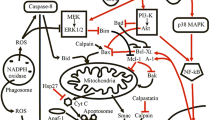
Abstract
Phagocytic leukocytes such as neutrophils and macrophages are essential for the innate immune response against invading bacteria. Binding and ingestion of bacteria by these host cells triggers potent anti-microbial activity, including production of reactive oxygen species. Although phagocytes are highly adept at destroying bacteria, modulation of leukocyte apoptosis or cell death by bacteria has emerged as a mechanism of pathogenesis. Whereas induction of macrophage apoptosis by pathogens may adversely affect the host immune response to infection, acceleration of neutrophil apoptosis following phagocytic interaction with bacteria appears essential for the resolution of infection. This idea is supported by the finding that some bacterial pathogens alter normal phagocytosis-induced neutrophil apoptosis to survive and cause disease. This review summarizes what is currently known about modulation of phagocyte apoptosis by bacteria and describes a paradigm whereby bacteria-induced neutrophil apoptosis plays a role in the resolution of infection.
Similar content being viewed by others
References
Nauseef WM, Clark RA. Granulocytes. In: Mandell GL, Bennett JE, Dolin R, eds. Basic Principles in the Diagnosis and Management of Infectious Diseases. New York: Churchill Livingstone 2000: 89–112.
Jones SL, Lindberg FP, Brown EJ. Phagocytosis. In: Paul WF, ed. Fundamental Immunology. New York: Lippincott-Raven 1999: 97–1020.
Gordon S. Development and distribution of mononu-clear phagocytes: Relevance to inflammation. In: Gallin JI, Snyderman R, eds. Inflammation, Basic Principles and Clinical Correlates. New York: Lippincott-Raven 1999: 35–48.
Yamaguchi T, Kakinuma K, Kaneda M, Shimada K. Comparative studies on alveolar macrophages and polymorphonuclear leukocytes. I. H2O2 and O2-generation by rabbit alveolar macrophages. J Biochem (Tokyo) 1980; 87: 1449–1455.
Nakagawara A, Nathan CF, Cohn ZA. Hydrogen peroxide metabolism in human monocytes during differentiation in vitro. J Clin Invest 1981; 68: 1243–1252.
Yagisawa M, Yuo A, Yonemaru M, et al. Superoxide release and NADPH oxidase components in mature human phago-cytes: Correlation between functional capacity and amount of functional proteins. Biochem Biophys Res Commun 1996; 228: 510–516.
Johansson A, Jesaitis AJ, Lundqvist H, et al. Different subcellular localization of cytochrome b and the dormant NADPH-oxidase in neutrophils and macrophages: Effect on the production of reactive oxygen species during phagocytosis. Cell Immunol 1995; 161: 61–71.
Kakinuma K, Yamaguchi T, Shimada K, Sato N. Comparative studies on alveolar macrophages and polymorphonu-clear leukocytes. III. Difference spectra of their cellular and subcellular fractions. J Biochem (Tokyo) 1980; 88: 1467–1474.
Sasada M, Kubo A, Nishimura T, Kakita T, et al. Candidacidal activity of monocyte-derived human macrophages: Relationship between Candida killing and oxygen radical generation by human macrophages. J Leukoc Biol 1987; 41: 289–294.
Weissler JC, Lipscomb MF, Lem VM, Toews GB. Tumor killing by human alveolar macrophages and blood monocytes Decreased cytotoxicity of human alveolar macrophages. Am Rev Respir Dis 1986; 134: 532–537.
Locksley RM, Nelson CS, Fankhauser JE, Klebanoff SJ. Loss of granule myeloperoxidase during in vitro culture of human monocytes correlates with decay in antiprotozoa activity. Am J Trop Med Hyg 1987; 36: 541–548.
Devalon ML, Elliott GR, Regelmann WE. Oxidative response of human neutrophils, monocytes, and alveolar macrophages induced by unopsonized surface-adherent Staphylococcus aureus. Infect Immun 1987; 55: 2398–2403.
Lincoln JA, Lefkowitz DL, Cain T, et al. Exogenous myeloper-oxidase enhances bacterial phagocytosis and intracellular killing by macrophages. Infect Immun 1995; 63: 3042–3047.
Mathy-Hartert M, Deby-Dupont G, Melin P, Lamy M, Deby C. Bactericidal activity against Pseudomonas aeruginosa is acquired by cultured human monocyte-derived macrophages after uptake of myeloperoxidase. Experientia 1996; 52: 167–174.
Lefkowitz SS, Gelderman MP, Lefkowitz DL, Moguilevsky N, Bollen A. Phagocytosis and intracellular killing of Candida albicans by macrophages exposed to myeloperoxidase. J Infect Dis 1996; 173: 1202–1207.
Zychlinsky A, Prevost MC, Sansonetti PJ. Shigella flexneri induces apoptosis in infected macrophages. Nature 1992; 358: 167–169.
Zychlinsky A, Kenny B, Menard R, Prevost MC, Holland IB, Sansonetti PJ. IpaB mediates macrophage apoptosis induced by Shigella flexneri. Mol Microbiol 1994; 11: 619–627.
Zychlinsky A, Fitting C, Cavaillon JM, Sansonetti PJ. Interleukin-1 is released by murine macrophages during apoptosis induced by Shigella flexneri. J Clin Invest 1994; 94: 1328–1332.
Hilbi H, Chen Y, Thirumalai K, Zychlinsky A. The interleukin-1beta-converting enzyme, caspase 1, is activated during Shigella flexneri-induced apoptosis in human monocyte-derived macrophages. Infect Immun 1997; 65: 5165–5170.
Hilbi H, Moss JE, Hersh D, et al. Shigella-induced apoptosis is dependent on caspase-1 which binds to IpaB. J Biol Chem 1998; 273: 32895–32900.
Chen Y, Smith MR, Thirumalai K, Zychlinsky A. A bacterial invasin induces macrophage apoptosis by binding directly to ICE. EMBO J 1996; 15: 3853–3860.
Fernandez-Prada CM, Hoover DL, Tall BD, Venkatesan MM. Human monocyte-derived macrophages infected with virulent Shigella flexneri in vitro undergo a rapid cytolytic event similar to oncosis but not apoptosis. Infect Immun 1997; 65: 1486–1496.
Guichon A, Zychlinsky A. Clinical isolates of Shigella species induce apoptosis in macrophages. J Infect Dis 1997; 175: 470–473.
Raqib R, Ekberg C, Sharkar P, et al. Apoptosis in acute shigellosis is associated with increased production of Fas/Fas ligand, perforin, caspase-1, and caspase-3 but reduced production of Bcl-2 and interleukin-2. Infect Immun 2002; 70: 3199–3207.
Zychlinsky A, Kenny B, Prevost MC, Holland IB, Sansonetti PJ. The ipaB gene of Shigella flexneri and macrophage-programmed cell death. Infect Agents Dis 1993; 2: 212–214.
Nonaka T, Kuwae A, Sasakawa C, Imajoh-Ohmi S. Shigella flexneri YSH6000 induces two types of cell death, apoptosis and oncosis, in the differentiated human monoblastic cell line U937. FEMS Microbiol Lett 1999; 174: 89–95.
Hilbi H, Puro RJ, Zychlinsky A. Tripeptidyl peptidase II promotes maturation of caspase-1 in Shigella flexneri-induced macrophage apoptosis. Infect Immun 2000; 68: 5502–5508.
Fernandez-Prada CM, Hoover DL, Tall BD, Hartman AB, Kopelowitz J, Venkatesan MM. Shigella flexneri IpaH(7.8) facilitates escape of virulent bacteria from the endocytic vacuoles of mouse and human macrophages. Infect Immun 2000; 68: 3608–3619.
Yu J, Oragui EE, Stephens A, Kroll JS, Venkatesan MM. Inactivation of DsbA alters the behaviour of Shigella flexneri towards murine and human-derived macrophage-like cells. FEMS Microbiol Lett 2001; 204: 81–88.
Hathaway LJ, Griffin GE, Sansonetti PJ, Edgeworth JD. Human monocytes kill Shigella flexneri but then die by apoptosis associated with suppression of proinflammatory cytokine production. Infect Immun 2002; 70: 3833–3842.
