Abstract
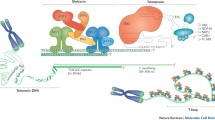
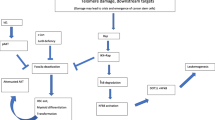
Chromosomal instability appears early during breast carcinogenesis and is considered a major driving force in malignant transformation. While current evidence suggests that centrosomal and mitotic checkpoint defects may, in large part, account for numerical chromosomal abnormalities, the mechanisms underlying structural chromosomal abnormalities remain largely unknown. Telomeres stabilize and protect chromosomal termini, but shorten due to cell division and oxidative damage. Moderate telomere shortening signals a tumor suppressive growth arrest in normal cells. Critically short telomeres, in the setting of abrogated DNA damage checkpoints, cause chromosomal instability due to end-to-end chromosomal fusions, subsequent breakage, and rearrangement, resulting in an increased cancer incidence in animal models. Recent results from high resolution in situ telomere length assessment in human breast tissues indicate that significant telomere shortening is prevalent in preinvasive breast lesions (DCIS), as well as focal areas of histologically normal epithelium from which breast carcinoma is thought to arise. Telomere shortening is therefore a strong candidate for the cause of structural chromosome defects that contribute to breast cancer development.
Similar content being viewed by others
REFERENCES
E. R. Fearon and B. Vogelstein (1990). A genetic model for colorectal tumorigenesis. Cell 61(5):759-767.
C. D. Heinen, C. Schmutte, and R. Fishel (2002). DNA repair and tumorigenesis: Lessons from hereditary cancer syndromes. Cancer Biol. Ther. 1(5):477-485.
P. L. Welcsh and M. C. King (2001). BRCA1 and BRCA2 and the genetics of breast and ovarian cancer. Hum. Mol. Genet. 10(7):705-713.
K. L. Nathanson and B. L. Weber (2001). “Other” breast cancer susceptibility genes: Searching for more holy grail. Hum. Mol. Genet. 10(7):715-720.
C. Lengauer, K. W. Kinzler, and B. Vogelstein (1998). Genetic instabilities in human cancers. Nature 396(6712):643-649.
C. Adem, C. L. Soderberg, J. M. Cunningham, C. Reynolds, T. J. Sebo, S. N. Thibodeau, et al.(2003). Microsatellite instability in hereditary and sporadic breast cancers. Int. J. Cancer 107(4):580-582.
B. L. King, S. C. Tsai, M. E. Gryga, T. G. D'Aquila, S. A. Seelig, L. E. Morrison, et al.(2003). Detection of chromosomal instability in paired breast surgery and ductal lavage specimens by interphase fluorescence in situ hybridization. Clin. Cancer Res. 9(4):1509-1516.
D. S. Yoon, R. P. Wersto, W. Zhou, F. J. Chrest, E. S. Garrett, T. K. Kwon, et al.(2002). Variable levels of chromosomal instability and mitotic spindle checkpoint defects in breast cancer. Am. J. Pathol. 161(2):391-397.
J. L. Salisbury (2001). The contribution of epigenetic changes to abnormal centrosomes and genomic instability in breast cancer. J. Mammary Gland Biol. Neoplasia 6(2):203-212.
P. O'Connell (2003). Genetic and cytogenetic analyses of breast cancer yield different perspectives of a complex disease. Breast Cancer Res. Treat. 78(3):347-357.
B. J. Miller, D. Wang, R. Krahe, and F. A. Wright (2003). Pooled analysis of loss of heterozygosity in breast cancer: A genome scan provides comparative evidence for multiple tumor suppressors and identifies novel candidate regions. Am. J. Hum. Genet. 73(4):748-767.
M. R. Teixeira, N. Pandis, and S. Heim (2002). Cytogenetic clues to breast carcinogenesis. Genes Chromosomes Cancer 33(1):1-16.
F. M. Waldman, S. DeVries, K. L. Chew, D. H. Moore, 2nd, K. Kerlikowske, and B. M. Ljung (2000). Chromosomal alterations in ductal carcinomas in situ and their in situ recurrences. J. Natl. Cancer Inst. 92(4):313-320.
