleost fishes. Fish Physiol. Biochem. 26: 57–70.
Reith, M., Munholland, J., Kelly, J., Finn, R.N. and Fyhn, H.J. 2001. Lipovitellins derived from two forms of vitellogenin are differentially processed during oocyte maturation in haddock (Melanogrammus aeglefinus). J. Exp. Zool. 291: 58–67.
Shimizu, M., Fujita, T. and Hara, A. 1998. Purification of the precursors to vitelline envelope proteins from serum of Sakhalin taimen, Hucho perryi. J. Exp. Zool. 282: 385–395.
Shimizu, M., Fujiwara, Y., Fukada, H. and Hara, A. 2002. Purification and identification of a second form of vitellogenin from ascites of medaka (Oryzias latipes) treated with estrogen. J. Exp. Zool. 293: 726–735.
Sullivan, C.V., Berlinsky, D.L. and Hodson, R.G. 1997. In: Striped Bass and Other Morone Culture. pp. 11–73. Edited by Harrell, R.M. Elsevier Science, Amsterdam.
Specker, J.L. and Sullivan, C.V. 1994. Vitellogenesis in fishes: Status and perspectives. In: Perspectives in Comparative Endocrinology. pp. 304–315. Edited by Davey, K.G. and Peter, R.E. and Tobe, S.E. National Research Council, Ottawa.
Stone, K.L., LoPresti, M.B., Crawford, J.M., DeAngelis, R. and Williams, K.R. 1989. Enzymatic digestion of proteins and HPLC peptide isolation. In: A practical guide to protein and peptide purification for microsequencing. pp. 31–47. Edited by Matsudaira, P.T. Academic Press, California.
Takemura, A. and Kim, B.H. 2001. Effects of estradiol-17β treatment on in vitro and in vivo synthesis of two distinct vitellogenins in tilapia. Comp. Biochem. Physiol. 129A: 641–651.
Tao, Y., Berlinsky, D.L. and Sullivan, C.V. 1996. Vitellogenin receptors in white perch (Morone americana). Biol. Reprod. 55: 646–656.
Tao, Y., Hara, A., Hodson, R.G., Woods, L.C. III and Sullivan, C.V. 1993. Purification, characterization and immunoassay of striped bass (Morone saxatilis) vitellogenin. Fish Physiol. Biochem. 12: 31–46.
Trichet, V., Buisine, N., Mouchel, N., Morn, P., Pends, A.M., Le Pennec, J.P. and Wolff, J. 2000. Genomic analysis of the vitellogenin locus in rainbow trout (Oncorhynchus mykiss) reveals a complex history of gene amplification and retroposon activity. Mol. Gen. Genet. 263: 828–837.
Wallace, R.A. 1985. Vitellogenesis and oocyte growth in nonmammalian vertebrate. In: Developmental Biology. Vol. 1. pp. 127–177. Edited by Browder, L.W. Plenum Press, New York.
Wallace, R.A. and Begovac, P.C. 1985. Phosvitins in Fundulus oocytes and eggs. Preliminary chromatographic and electrophoretic analyses together with biological considerations. J. Biol. Chem. 260: 11268–11274.
Wallace, R.A. and Selman, K. 1985. Major protein changes during vitellogenesis and maturation of Fundulus oocytes. Dev. Biol. 110: 492–498.
Wang, H., Yan, T., Tan, J.T.T. and Gong, Z. 2000. A zebrafish vitellogenin gene (vg3) encodes a novel vitellogenin without a phosvitin domain and may represent a primitive vertebrate vitellogenin gene. Gene 256: 303–310.
Wang, S.Y., Smith, D.E. and Williams, D.L. 1983. Purification of avian vitellogenin III: Comparison with vitellogenins I and II. Biochemistry 22: 6206–6212.
Wang, S.Y. and Williams, D.L. 1980. Identification, purification, and characterization of two distinct avian vitellogenins. Biochemistry 19: 1557–1563.
Weber, G.M. and Sullivan, C.V. 2000. Effects of insulin-like growth factor I on in vitro final oocyte maturation and ovarian steroid production in striped bass (Morone saxatilis). Biol. Reprod. 63: 1049–1057.
Yamagami, K. 1996. Studies on the hatching enzyme (choriolysin) and its substrate, egg envelope, constructed of the precursors (choriogenins) in Oryzias latipes: A sequel to the information in 1991/1992. Zool. Sci. 13: 331–340.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hiramatsu, N., Matsubara, T., Hara, A. et al. Identification, purification and classification of multiple forms of vitellogenin from white perch (Morone americana). Fish Physiology and Biochemistry 26, 355–370 (2002). https://doi.org/10.1023/B:FISH.0000009266.58556.9a
Issue Date:
DOI: https://doi.org/10.1023/B:FISH.0000009266.58556.9a