Monack DM, Raupach B, Hromockyj AE, Falkow S. Salmonella typhimurium invasion induces apoptosis in infected macrophages. Proc Natl Acad Sci USA 1996; 93: 9833–9838.
Chen LM, Kaniga K, Galan JE. Salmonella spp. are cytotoxic for cultured macrophages. Mol Microbiol 1996; 21: 1101–1115.
Hersh D, Monack DM, Smith MR, Ghori N, Falkow S, Zychlinsky A. The Salmonella invasin SipB induces macrophage apoptosis by binding to caspase-1. Proc Natl Acad Sci USA 1999; 96: 2396–2401.
Lundberg U, Vinatzer U, Berdnik D, von Gabain A, Baccarini M. Growth phase-regulated induction of Salmonella-induced macrophage apoptosis correlates with transient expression of SPI-1 genes. J Bacteriol 1999; 181: 3433–3437.
Yrlid U, Wick MJ. Salmonella-induced apoptosis of infected macrophages results in presentation of a bacteria-encoded antigen after uptake by bystander dendritic cells. J Exp Med 2000; 191: 613–624.
Schwan WR, Huang XZ, Hu L, Kopecko DJ. Differential bacterial survival, replication, and apoptosis-inducing ability of Salmonella serovars within human and murine macrophages. Infect Immun 2000; 68: 1005–1013.
Watson PR, Gautier AV, Paulin SM, Bland AP, Jones PW, Wallis TS. Salmonella enterica serovars Typhimurium and Dublin can lyse macrophages by a mechanism distinct from apoptosis. Infect Immun 2000; 68: 3744–3747.
Watson PR, Paulin SM, Jones PW, Wallis TS. Interaction of Salmonella serotypes with porcine macrophages in vitro does not correlate with virulence. Microbiology 2000; 146: 1639–1649.
van der Velden AW, Lindgren SW, Worley MJ, Heffron F. Salmonella pathogenicity island 1-independent induction of apoptosis in infected macrophages by Salmonella enterica serotype Typhimurium. Infect Immun 2000; 68: 5702–5709.
Jesenberger V, Procyk KJ, Yuan J, Reipert S, Baccarini M. Salmonella-induced caspase-2 activation in macrophages: A novel mechanism in pathogen-mediated apoptosis. J Exp Med 2000; 192: 1035–1046.
Brennan MA, Cookson BT. Salmonella induces macrophage death by caspase-1-dependent necrosis. Mol Microbiol 2000; 38: 31–40.
Jesenberger V, Procyk KJ, Ruth J, Schreiber M, Theussl HC, Wagner EF, Baccarini M. Protective role of Raf-1 in Salmonella-induced macrophage apoptosis. J Exp Med 2001; 193: 353–364.
Libby SJ, Lesnick M, Hasegawa P, Weidenhammer E, Guiney DG. The Salmonella virulence plasmid spv genes are required for cytopathology in human monocyte-derived macrophages. Cell Microbiol 2000; 2: 49–58.
Zhou X, Mantis N, Zhang XR, Potoka DA, Watkins SC, Ford HR. Salmonella typhimurium induces apoptosis in human monocyte-derived macrophages. Microbiol Immunol 2000; 44: 987–995.
Santos RL, Tsolis RM, Baumler AJ, Smith R 3rd, Adams LG. Salmonella enterica serovar Typhimurium induces cell death in bovine monocyte-derived macrophages by early sipB dependent and delayed sipB-independent mechanisms. Infect Immun 2001; 69: 2293–2301.
Monack DM, Detweiler CS, Falkow S. Salmonella pathogenicity island 2-dependent macrophage death is mediated in part by the host cysteine protease caspase-1. Cell Microbiol 2001; 3: 825–837.
Richter-Dahlfors A, Buchan AM, Finlay BB. Murine salmonellosis studied by confocal microscopy: Salmonella typhimurium resides intracellularly inside macrophages and exerts a cytotoxic effect on phagocytes in vivo. J Exp Med 1997; 186: 569–580.
Forsberg M, Blomgran R, Lerm M, et al. Differential effects of invasion by and phagocytosis of Salmonella typhimurium on apoptosis in human macrophages: potential role of Rho-GTPases and Akt. J Leukoc Biol 2003; 74: 620–629.
Basso H, Rharbaoui F, Staendner LH, Medina E, Garcia-Del Portillo F, Guzman CA. Characterization of a novel intracellularly activated gene from Salmonella enterica serovar Typhi. Infect Immun 2002; 70: 5404–5411.
Monack DM, Hersh D, Ghori N, Bouley D, Zychlinsky A, Falkow S. Salmonella exploits caspase-1 to colonize Peyer’s patches in a murine typhoid model. J Exp Med 2000; 192: 249–58.
Monack DM, Mecsas J, Ghori N, Falkow S. Yersinia signals macrophages to undergo apoptosis and YopJ is necessary for this cell death. Proc Natl Acad Sci USA 1997; 94: 10385–10390.
Monack DM, Mecsas J, Bouley D, Falkow S. Yersinia-induced apoptosis in vivo aids in the establishment of a systemic infection of mice. J Exp Med 1998; 188: 2127–2137.
Zhang Y, Bliska JB. Role of Toll-like receptor signaling in the apoptotic response of macrophages to Yersinia infection. Infect Immun 2003; 71: 1513–1519.
Mills SD, Boland A, Sory MP, et al. Yersinia enterocolitica induces apoptosis in macrophages by a process requiring functional type III secretion and translocation mechanisms and involving YopP, presumably acting as an effector protein. Proc Natl Acad Sci USA 1997; 94: 12638–12643.
Ruckdeschel K, Roggenkamp A, Lafont V, Mangeat P, Heesemann J, Rouot B. Interaction of Yersinia enterocolitica with macrophages leads to macrophage cell death through apoptosis. Infect Immun 1997; 65: 4813–4821.
Ruckdeschel K, Harb S, Roggenkamp A, et al. Yersinia enterocolitica impairs activation of transcription factor NF-kappaB: involvement in the induction of programmed cell death and in the suppression of the macrophage tumor necrosis factor alpha production. J Exp Med 1998; 187: 1069–1079.
Ruckdeschel K, Mannel O, Richter K, et al. Yersinia outer protein P of Yersinia enterocolitica simultaneously blocks the nuclear factor-kappa B pathway and exploits lipopolysaccha-ride signaling to trigger apoptosis in macrophages. J Immunol 2001; 166: 1823–1831.
Denecker G, Declercq W, Geuijen CA, et al. Yersinia enterocolitica YopP-induced apoptosis of macrophages involves the apoptotic signaling cascade upstream of Bid. J Biol Chem 2001; 276: 19706–19714.
Ruckdeschel K, Richter K, Mannel O, Heesemann J. Arginine-143 of Yersinia enterocolitica YopP crucially determines isotype-related NF-kappaB suppression and apoptosis induction in macrophages. Infect Immun 2001; 69: 7652–7662.
Ruckdeschel K, Mannel O, Schrottner P. Divergence of apoptosis-inducing and preventing signals in bacteria-faced macrophages through myeloid differentiation factor 88 and IL-1 receptor-associated kinase members. J Immunol 2002; 168: 4601–4611.
Ruckdeschel K, Richter K. Lipopolysaccharide desensitization of macrophages provides protection against Yersinia enterocolitica-induced apoptosis. Infect Immun 2002; 70: 5259–5264.
Weeks S, Hill J, Friedlander A, Welkos S. Anti-V antigen antibody protects macrophages from Yersinia pestis-induced cell death and promotes phagocytosis. Microb Pathog 2002; 32: 227–237.
Ng LC, Forslund O, Koh S, Kuoppa K, Sjostedt A. The response of murine macrophages to infection with Yersinia pestis as revealed by DNA microarray analysis. Adv Exp Med Biol 2003; 529: 155–160.
Haase R, Kirschning CJ, Sing A, et al. A dominant role of Toll-like receptor 4 in the signaling of apoptosis in bacteria-faced macrophages. J Immunol 2003; 171: 4294–4303.