E. Moore, H. Magee, J. Coyne, T. Gorey, and P. A. Dervan (1999). Widespread chromosomal abnormalities in high-grade ductal carcinoma in situ of the breast. Comparative genomic hybridization study of pure high-grade DCIS. J. Pathol. 187(4):403-409.
H. Buerger, F. Otterbach, R. Simon, C. Poremba, R. Diallo, T. Decker, et al.(1999). Comparative genomic hybridization of ductal carcinoma in situ of the breast-evidence of multiple genetic pathways. J. Pathol. 187(4):396-402.
M. Ciullo, M. A. Debily, L. Rozier, M. Autiero, A. Billault, V. Mayau, et al.(2002). Initiation of the breakage-fusion-bridge mechanism through common fragile site activation in human breast cancer cells: The model of PIP gene duplication from a break at FRA7I. Hum. Mol. Genet. 11(23):2887- 2894.
J. C. Lung, J. S. Chu, J. C. Yu, C. T. Yue, Y. L. Lo, C. Y. Shen, et al.(2002). Aberrant expression of cell-cycle regulator cyclin D1 in breast cancer is related to chromosomal genomic instability. Genes Chromosomes Cancer 34(3):276-284.
J. A. O'shaughnessy, G. J. Kelloff, G. B. Gordon, A. J. Dannenberg, W. K. Hong, C. J. Fabian, et al.(2002). Treatment and prevention of intraepithelial neoplasia: An important target for accelerated new agent development. Clin. Cancer Res. 8(2):314-346.
E. H. Blackburn (1991). Structure and function of telomeres. Nature 350:569-572.
T. von Zglinicki (2002). Oxidative stress shortens telomeres. Trends Biochem. Sci. 27(7):339-344.
C. W. Greider and E. H. Blackburn (1985). Identification of a specific telomere terminal transferase activity in Tetrahymena extracts. Cell 43:405-413.
R. R. Reddel (2003). Alternative lengthening of telomeres, telomerase, and cancer. Cancer Lett. 194(2):155-162.
C. Counter, A. Avilion, C. LeFeuvre, N. Stewart, C. Greider, C. Harley, et al.(1992). Telomere shortening associated with chromosome instability is arrested in immortal cells which express telomerase activity. EMBO J. 11:1921-1929.
B. McClintock (1941). The stability of broken ends of chromosomes in Zea mays. Genetics 26:234-282.
M. A. Blasco (2002). Telomerase beyond telomeres. Nat. Rev. Cancer 2(8):627-633.
S. E. Artandi and R. A. DePinho (2000). A critical role for telomeres in suppressing and facilitating carcinogenesis. Curr. Opin. Genet. Dev. 10(1):39-46.
D. M. Feldser, J. A. Hackett, and C. W. Greider (2003). Telomere dysfunction and the initiation of genome instability. Nat. Rev. Cancer 3(8):623-627.
M. A. Blasco, H. W. Lee, M. P. Hande, E. Samper, P. M. Lansdorp, R. A. DePinho, et al.(1997). Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91(1):25-34.
K. L. Rudolph, S. Chang, H. W. Lee, M. Blasco, G. J. Gottlieb, C. Greider, et al.(1999). Longevity, stress response, and cancer in aging telomerase-deficient mice. Cell 96(5):701-712.
R. A. DePinho and K. K. Wong (2003). The age of cancer: Telomeres, checkpoints, and longevity. J. Clin. Invest. 111(7):S9-S14.
S. E. Artandi, S. Chang, S. L. Lee, S. Alson, G. J. Gottlieb, L. Chin, et al.(2000). Telomere dysfunction promotes nonre-ciprocal translocations and epithelial cancers in mice. Nature 406(6796):641-645.
R. C. OHagan, S. Chang, R. S. Maser, R. Mohan, S. E. Artandi, L. Chin, et al.(2002). Telomere dysfunction provokes regional amplification and deletion in cancer genomes. Cancer Cell 2(2):149-155.