Muller A, Hacker J, Brand BC. Evidence for apoptosis of human macrophage-like HL-60 cells by Legionella pneumophila infection. Infect Immun 1996; 64: 4900–4906.
Hagele S, Hacker J, Brand BC. Legionella pneumophila kills human phagocytes but not protozoan host cells by inducing apoptotic cell death. FEMS Microbiol Lett 1998; 169: 51–58.
Gao LY, Abu Kwaik Y. Apoptosis in macrophages and alveolar epithelial cells during early stages of infection by Legionella pneumophila and its role in cytopathogenicity. Infect Immun 1999; 67: 862–870.
Gao LY, Abu Kwaik Y. Activation of caspase 3 during Legionella pneumophila-induced apoptosis. Infect Immun 1999; 67: 4886–4694.
Zink SD, Pedersen L, Cianciotto NP, Abu-Kwaik-Y. The Dot/Icm type IV secretion system of Legionella pneumophila is essential for the induction of apoptosis in human macrophages. Infect Immun 2002; 70: 1657–1663.
Neumeister B, Faigle M, Lauber K, Northoff H, Wesselborg S. Legionella pneumophila induces apoptosis via the mitochondrial death pathway. Microbiology 2002; 148: 3639–3650.
Walz JM, Gerhardt H, Faigle M, Wolburg H, Neumeister B. Legionella species of different human prevalence induce different rates of apoptosis in human monocytic cells. APMIS 2000; 108: 398–408.
Alli OA, Gao LY, Pedersen LL, et al. Temporal pore formation-mediated egress from macrophages and alveolar epithelial cells by Legionella pneumophila. Infect Immun 2000; 68: 6431–6440.
Fratazzi C, Arbeit RD, Carini C, Remold HG. Programmed cell death of Mycobacterium avium serovar 4-infected human macrophages prevents the mycobacteria from spreading and induces mycobacterial growth inhibition by freshly added, uninfected macrophages. J Immunol 1997; 158: 4320–4327.
Bermudez LE, Parker A, Petrofsky M. Apoptosis of Mycobacterium avium-infected macrophages is mediated by both tumour necrosis factor (TNF) and Fas, and involves the activation of caspases. Clin Exp Immunol 1999; 116: 94–99.
Balcewicz-Sablinska MK, Gan H, Remold HG. Interleukin 10 produced by macrophages inoculated with Mycobacterium avium attenuates mycobacteria-induced apoptosis by reduction of TNF-alpha activity. J Infect Dis 1999; 180: 1230–1237. ORRECTED PROOF
Mohagheghpour N, van Vollenhoven A, Goodman J, Bermudez LE. Interaction of Mycobacterium avium with human monocyte-derived dendritic cells. Infect Immun 2000; 68: 5824–5829.
Allen S, Sotos J, Sylte MJ, Czuprynski CJ. Use of Hoechst 33342 staining to detect apoptotic changes in bovine mononuclear phagocytes infected with Mycobacterium avium subsp. paratuberculosis. Clin Diagn Lab Immunol 2001; 8: 460–464.
Feng CG, Kullberg MC, Jankovic D, et al. Transgenic mice expressing human interleukin-10 in the antigen-presenting cell compartment show increased susceptibility to infection with Mycobacterium avium associated with decreased macrophage effector function and apoptosis. Infect Immun 2002; 70: 6672–6679.
Bhattacharyya A, Pathak S, Basak C, Law S, Kundu M, Basu J. Execution of macrophage apoptosis by Mycobacterium avium through apoptosis signal-regulating kinase 1/p38 mitogen-activated protein kinase signaling and caspase 8 activation. J Biol Chem 2003; 278: 26517–26525.
Hayashi T, Catanzaro A, Rao SP. Apoptosis of human mono-cytes and macrophages by Mycobacterium avium sonicate. Infect Immun 1997; 65: 5262–5271.
Ahmad A, Khan M, Raykundalia C, Catty D. Study of the mechanisms of killing of Mycobacterium bovisBCGby apoptosis in J774 murine macrophages. J Pak Med Assoc 1999; 49: 273–278.
Kremer L, Estaquier J, Brandt E, Ameisen JC, Locht C. Mycobacterium bovis Bacillus Calmette Guerin infection prevents apoptosis of resting human monocytes. Eur J Immunol 1997; 27: 2450–2456.
Keane J, Remold HG, Kornfeld H. Virulent Mycobacterium tuberculosis strains evade apoptosis of infected alveolar macrophages. J Immunol 2000; 164: 2016–2020.
Gutierrez-Pabello JA, McMurray DN, Adams LG. Upregulation of thymosin beta-10 by Mycobacterium bovis infection of bovine macrophages is associated with apoptosis. Infect Immun 2002; 70: 2121–2127.
Riendeau CJ, Kornfeld H. THP-1 cell apoptosis in response to Mycobacterial infection. Infect Immun 2003; 71: 254–259.
Molloy A, Laochumroonvorapong P, Kaplan G. Apoptosis, but not necrosis, of infected monocytes is coupled with killing of intracellular bacillus Calmette-Guerin. J Exp Med 1994; 180: 1499–1509.
Kausalya S, Somogyi R, Orlofsky A, Prystowsky MB. Requirement of A1-a for bacillus Calmette-Guerin-mediated protection of macrophages against nitric oxide-induced apoptosis. J Immunol 2001; 166: 4721–4727.
Guerardel Y, Maes E, Briken V, et al. Lipomannan and lipoara binomannan from a clinical isolate of Mycobacterium kansasii: Novel structural features and apoptosis-inducing properties. J Biol Chem 2003; 278: 36637–36651.
Hernandez MO, Neves I, Sales JS, Carvalho DS, Sarno EN, Sampaio EP. Induction of apoptosis in monocytes by Mycobacterium leprae in vitro: A possible role for tumour necrosis factoralpha. Immunology 2003; 109: 156–164.
Durrbaum-Landmann I, Gercken J, Flad HD, Ernst M. Effect of in vitro infection of human monocytes with low numbers of Mycobacterium tuberculosis bacteria on monocyte apoptosis. Infect Immun 1996; 64: 5384–5389.
Keane J, Balcewicz-Sablinska MK, Remold HG, et al. Infection by Mycobacterium tuberculosis promotes human alveolar macrophage apoptosis. Infect Immun 1997; 65: 298–304.
Placido R, Mancino G, Amendola A, et al. Apoptosis of human monocytes/macrophages in Mycobacterium tuberculosis infection. J Pathol 1997; 181: 31–38.
Rojas M, Barrera LF, Puzo G, Garcia LF. Differential induction of apoptosis by virulent Mycobacterium tuberculosis in resistant and susceptible murine macrophages: Role of nitric oxide and mycobacterial products. J Immunol 1997; 159: 1352–1361.
Klingler K, Tchou-Wong KM, Brandli O, et al. Effects of mycobacteria on regulation of apoptosis in mononuclear phagocytes. Infect Immun 1997; 65: 5272–52788.
Oddo M, Renno T, Attinger A, Bakker T, MacDonald HR, Meylan PR. Fas ligand-induced apoptosis of infected human macrophages reduces the viability of intracellular Mycobacterium tuberculosis. J Immunol 1998; 160: 5448–5454.
Rojas M, Barrera LF, Garcia LF. Induction of apoptosis in murine macrophages by Mycobacterium tuberculosis is reactive oxygen intermediates-independent. Biochem Biophys Res Commun 1998; 247: 436–442.
Ragno S, Estrada-Garcia I, Butler R, Colston MJ. Regulation of macrophage gene expression by Mycobacterium tuberculosis: down-regulation of mitochondrial cytochrome c oxidase. Infect Immun 1998; 66: 3952–3958.
Rojas M, Olivier M, Gros P, Barrera LF, Garcia LF. TNF-alpha and IL-10 modulate the induction of apoptosis by virulent Mycobacterium tuberculosis in murine macrophages. J Immunol 1999; 162: 6122–6131.
Mustafa T, Phyu S, Nilsen R, Bjune G, Jonsson R. Increased expression of Fas ligand on Mycobacterium tuberculosis infected macrophages: A potential novel mechanism of immune evasion by Mycobacterium tuberculosis? Inflammation 1999; 23: 507–521.
Santucci MB, Amicosante M, Cicconi R, et al. Mycobacterium tuberculosis-induced apoptosis in monocytes/macrophages: Early membrane modifications and intracellular mycobacterial viability. J Infect Dis 2000; 181: 1506–1509.
Rojas M, Garcia LF, Nigou J, Puzo G, Olivier M. Mannosylated lipoarabinomannan antagonizes Mycobacteriumtuberculosis-induced macrophage apoptosis by altering Ca+2-dependent cell signaling. J Infect Dis 2000; 182: 240–251.
Ciaramella A, Martino A, Cicconi R, Colizzi V, Fraziano M. Mycobacterial 19-kDa lipoprotein mediates Mycobacteriumtuberculosis-induced apoptosis in monocytes/macrophages at early stages of infection. Cell Death Differ 2000; 7: 1270–1272.
Duan L, Gan H, Arm J, Remold HG. Cytosolic phospholipase A2 participates with TNF-alpha in the induction of apoptosis of human macrophages infected with Mycobacterium tuberculosis H37Ra. J Immunol 2001; 166: 7469–7476.
Keane J, Shurtleff B, Kornfeld H. TNF-dependent BALB/c murine macrophage apoptosis following Mycobacterium tuberculosis infection inhibits bacillary growth in an IFN-gamma independent manner. Tuberculosis (Edinb). 2002; 82: 55–61.
Duan L, Gan H, Golan DE, Remold HG. Critical role of mitochondrial damage in determining outcome of macrophage infection with Mycobacterium tuberculosis. J Immunol 2002; 169: 5181–5187.
Rojas M, Olivier M, Garcia LF. Activation of JAK2/STAT1-alpha-dependent signaling events during Mycobacterium tuberculosis-induced macrophage apoptosis. Cell Immunol 2002; 217: 58–66.
Lopez M, Sly LM, Luu Y, Young D, Cooper H, Reiner NE. The 19-kDa Mycobacterium tuberculosis protein induces macrophage apoptosis through Toll-like receptor-2. J Immunol 2003; 170: 2409–2416. UNCORRECTED PROOF
Danelishvili L, McGarvey J, Li YJ, Bermudez LE. Mycobacterium tuberculosis infection causes different levels of apoptosis and necrosis in human macrophages and alveolar epithelial cells. Cell Microbiol 2003; 5: 649–660.
Ciaramella A, Cavone A, Santucci MB, et al. Proinflammatory cytokines in the course of Mycobacterium tuberculosis-induced apoptosis in monocytes/macrophages. J Infect Dis 2002; 186: 1277–1282.
Sly LM, Hingley-Wilson SM, Reiner NE, McMaster WR. Survival of Mycobacterium tuberculosis in host macrophages involves resistance to apoptosis dependent upon induction of antiapoptotic Bcl-2 family member Mcl-1. J Immunol 2003; 170: 430–437.
Spira A, Carroll JD, Liu G, et al. Apoptosis genes in human alveolar macrophages infected with virulent or attenuated Mycobacterium tuberculosis: A pivotal role for tumor necrosis factor. Am J Respir Cell Mol Biol 2003; 29: 545–551.
George KM, Pascopella L, Welty DM, Small PL. A Mycobacterium ulcerans toxin, mycolactone, causes apoptosis in guinea pig ulcers and tissue culture cells. Infect Immun 2000; 68: 877–883.
Kato S, Muro M, Akifusa S, et al. Evidence for apoptosis of murine macrophages by Actinobacillus actinomycetemcomitans infection. Infect Immun 1995; 63: 3914–3919.
Muro M, Koseki T, Akifusa S, et al. Role of CD14 molecules in internalization of Actinobacillus actinomycetemcomitans by macrophages and subsequent induction of apoptosis. Infect Immun 1997; 65: 1147–1151.
Nonaka K, Ishisaki A, Muro M, et al. Possible involvement of protein kinase C in apoptotic cell death of macrophages infected with Actinobacillus actinomycetemcomitans. FEMS Microbiol Lett 1998; 159: 247–254.
Muro M, Nakashima K, Tomioka J, et al. Inhibitory effect of lipopolysaccharide on apoptotic cell death in macrophages infected with Actinobacillus actinomycetemcomitans. FEMS Microbiol Lett 1999; 175: 211–216.
Nonaka K, Ishisaki A, Okahashi N, et al. Involvement of caspases in apoptotic cell death of murine macrophages infected with Actinobacillus actinomycetemcomitans. J Periodontal Res 2001; 36: 40–47.
Nakashima K, Tomioka J, Kato S, Nishihara T, Kowashi Y. Nitric oxide-mediated protection of Actinobacillus actinomycetemcomitans-infected murine macrophages against apoptosis. Nitric Oxide 2002; 6: 61–68.
Nalbant A, Zadeh HH. Actinobacillus actinomycetemcomitans induces apoptosis of T lymphocytes by the Fas and Fas ligand pathway. Oral Microbiol Immunol 2002; 17: 277–284.
Schaible UE, Winau F, Sieling PA, et al. Apoptosis facilitates antigen presentation to T lymphocytes through MHC-I and CD1 in tuberculosis. Nat Med 2003; 9: 1039–1046.
Fairbairn IP, Stober CB, Kumararatne DS, Lammas DA. ATP-mediated killing of intracellular mycobacteria by macrophages is a P2X(7)-dependent process inducing bacterial death by phagosome-lysosome fusion. J Immunol 2001; 167: 3300–3307.
Dockrell DH, Lee M, Lynch DH, Read RC. Immune mediated phagocytosis and killing of Streptococcus pneumoniae are associated with direct and bystander macrophage apoptosis. J Infect Dis 2001; 184: 713–722.
Ali F, Lee ME, Iannelli F, et al. Streptococcus pneumoniae associated human macrophage apoptosis after bacterial internalization via complement and Fcgamma receptors correlates with intracellular bacterial load. J Infect Dis 2003; 188: 1119–1131.
Dockrell DH, Marriott HM, Prince LR, et al. Alveolar macrophage apoptosis contributes to pneumococcal clearance in a resolving model of pulmonary infection. J Immunol 2003; 171: 5380–5388.
Khelef N, Zychlinsky A, Guiso N. Bordetella pertussis induces apoptosis in macrophages: Role of adenylate cyclase hemolysin. Infect Immun 1993; 61: 4064–4071.
Khelef N, Guiso N. Induction of macrophage apoptosis by Bordetella pertussis adenylate cyclase-hemolysin. FEMS Microbiol Lett 1995; 134: 27–32.
Gueirard P, Druilhe A, Pretolani M, Guiso N. Role of adenylate cyclase-hemolysin in alveolar macrophage apoptosis during Bordetella pertussis infection in vivo. Infect Immun 1998; 66: 1718–1725.
Khelef N, Gounon P, Guiso N. Internalization of Bordetella pertussis adenylate cyclase-haemolysin into endocytic vesicles contributes to macrophage cytotoxicity. Cell Microbiol 2001; 3: 721–730.
Abramson T, Kedem H, Relman DA. Proinflammatory and proapoptotic activities associated with Bordetella pertussis filamentous hemagglutinin. Infect Immun 2001; 69: 2650–2658.
Khelef N, DeShazer D, Friedman RL, Guiso N. In vivo and in vitro analysis of Bordetella pertussis catalase and Fe-superoxide dismutase mutants. FEMS Microbiol Lett 1996; 142: 231–235.
Dellacasagrande J, Capo C, Raoult D, Mege JL. IFN-gamma-mediated control of Coxiella burnetii survival in monocytes: The role of cell apoptosis and TNF. J Immunol 1999; 162: 2259–2265.
Liles WC, Thomsen AR, O’Mahony DS, Klebanoff SJ. Stimulation of human neutrophils and monocytes by staphylococcal phenol-soluble modulin. J Leukoc Biol 2001; 70: 96–102.