D. Broccoli, L. A. Godley, L. A. Donehower, H. E. Varmus, and T. de Lange (1996). Telomerase activation in mouse mammary tumors: Lack of detectable telomere shortening and evidence for regulation of telomerase RNA with cell proliferation. Mol. Cell Biol. 16(7):3765-3772.
S. E. Artandi, S. Alson, M. K. Tietze, N. E. Sharpless, S. Ye, R.A. Greenberg,et al.(2002). Constitutive telomerase expression promotes mammary carcinomas in aging mice. Proc. Natl. Acad. Sci. U.S.A. 99(12):8191-8196.
E. Odagiri, N. Kanada, K. Jibiki, R. Demura, E. Aikawa, and H. Demura (1994). Reduction of telomeric length and c-erbB-2 gene amplification in human breast cancer, fibroade-noma, and gynecomastia. Relationship to histologic grade and clinical parameters. Cancer 73(12):2978-2984.
P. Rogalla, C. Rohen, U. Bonk, and J. Bullerdiek (1996). Telomeric repeat fragment lengths are not correlated to histological grading in 85 breast cancers. Cancer Lett. 106(2):155-161.
K. Takubo, K. Nakamura, T. Arai, K. Nakachi, and M. Ebuchi (1998). Telomere length in breast carcinoma of the young and aged. Nippon Rinsho 56(5):1283-1286.
S.Y. Rha, K.H. Park, T.S. Kim, N.C. Yoo, W.I. Yang, J. K. Roh, et al.(1999). Changes of telomerase and telomere lengths in paired normal and cancer tissues of breast. Int. J. Oncol. 15(4):839-845.
J. K. Griffith, J. E. Bryant, C. A. Fordyce, F. D. Gilliland, N. E. Joste, and R. K. Moyzis (1999). Reduced telomere DNA content is correlated with genomic instability and metastasis in invasive human breast carcinoma. Breast Cancer Res. Treat 54(1):59-64.
N. W. Kim, M. A. Piatyszek, K. R. Prowse, C. B. Harley, M.D. West, P.L. Ho,et al.(1994). Specific association of human telomerase activity with immortal cells and cancer. Science 266(5193):2011-2015.
E. Hiyama, L. Gollahon, T. Kataoka, K. Kuroi, T. Yokoyama, A. F. Gazdar, et al.(1996). Telomerase activity in human breast tumors. J. Natl. Cancer Inst. 88(2):116-122.
G. M. Clark, C. K. Osborne, D. Levitt, F. Wu, and N. W. Kim (1997). Telomerase activity and survival of patients with node-positive breast cancer. J. Natl. Cancer Inst. 89(24):1874-1881.
A. Hoos, H. H. Hepp, S. Kaul, T. Ahlert, G. Bastert, and D. Wallwiener (1998). Telomerase activity correlates with tumor aggressiveness and reflects therapy effect in breast cancer. Int. J. Cancer 79(1):8-12.
K. Mokbel, C. N. Parris, M. Ghilchik, G. Williams, and R. F. Newbold (1999). The association between telomerase, histopathological parameters, and KI-67 expression in breast cancer. Am. J. Surg. 178(1):69-72.
K. M. Mokbel, C. N. Parris, M. Ghilchik, C. N. Amerasinghe, and R. F. Newbold (2000). Telomerase activity and lym-phovascular invasion in breast cancer. Eur. J. Surg. Oncol. 26(1):30-33.
A. Papadopoulou, T. Trangas, M. R. Teixeira, S. Heim, E. Dimitriadis, H. Tsarouha, et al.(2003). Telomerase activity and genetic alterations in primary breast carcinomas. Neoplasia 5(2):170-178.
I. Bieche, C. Nogues, V. Paradis, M. Olivi, P. Bedossa, R. Lidereau, et al.(2000). Quantitation of hTERT gene expression in sporadic breast tumors with a real-time reverse transcription-polymerase chain reaction assay. Clin. Cancer Res. 6(2):452-459.
R. L. Loveday, J. Greenman, P. J. Drew, J. R. Monson, and M. J. Kerin (1999). Genetic changes associated with telom-erase activity in breast cancer. Int. J. Cancer 84(5):516-520.