Bratton DL, May KR, Kailey JM, Doherty DE, Leung DY. Staphylococcal toxic shock syndrome toxin-1 inhibits monocyte apoptosis. J Allergy Clin Immunol 1999; 103: 895–900.
Baran J, Guzik K, Hryniewicz W, Ernst M, Flad HD, Pryjma J. Apoptosis of monocytes and prolonged survival of granulocytes as a result of phagocytosis of bacteria. Infect Immun 1996; 64: 4242–4248.
Cerquetti MC, Goren NB, Ropolo AJ, Grasso D, Giacomodonato MN, Vaccaro MI. Nitric oxide and apoptosis induced in Peyer’s patches by attenuated strains of Salmonella enterica serovar Enteritidis. Infect Immun 2002; 70: 964–969.
Allen S, Sotos J, Sylte MJ, Czuprynski CJ. Use of Hoechst 33342 staining to detect apoptotic changes in bovine mononuclear phagocytes infected with Mycobacterium avium subsp. paratuberculosis. Clin Diagn Lab Immunol 2001; 8: 460–464.
Betten A, Bylund J, Cristophe T, et al. A proinflammatory peptide from Helicobacter pylori activates monocytes to induce lymphocyte dysfunction and apoptosis. J Clin Invest 2001;108: 1221–1228.
Fernandez-Prada CM, Zelazowska EB, Nikolich M, et al. Interactions between Brucella melitensis and human phagocytes: Bacterial surface O-Polysaccharide inhibits phagocytosis, bacterial killing, and subsequent host cell apoptosis. Infect Immun 2003; 71: 2110–2119.
Gross A, Terraza A, Ouahrani-Bettache S, Liautard JP, Dornand J. In vitro Brucella suis infection prevents the programmed cell death of human monocytic cells. Infect Immun 2000; 68: 342–351.
Galdiero E, Romano Carratelli C, Vitiello M, et al. HSP and apoptosis in leukocytes from infected or vaccinated animals by Brucella abortus. New Microbiol 2000; 23: 271. UNCORRECTED
Jewett A, Hume WR, Le H, et al. Induction of apoptotic cell death in peripheral blood mononuclear and polymorphonuclear cells by an oral bacterium, Fusobacterium nucleatum. Infect Immun 2000; 68: 1893–1898.
Ozaki K, Hanazawa S. Porphyromonas gingivalis fimbriae inhibit caspase-3-mediated apoptosis of monocytic THP-1 cells under growth factor deprivation via extracellular signal-regulated kinase-dependent expression of p21 Cip/WAF1. Infect Immun 2001; 69: 4944–4950.
Hall RE, Agarwal S, Kestler DP. Induction of leukemia cell differentiation and apoptosis by recombinant P48, a modulin derived from Mycoplasma fermentans. Biochem Biophys Res Commun 2000; 269: 284–289.
Into T, NodasakaY, Hasebe A, et al. Mycoplasmal lipoproteins induce toll-like receptor 2-and caspases-mediated cell death in lymphocytes and monocytes. Microbiol Immunol 2002; 46: 265–276.
Kuo CF, Wu JJ, Tsai PJ, et al. Streptococcal pyrogenic exotoxin B induces apoptosis and reduces phagocytic activity in U937 cells. Infect Immun 1999; 67: 126–130.
Wang M, Qureshi N, Soeurt N, Splitter G. High levels of nitric oxide production decrease early but increase late survival of Brucella abortus in macrophages. Microb Pathog 2001; 31: 221–230.
Airenne S, Surcel HM, Tuukkanen J, Leinonen M, Saikku P. Chlamydia pneumoniae inhibits apoptosis in human epithelial and monocyte cell lines. Scand J Immunol 2002; 55: 390–398.
Carratelli CR, Rizzo A, Catania MR, et al. Chlamydia pneumoniae infections prevent the programmed cell death on THP-1 cell line. FEMS Microbiol Lett 2002; 215: 69–74.
Lin CG, Kao YT, Liu WT, et al. Calyculin A sensitive protein phosphatase is required for Bacillus anthracis lethal toxin induced cytotoxicity. Curr Microbiol 1996; 33: 224–227.
Popov SG, Villasmil R, Bernardi J, et al. Lethal toxin of Bacillus anthracis causes apoptosis of macrophages. Biochem Biophys Res Commun 2002; 293: 349–355.
Popov SG, Villasmil R, Bernardi J, et al. Effect of Bacillus anthracis lethal toxin on human peripheral blood mononuclear cells. FEBS Lett 2002; 527: 211–215.
Lai XH, Xu JG, Melgar S, Uhlin BE. An apoptotic response by J774 macrophage cells is common upon infection with diarrheagenic Escherichia coli. FEMS Microbiol Lett 1999; 172: 29–34.
Stravodimos KG, Singhal PC, Sharma S, Reddy K, Smith AD. Escherichia coli promotes macrophage apoptosis. J Endourol 1999; 13: 273–277.
Rodrigues VS, Vidotto MC, Felipe I, Santos DS, Gaziri LC. Apoptosis of murine peritoneal macrophages induced by an avian pathogenic strain of Escherichia coli. FEMS Microbiol Lett 1999; 179: 73–78.
Lai XH, Arencibia I, Johansson A, et al. Cytocidal and apoptotic effects of the ClyA protein from Escherichia coli on primary and cultured monocytes and macrophages. Infect Immun 2000; 68: 4363–4367.
Hacker H, Furmann C, Wagner H, Hacker G. Caspase-9/-3 activation and apoptosis are induced in mouse macrophages upon ingestion and digestion of Escherichia coli bacteria. J Immunol 2002; 169: 3172–3179.
Fernandez-Prada C, Tall BD, Elliott SE, Hoover DL, Nataro JP, Venkatesan MM. Hemolysin-positive enteroaggregative and cell-detaching Escherichia coli strains cause oncosis of human monocyte-derived macrophages and apoptosis of murine J774 cells. Infect Immun 1998; 66: 3918–3924.
Goth SR, Stephens RS. Rapid, transient phosphatidylserine externalization induced in host cells by infection with Chlamydia spp. Infect Immun 2001; 69: 1109–1119.
Ojcius DM, Souque P, Perfettini JL, Dautry-Varsat A. Apoptosis of epithelial cells and macrophages due to infection with the obligate intracellular pathogen Chlamydia psittaci. J Immunol 1998; 161: 4220–4226.
Punj V, Sharma R, Zaborina O, Chakrabarty AM. Energy-generating enzymes of Burkholderia cepacia and their interactions with macrophages. J Bacteriol 2003; 185: 3167–3178.
Galindo CL, Sha J, Ribardo DA, Fadl AA, Pillai L, Chopra AK. Identification of Aeromonas hydrophila cytotoxic enterotoxin-induced genes in macrophages using microarrays. J Biol Chem 2003; 278: 40198–40212.
Forde CB, Shi X, Li J, Roberts M. Bordetella bronchiseptica-mediated cytotoxicity to macrophages is dependent on bvg-regulated factors, including pertactin. Infect Immun 1999; 67: 5972–5978.
Stockbauer KE, Foreman-Wykert AK, Miller JF. Bordetella type III secretion induces caspase 1-independent necrosis. Cell Microbiol 2003; 5: 123–132.
Lai XH, Golovliov I, Sjostedt A. Francisella tularensis induces cytopathogenicity and apoptosis in murine macrophages via a mechanism that requires intracellular bacterial multiplication. Infect Immun 2001; 69: 4691–4694.
Lai XH, Sjostedt A. Delineation of the molecular mechanisms of Francisella tularensis-induced apoptosis in murine macrophages. Infect Immun 2003; 71: 4642–4646.
Gobert AP, ChengY, Wang JY, et al. Helicobacter pylori induces macrophage apoptosis by activation of arginase II. J Immunol 2002; 168: 4692–4700.
Arakaki N, Higa F, Koide M, Tateyama M, Saito A. Induction of apoptosis of human macrophages in vitro by Legionella longbeachae through activation of the caspase pathway. J Med Microbiol 2002; 51: 159–168.
Merien F, Baranton G, Perolat P. Invasion of Vero cells and induction of apoptosis in macrophages by pathogenic Leptospira interrogans are correlated with virulence. Infect Immun 1997; 65: 729–738.
Guzman CA, Domann E, Rohde M, et al. Apoptosis of mouse dendritic cells is triggered by listeriolysin, the major virulence determinant of Listeria monocytogenes. Mol Microbiol 1996; 20: 119–126.
Barsig J, Kaufmann SH. The mechanism of cell death in Listeria monocytogenes-infected murine macrophages is distinct from apoptosis. Infect Immun 1997; 65: 4075–4081.
Greub G, Mege JL, Raoult D. Parachlamydia acanthamoeba enters and multiplies within human macrophages and induces their apoptosis. Infect Immun 2003; 71: 5979–5985.
Dacheux D, Toussaint B, Richard M, Brochier G, Croize J, Attree I. Pseudomonas aeruginosa cystic fibrosis isolates induce rapid, type III secretion-dependent, but ExoU-independent, oncosis of macrophages and polymorphonuclear neutrophils. Infect Immun 2000; 68: 2916–2924.
Hauser AR, Engel JN. Pseudomonas aeruginosa induces type-III-secretion-mediated apoptosis of macrophages and epithelial cells. Infect Immun 1999; 67: 5530–5537.
Zaborina O, Dhiman N, Ling Chen M, Kostal J, Holder IA, Chakrabarty AM. Secreted products of a nonmucoid Pseudomonas aeruginosa strain induce two modes of macrophage killing: external-ATP-dependent, P2Z-receptor-mediated necrosis and ATP-independent, caspase-mediated apoptosis. Microbiology 2000; 146: 2521–2530.
Worgall S, Martushova K, Busch A, Lande L, Crystal RG. Apoptosis induced by Pseudomonas aeruginosa in antigen presenting cells is diminished by genetic modification with CD40 ligand. Pediatr Res 2002; 52: 636–644. UNCORRECTED PROOF
Zhang J, Takayama H, Matsuba T, Jiang R, Tanaka Y. Induction of apoptosis in macrophage cell line, J774, by the cell-free supernatant from Pseudomonas aeruginosa. Microbiol Immunol 2003; 47: 199–206.
Yamada T, Goto M, Punj V, et al. The bacterial redox protein azurin induces apoptosis in J774 macrophages through complex formation and stabilization of the tumor suppressor protein p53. Infect Immun 2002; 70: 7054–7062.
Goto M, Yamada T, Kimbara K, et al. Induction of apoptosis in macrophages by Pseudomonas aeruginosa azurin: Tumour-suppressor protein p53 and reactive oxygen species, but not redox activity, as critical elements in cytotoxicity. Mol Microbiol 2003; 47: 549–559.
Huang YT, Jeng CR, Cheng CH, Chueh LL, Liu JJ, Pang VF. Morphological and immunological evidence of a unique selective production and endoplasmic reticular accumulation of interleukin-1alpha in rat peritoneal macrophages induced by Pseudomonas aeruginosa exotoxin A. Cell Immunol 2003; 221: 143–156.
Kim MK, Seong SY, Seoh JY, et al. Orientia tsutsugamushi inhibits apoptosis of macrophages by retarding intracellular calcium release. Infect Immun 2002; 70: 4692–4696.
Arai T, Hiromatsu K, Nishimura H, et al. Endogenous interleukin-10 prevents apoptosis in macrophages during Salmonella infection. Biochem Biophys Res Commun 1995; 213: 600–607.
Minamino M, Sakaguchi I, Naka T, et al. Bacterial ceramides and sphingophospholipids induce apoptosis of human leukaemic cells. Microbiology 2003; 149: 2071–2081.
Guzik K, Bzowska M, Dobrucki J, Pryjma J. Heat-shocked monocytes are resistant to Staphylococcus aureus-induced apoptotic DNA fragmentation due to expression of HSP72. Infect Immun 1999; 67: 4216–4222.
Zlotkin A, Chilmonczyk S, Eyngor M, Hurvitz A, Ghittino C, Eldar A. Trojan horse effect: phagocyte-mediated Streptococcusiniae infection of fish. Infect Immun 2003; 71: 2318–2325.
Fettucciari K, Rosati E, Scaringi L, et al. Group B Streptococcus induces apoptosis in macrophages. J Immunol 2000; 165: 3923–3933.
Buratta S, Fettucciari K, Mambrini R, Fetriconi I, Marconi P, Mozzi R. Group B Streptococcus (GBS) modifies macrophage phosphatidylserine metabolism during induction of apoptosis. FEBS Lett 2002; 520: 68–72.
Ulett GC, Bohnsack JF, Armstrong J, Adderson EE. Beta-hemolysin-independent induction of apoptosis of macrophages infected with serotype III group B Streptococcus. J Infect Dis 2003; 188: 1049–1053.
Kashimoto T, Ueno S, Hanajima M, et al. Vibrio vulnificus induces macrophage apoptosis in vitro and in vivo. Infect Immun 2003; 71: 533–535.
Dacheux D, Goure J, Chabert J, Usson Y, Attree I. Poreforming activity of type III system-secreted proteins leads to oncosis of Pseudomonas aeruginosa-infected macrophages. Mol Microbiol 2001; 40: 76–85.
Herron MJ, Nelson CM, Larson J, Snapp KR, Kansas GS, Goodman JL. Intracellular parasitism by the human granulocytic ehrlichiosis bacterium through the P-selectin ligand, PSGL-1. Science 2000; 288: 1653–1656.
Chen SM, Dumler JS, Bakken JS, Walker DH. Identification of a granulocytotropic Ehrlichia species as the etiologic agent of human disease. J Clin Microbiol 1994; 32: 589–595.
Scaife H, Woldehiwet Z, Hart CA, Edwards SW. Anaplasma phagocytophilum reduces neutrophil apoptosis in vivo. Infect Immun 2003; 71: 1995–2001.
Yoshiie K, Kim HY, Mott J, RikihisaY. Intracellular infection by the human granulocytic ehrlichiosis agent inhibits human neutrophil apoptosis. Infect Immun 2000; 68: 1125–1133.
Van Zandbergen G, Gieffers J, Kothe H, et al. Chlamydia pneumoniae multiply in neutrophil granulocytes and delay their spontaneous apoptosis. J Immunol 2004; 172: 1768–1776.
Cline MJ. Production, destruction, and distribution of neutrophilic granulocytes. In: Cline MJ, ed. The White Cell. Massachusetts: Harvard University Press, 1975: 22–38.
Newman SL, Henson JE, Henson PM. Phagocytosis of senescent neutrophils by human monocyte-derived macrophages and rabbit inflammatory macrophages. J Exp Med 1982; 156: 430–442.
Savill JS, Wyllie AH, Henson JE, Walport MJ, Henson PM, Haslett C. Macrophage phagocytosis of aging neutrophils in inflammation. Programmed cell death in the neutrophil leads to its recognition by macrophages. J Clin Invest 1989; 83: 865–875.
Savill JS, Henson PM, Haslett C. Phagocytosis of aged human neutrophils by macrophages is mediated by a novel ”charge-sensitive” recognition mechanism. J Clin Invest 1989; 84: 1518–1527.
Savill J, Dransfield I, Hogg N, Haslett C. Vitronectin receptor-mediated phagocytosis of cells undergoing apoptosis. Nature 1990; 343: 170–173.
Savill J. Apoptosis in the resolution of inflammation. J Leukoc Biol 1997; 61: 375–380.
Sansonetti PJ, Phalipon A, Arondel J, et al. Caspase-1 activation of IL-1beta and IL-18 are essential for Shigella flexneri induced inflammation. Immunity 2000; 12: 581–590.
Perdomo OJ, Cavaillon JM, Huerre M, Ohayon H, Gounon P, Sansonetti PJ. Acute inflammation causes epithelial invasion and mucosal destruction in experimental shigellosis. J Exp Med 1994; 180: 1307–1319.
Majno G, Joris I. Apoptosis, oncosis, and necrosis. An overview of cell death. Am J Pathol 1995; 146: 3–15.
Pais TF, Appelberg R. Macrophage control of mycobacterial growth induced by picolinic acid is dependent on host cell poptosis. J Immunol 2000; 164: 389–397.