L. A. Carey, N. W. Kim, S. Goodman, J. Marks, G. Henderson, C. B. Umbricht, et al.(1999). Telomerase activity and prognosis in primary breast cancers. J. Clin. Oncol. 17(10):3075-3081.
L. A. Carey, C. A. Hedican, G. S. Henderson, C. B. Umbricht, J. S. Dome, D. Varon, et al.(1998). Careful histological confirmation and microdissection reveal telomerase activity in otherwise telomerase-negative breast cancers. Clin. Cancer Res. 4(2):435-440.
T. Sugino, K. Yoshida, J. Bolodeoku, D. Tarin, and S. Goodison (1997). Telomerase activity and its inhibition in benign and malignant breast lesions. J. Pathol. 183(1):57-61.
C. Poremba, K. R. Shroyer, M. Frost, R. Diallo, F. Fogt, K. L. Schafer, et al.(1999). Telomerase is a highly sensitive and specific molecular marker in fine-needle aspirates of breast lesions. J. Clin. Oncol. 17(7):2020-2026.
E. Hiyama, T. Saeki, K. Hiyama, S. Takashima, J. W. Shay, Y. Matsuura, et al.(2000). Telomerase activity as a marker of breast carcinoma in fine-needle aspirated samples. Cancer 90(4):235-238.
J. I. Tsao, Y. L. Zhao, J. Lukas, X. W. Yang, A. Shah, M. Press, et al.(1997). Telomerase activity in normal and neoplastic breast. Clinical Cancer Research 3(4):627-631.
C. Poremba, W. Bocker, H. Willenbring, K. L. Schafer, F. Otterbach, H. Burger, et al.(1998). Telomerase activity in human proliferative breast lesions. Int. J. Oncol. 12(3):641- 648.
C. B. Umbricht, M. E. Sherman, J. Dome, L. A. Carey, J. Marks, N. Kim, et al.(1999). Telomerase activity in ductal carcinoma in situ and invasive breast cancer. Oncogene 18(22):3407-3414.
A. K. Meeker, W. R. Gage, J. L. Hicks, I. Simon, J. R. Coffman, E. A. Platz, et al.(2002). Telomere length assessment in human archival tissues: Combined telomere fluorescence in situ hybridization and immunostaining. Am. J. Pathol. 160(4):1259-1268.
A. K. Meeker, J. L. Hicks, E. A. Platz, G. E. March, C. J. Bennett, M. J. Delannoy, et al.(2002). Telomere shortening is an early somatic DNA alteration in human prostate tumorigenesis. Cancer Res. 62(22):6405-6409.
N. T. van Heek, A. K. Meeker, S. E. Kern, C. J. Yeo, K. D. Lillemoe, J. L. Cameron, et al.(2002). Telomere shortening is nearly universal in pancreatic intraepithelial neoplasia. Am. J. Pathol. 161(5):1541-1547.
J. N. O'sullivan, M. P. Bronner, T. A. Brentnall, J. C. Finley, W. T. Shen, S. Emerson, et al.(2002). Chromosomal instability in ulcerative colitis is related to telomere shortening. Nat. Genet. 32(2):280-284.
B. Vukovic, P. C. Park, J. Al-Maghrabi, B. Beheshti, J. Sweet, A. Evans, et al.(2003). Evidence of multifocality of telomere erosion in high-grade prostatic intraepithelial neoplasia (HPIN) and concurrent carcinoma. Oncogene 22(13):1978-1987.
F. A. Tavassoli and P. Devilee (eds.) (2003). Tumors of the Breast and Female Genital Organs, World Health Organization Classification of Tumors, International Agency for Research on Cancer (IARC), Lyon, France.
C. W. Elston and I. O. Ellis (1998). Classification of malignant breast disease. In C. W. Elston and I. O. Ellis (eds.), The Breas t, Churchill Livingston, London, p. 243.
S. R. Romanov, B. K. Kozakiewicz, C. R. Holst, M. R. Stampfer, L. M. Haupt, and T. D. Tlsty (2001). Normal human mammary epithelial cells spontaneously escape senescence and acquire genomic changes. Nature 409(6820):633- 637.