Clemens DL. Characterization of the Mycobacterium tuberculosis phagosome. Trends Microbiol 1996; 4: 113–118.
Schlesinger LS. Entry of Mycobacterium tuberculosis into mononuclear phagocytes. Curr Top Microbiol Immunol 1996; 215: 71–96.
Watson RW, Redmond HP, Wang JH, Condron C, Bouchier-Hayes D. Neutrophils undergo apoptosis following ingestion of Escherichia coli. J Immunol 1996; 156: 3986–3992.
Coxon A, Rieu P, Barkalow FJ, et al. A novel role for the beta 2 integrin CD11b/CD18 in neutrophil apoptosis: A homeo-static mechanism in inflammation. Immunity 1996; 5: 653–666.
Whyte MK, Meagher LC, MacDermot J, Haslett C. Impairment of function in aging neutrophils is associated with apoptosis. J Immunol 1993; 150: 5124–5134.
Kobayashi SD, Braughton KR, Whitney AR, et al. Bacterial pathogens modulate an apoptosis differentiation program in human neutrophils. Proc Natl Acad Sci USA 2003; 100: 10948–10953.
Harvill ET, Cotter PA, Miller JF. Pregenomic comparative analysis between Bordetella bronchiseptica RB50 and Bordetella pertussis tohama I in murine models of respiratory tract infection. Infect Immun 1999; 67: 6109–6118.
Hutchison ML, Poxton IR, Govan JR. Burkholderia cepacia produces a hemolysin that is capable of inducing apoptosis and degranulation of mammalian phagocytes. Infect Immun 1998; 66: 2033–2039. UNCORRECTED PROOF
Mahida YR, Galvin A, Makh S, et al. Effect of Clostridium difficile toxin A on human colonic lamina propria cells: Early loss of macrophages followed by T-cell apoptosis. Infect Immun 1998; 66: 5462–5469.
Colamussi ML, White MR, Crouch E, Hartshorn KL. Influenza A virus accelerates neutrophil apoptosis and markedly potentiates apoptotic effects of bacteria. Blood 1999; 93: 2395–2403.
Watson RW, Redmond HP, Wang JH, Bouchier-Hayes D. Bacterial ingestion, tumor necrosis factor-alpha, and heat induce programmed cell death in activated neutrophils. Shock 1996; 5: 47–51.
Matsuda T, Saito H, Inoue T, et al. Ratio of bacteria to polymorphonuclear neutrophils (PMNs) determines PMN fate. Shock 1999; 12: 365–372.
Yagi Y, Shiono H, Shibahara T, Chikayama Y, Nakamura I, Ohnuma A. Increase in apoptotic polymorphonuclear neutrophils in peripheral blood after intramammary infusion of Escherichia coli lipopolysaccharide. Vet Immunol Immunopathol 2002; 89: 115–125.
Feterowski C, Weighardt H, Emmanuilidis K, Hartung T, Holzmann B. Immune protection against septic peritonitis in endotoxin-primed mice is related to reduced neutrophil apoptosis. Eur J Immunol 2001; 31: 1268–1277.
Liu J, Akahoshi T, Sasahana T, et al. Inhibition of neutrophil apoptosis by verotoxin 2 derived from Escherichia coli O157:H7. Infect Immun 1999; 67: 6203–6205.
Narayanan S, Stewart GC, Chengappa MM, et al. Fusobacterium necrophorum leukotoxin induces activation and apoptosis of bovine leukocytes. Infect Immun 2002; 70: 4609–4620.
Yang YF, Sylte MJ, Czuprynski CJ. Apoptosis: A possible tactic of Haemophilus somnus for evasion of killing by bovine neutrophils? Microb Pathog 1998; 24: 351–359.
Kim JS, Kim JM, Jung HC, Song IS, Kim CY. Inhibition of apoptosis in human neutrophils by Helicobacter pylori watersoluble surface proteins. Scand J Gastroenterol 2001; 36: 589–600.
Hofman V, Ricci V, Mograbi B, et al. Helicobacter pylori lipopolysaccharide hinders polymorphonuclear leucocyte apoptosis. Lab Invest 2001; 81: 375–384.
Cudd LA, Ownby CL, Clarke CR, Sun Y, Clinkenbeard KD. Effects of Mannheimia haemolytica leukotoxin on apoptosis and oncosis of bovine neutrophils. Am J Vet Res 2001; 62: 136–141.
Suttmann H, Lehan N, Bohle A, Brandau S. Stimulation of neutrophil granulocytes with Mycobacterium bovis bacillus Calmette-Guerin induces changes in phenotype and gene expression and inhibits spontaneous apoptosis. Infect Immun 2003; 71: 4647–4656.
Kasahara K, Sato I, Ogura K, Takeuchi H, Kobayashi K, Adachi M. Expression of chemokines and induction of rapid cell death in human blood neutrophils by Mycobacterium tuberculosis. Expression of chemokines and induction of rapid cell death in human blood neutrophils by Mycobacterium tuberculosis. J Infect Dis 1998; 178: 127–137.
Kisich KO, Higgins M, Diamond G, Heifets L. Tumor necrosis factor alpha stimulates killing of Mycobacterium tuberculosis by human neutrophils. Infect Immun 2002; 70: 4591–4599.
Perskvist N, Long M, Stendahl O, Zheng L. Mycobacterium tuberculosis promotes apoptosis in human neutrophils by activating caspase-3 and altering expression of Bax/Bcl-xL via an oxygen-dependent pathway. J Immunol 2002; 168: 6358–6365.
Aleman M, Garcia A, Saab MA, et al. Mycobacterium tuberculosis induced activation accelerates apoptosis in peripheral blood neutrophils from patients with active tuberculosis. AmJ Respir Cell Mol Biol 2002; 27: 583–592.
Stevens PK, Czuprynski CJ. Pasteurella haemolytica leukotoxin induces bovine leukocytes to undergo morphologic changes consistent with apoptosis in vitro. Infect Immun 1996; 64: 2687–2694.
Dacheux D, Attree I, Schneider C, Toussaint B. Cell death of human polymorphonuclear neutrophils induced by a Pseudomonas aeruginosa cystic fibrosis isolate requires a functional type III secretion system. Infect Immun 1999; 67: 6164–6167.
Usher LR, Lawson RA, Geary I, et al. Induction of neutrophil apoptosis by the Pseudomonas aeruginosa exotoxin pyocyanin: A potential mechanism of persistent infection. J Immunol 2002; 168: 1861–1868.
Tateda K, Ishii Y, Horikawa M, et al. The Pseudomonas aeruginosa autoinducer N-3-oxododecanoyl homoserine lactone accelerates apoptosis in macrophages and neutrophils. Infect Immun. 2003; 71: 5785–5793.
Preshaw PM, Schifferle RE, Walters JD. Porphyromonas gingivalis lipopolysaccharide delays human polymorphonuclear leukocyte apoptosis in vitro. J Periodontal Res 1999; 34: 197–202.
do Vale A, Marques F, Silva MT. Apoptosis of sea bass (Dicentrarchus labrax L.) neutrophils and macrophages induced by experimental infection with Photobacterium damselae subsp. piscicida. Fish Shellfish Immunol 2003; 15: 129–144.
Francois M, Le Cabec V, Dupont MA, Sansonetti PJ, Maridonneau-Parini I. Induction of necrosis in human neutrophils by Shigella flexneri requires type III secretion, IpaB and IpaC invasins, and actin polymerization. Infect Immun 2000; 68: 1289–1296.
Yamamoto A, Taniuchi S, Tsuji S, Hasui M, and Kobayashi Y. Role of reactive oxygen species in neutrophil apoptosis following ingestion of heat-killed Staphylococcus aureus. Clin Exp Immunol 2002; 129: 479–484.
Lundqvist-Gustafsson H, Norrman S, Nilsson J, Wilsson A. Involvement of p38-mitogen-activated protein kinase in Staphylococcus aureus-induced neutrophil apoptosis. J Leukoc Biol 2001; 70: 642–648.