P. Yaswen and M. R. Stampfer (2002). Molecular changes accompanying senescence and immortalization of cultured human mammary epithelial cells. Int. J. Biochem. Cell Biol. 34(11):1382-1394.
T. D. Tlsty, S. R. Romanov, B. K. Kozakiewicz, C. R. Holst, L. M. Haupt, and Y. G. Crawford (2001). Loss of chromosomal integrity in human mammary epithelial cells subsequent to escape from senescence. J. Mammary Gland Biol. Neoplasia 6(2):235-243.
C. R. Holst, G. J. Nuovo, M. Esteller, K. Chew, S. B. Baylin, J. G. Herman, et al.(2003). Methylation of p16(INK4a) promot-ers occurs in vivo in histologically normal human mammary epithelia. Cancer Res. 63(7):1596-1601.
M. R. Stampfer, J. Garbe, G. Levine, S. Lichtsteiner, A. P. Vasserot, and P. Yaswen (2001). Expression of the telomerase catalytic subunit, hTERT, induces resistance to transforming growth factor beta growth inhibition in p16INK4A( —) human mammary epithelial cells. Proc. Natl. Acad. Sci. U.S.A. 98(8):4498-4503.
K. A. Kolquist, L. W. Ellisen, C. M. Counter, M. Meyerson, L. K. Tan, R. A. Weinberg, et al.(1998). Expression of TERT in early premalignant lesions and a subset of cells in normal tissues. Nat. Genet. 19(2):182-186.
A. Zhang, C. Zheng, M. Hou, C. Lindvall, K. J. Li, F. Erlandsson, et al.(2003). Deletion of the telomerase reverse transcriptase gene and haploinsufficiency of telomere maintenance in Cri du chat syndrome. Am. J. Hum. Genet. 72(4):940- 948.
W. Cui, S. Aslam, J. Fletcher, D. Wylie, M. Clinton, and A. J. Clark (2002). Stabilization of telomere length and karyotypic stability are directly correlated with the level of hTERT gene expression in primary fibroblasts. J. Biol. Chem. 277(41):38531-38539.
Y. Liu, H. Kha, M. Ungrin, M. O. Robinson, and L. Harrington (2002). Preferential maintenance of critically short telomeres in mammalian cells heterozygous for mTert. Proc. Natl. Acad. Sci. U.S.A. 99(6):3597-3602.
K. S. Hathcock, M. T. Hemann, K. K. Opperman, M. A. Strong, C. W. Greider, and R. J. Hodes (2002). Haploinsufficiency of mTR results in defects in telomere elongation. Proc. Natl. Acad. Sci. U.S.A. 99(6):3591-3596.
J. W. Shay and W. E. Wright (2002). Telomerase: A target for cancer therapeutics. Cancer Cell 2(4):257-265.
W. C. Hahn, S. A. Stewart, M. W. Brooks, S. G. York, E. Eaton, A. Kurachi, et al.(1999). Inhibition of telomerase lim-its the growth of human cancer cells. Nat. Med. 5(10):1164- 1170.
B. Herbert, A. E. Pitts, S. I. Baker, S. E. Hamilton, W. E. Wright, J. W. Shay, et al.(1999). Inhibition of human telomerase in immortal human cells leads to progressive telomere shortening and cell death. Proc. Natl. Acad. Sci. U.S.A. 96(25):14276-14281.
X. Zhang, Z. Chen, Y. Chen, and Tong T. (2003). Delivering antisense telomerase RNA by a hybrid adenovirus/adeno-associated virus significantly suppresses the malignant phenotype and enhances cell apoptosis of human breast cancer cells. Oncogene 22(16):2405- 2416.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Meeker, A.K., Argani, P. Telomere Shortening Occurs Early During Breast Tumorigenesis: A Cause of Chromosome Destabilization Underlying Malignant Transformation?. J Mammary Gland Biol Neoplasia 9, 285–296 (2004). https://doi.org/10.1023/B:JOMG.0000048775.04140.92
Issue Date:
DOI: https://doi.org/10.1023/B:JOMG.0000048775.04140.92