Baran J, Guzik K, Hryniewicz W, Ernst M, Flad HD, Pryjma J. Apoptosis of monocytes and prolonged survival of granulocytes as a result of phagocytosis of bacteria. Infect Immun 1996; 64: 4242–4248.
Lotz S, Aga E, Wilde I, et al. Highly purified lipoteichoic acid activates neutrophil granulocytes and delays their spontaneous apoptosis via CD14 and TLR2. J Leukoc Biol 2003 [Epub ahead of print].
Moulding DA, Walter C, Hart CA, Edwards SW. Effects of staphylococcal enterotoxins on human neutrophil functions and apoptosis. Infect Immun 1999; 67: 2312–2318.
Zysk G, Bejo L, Schneider-Wald BK, Nau R, Heinz H. Induction of necrosis and apoptosis of neutrophil granulocytes by Streptococcus pneumoniae. Clin Exp Immunol 2000; 122: 61–66.
Engelich G, White M, Hartshorn KL. Neutrophil survival is markedly reduced by incubation with influenza virus and Streptococcus pneumoniae: Role of respiratory burst. J Leukoc Biol 2001; 69: 50–56.
Kisich KO, Higgins M, Diamond G, Heifets L. Tumor necrosis factor alpha stimulates killing of Mycobacterium tuberculosis by human neutrophils. Infect Immun 2002; 70: 4591–4599.
Kasahara K, Sato I, Ogura K, Takeuchi H, Kobayashi K, Adachi M. Expression of chemokines and induction of rapid cell death in human blood neutrophils by Mycobacterium tuberculosis. Expression of chemokines and induction of rapid cell death in human blood neutrophils by Mycobacterium tuberculosis. J Infect Dis 1998; 178: 127–137.
Weiss SJ. Tissue destruction by neutrophils. N Engl J Med 1989; 320: 365–376.
Gamberale R, Giordano M, Trevani AS, Andonegui G, Geffner JR. Modulation of human neutrophil apoptosis by immune complexes. J Immunol 1998; 161: 3666–3674.
Schettini J, Salamone G, Trevani A, et al. Stimulation of neutrophil apoptosis by immobilized IgA. J Leukoc Biol 2002; 72: 685–691.
Kobayashi SD, Voyich JM, Braughton KR, DeLeo FR. Down-regulation of proinflammatory capacity during apoptosis in human polymorphonuclear leukocytes. J Immunol 2003; 170: 3357–3368.
Suter S, Schaad UB, Roux L, Nydegger UE, Waldvogel FA. Granulocyte neutral proteases and Pseudomonas elastase as possible causes of airway damage in patients with cystic fibrosis. J Infect Dis 1984; 149: 523–531.
Amitani R, Wilson R, Rutman A, et al. Effects of human neutrophil elastase and Pseudomonas aeruginosa proteinases on human respiratory epithelium. Am J Respir Cell Mol Biol 1991; 4: 26–32.
Power C, O’Connor CM, MacFarlane D, et al. Neutrophil collagenase in sputum from patients with cystic fibrosis. Am J Respir Crit Care Med 1994; 150: 818–822.
Stone PJ, Konstan MW, Berger M, Dorkin HL, Franzblau C, Snider GL. Elastin and collagen degradation products inurine of patients with cystic fibrosis. Am J Respir Crit Care Med 1995; 152: 157–162.
Koller DY, Urbanek R, Gotz M. Increased degranulation of eosinophil and neutrophil granulocytes in cystic fibrosis. Am J Respir Crit Care Med 1995; 152: 629–633.
Gillet Y, Issartel B, Vanhems P, et al. Association between Staphylococcus aureus strains carrying gene for Panton-Valentine leukocidin and highly lethal necrotising pneumonia in young immunocompetent patients. Lancet 2002; 359: 753–759.
Cox G, Crossley J, Xing Z. Macrophage engulfment of apoptotic neutrophils contributes to the resolution of acute pulmonary inflammation in vivo. Am J Respir Cell Mol Biol 1995; 12: 232–237.
Kobayashi SD, Voyich JM, Braughton KR, et al. Gene expression profiling provides insight into the pathophysiology of chronic granulomatous disease. J Immunol 2004; 172: 636–643.
Hampton MB, Vissers MC, Keenan JI, Winterbourn C. Oxidant-mediated phosphatidylserine exposure and acrophage uptake of activated neutrophils: Possible impairment in chronic granulomatous disease. J Leukoc Biol 2002; 71: 775–781.
Trump BF, Berezesky IK, Chang SH, Phelps PC. The path ways of cell death: Oncosis, apoptosis, and necrosis. Toxicol Pathol 1997; 25: 82–88.
Colotta F, Re F, Polentarutti N, Sozzani S, Mantovani A. Modulation of granulocyte survival and programmed cell death by cytokines and bacterial products. Blood 1992; 80: 2012–2020.
Stehle HW, Leblebicioglu B, Walters JD. Short-chain carboxylic acids produced by gram-negative anaerobic bacteria can accelerate or delay polymorphonuclear leukocyte apoptosis in vitro. J Periodontol 2001; 72: 1059–1063.
Webster P, IJdo JW, Chicoine LM, Fikrig E. The agent of Human Granulocytic Ehrlichiosis resides in an endosomal compartment. J Clin Invest 1998; 101: 1932–1941.
Hannah S, Mecklenburgh K, Rahman I, et al. Hypoxia prolongs neutrophil survival in vitro. FEBS Lett 1995; 372: 233–237.
Kasahara Y, Iwai K, Yachie A, et al. Involvement of reactive oxygen intermediates in spontaneous and CD95 (Fas/APO-1)-mediated apoptosis of neutrophils. Blood 1997; 89: 1748–1753.
Oishi K, Machida K. Inhibition of neutrophil apoptosis by antioxidants in culture medium. Scand J Immunol 1997; 45: 21–27.
Rollet-Labelle E, Grange MJ, Elbim C, Marquetty C, Gougerot-Pocidalo MA, Pasquier C. Hydroxyl radical as a potential intracellular mediator of polymorphonuclear neutrophil apoptosis. Free Radic Biol Med 1998; 24: 563–572.
Gardai S, Whitlock BB, Helgason C, et al. Activation of SHIP by NADPH oxidase-stimulated Lyn leads to enhanced apoposis in neutrophils. J Biol Chem 2002; 277: 5236–5246.
Zhang B, Hirahashi J, Cullere X, Mayadas TN. Elucidation of molecular events leading to neutrophil apoptosis following phagocytosis: Cross-talk between caspase 8, reactive oxygen species, and MAPK/ERK activation. J Biol Chem 2003; 278: 28443–28454.
Kobayashi SD, Voyich JM, Buhl CL, Stahl RM, DeLeo FR. Global changes in gene expression by human polymorphonu clear leukocytes during receptor-mediated phagocytosis: Cell fate is regulated at the level of gene expression. Proc Natl Acad Sci USA 2002; 99: 6901–6906.
Ottonello L, Frumento G, Arduino N, Dapino P, Tortolina G, Dallegri F. Immune complex stimulation of neutrophil apoptosis: Investigating the involvement of oxidative and nonoxidative pathways. Free Radic Biol Med 2001; 30: 161–169.
Ottonello L, Frumento G, Arduino N, et al. Differential regulation of spontaneous and immune complex-induced neutrophil apoptosis by proinflammatory cytokines. Role of oxidants, Bax and caspase-3. J Leukoc Biol 2002; 72: 125–132.
Kobayashi SD, Voyich JM, Somerville GA, et al. Anapoptosis differentiation program in human polymorphonuclear leukocytes facilitates resolution of inflammation. J Leukoc Biol 2003; 73: 315–322.
Kobayashi SD, Voyich JM, DeLeo FR. Regulation of the neutrophil-mediated inflammatory response to infection. Microbes Infect 2003; 5: 1337–1344.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
DeLeo, F.R. Modulation of phagocyte apoptosis by bacterial pathogens. Apoptosis 9, 399–413 (2004). https://doi.org/10.1023/B:APPT.0000031448.64969.fa
Issue Date:
DOI: https://doi.org/10.1023/B:APPT.0000031448.64969.fa