Abstract
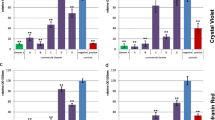
Epifluorescence microscope methods, namely BacLight, direct epifluorescence filter technique and Rhodamine 123, consistently underestimated plate bacterial counts in a 4-chlorophenol degrading consortium. Cells capable of passing through 0.2 μm filters, referred as `ultramicrocells', were found. Although cell counts were higher when traditional methods were used, BacLight and direct epifluorescence filter technique were convenient techniques for the systematic monitoring of bacteria involved in biodegradation processes, as results were consistent and available within a short time.
Similar content being viewed by others
References
Bartscht K, Cypionka H, Overmann J (1999) Evaluation of cell activity and of methods for the cultivation of bacteria from a natural lake community. FEMS Microbiol. Ecol. 28: 249–259.
Carvalho MF, Vasconcelos I, Bull AT, Castro PML (2001) A GAC biofilm reactor for the continuous degradation of 4-chlorophenol: treatment efficiency and microbial analysis. Appl. Microbiol. Biotechnol. 57: 419–426.
Eguchi M, Nishikawa T, MacDonald K, Cavicchioli R, Gottschal JC, Kjelleberg S (1996) Responses to stress and nutrient availability by the marine ultramicrobacterium Sphingomonas sp. strain RB2256. Appl. Environ. Microbiol. 62: 1287–1294.
Fiksdal L, Tryland I (1999) Effect of u.v. light irradiation, starvation and heat on Escherichia coli ?-D-galactosidase activity and other potential viability parameters. J. Appl. Microbiol. 87: 62–71.
Jones CR, Chamberlain AHL, Adams MR (1999) An investigation of the presence of ultramicrocells in natural mineral water. Lett. Appl. Microbiol. 28: 275–279.
Kaprelyants AS, Kell DB (1992) Rapid assessment of bacterial viability and vitality by Rhodamine 123 and flow cytometry. J. Appl. Bact. 72: 410–422.
McFeters G, Yu F, Pyle B, Stewart P (1995) Physiological assessment of bacteria using fluorochromes. J. Microbiol. Meth. 21: 1–13.
Morita RY (1995) Starvation and miniaturisation of heterotrophs, with special emphasis on maintenance of the starved viable state. In: Fletcher M, Floodgate GD, eds. Bacteria in their Natural Environments. London: Academic Press, pp. 111–130.
Pettipher GL, Mansell R, McKinnon CH, Cousins CM (1980) Rapid membrane filtration-epifluorescence microscopy technique for direct enumeration of bacteria in raw milk. Appl. Environ. Microbiol. 39: 423–429.
Ramalho R, Cunha J, Albano H, Teixeira P, Gibbs P (2001) Improved methods for the enumeration of heterotrophic bacteria in bottled mineral waters. J. Microbiol. Meth. 44: 97–103.
Rapposch S, Zangerl P, Ginzinger W (2000) Influence of fluorescence of bacteria stained with acridine orange on the enumeration of microorganisms in raw milk. J. Dairy Sci. 83: 2753–2758.
Vollertsen J, Jahn A, Nielsen JL, Hvitved-Jacobsen T, Nielsen PH (2001) Comparison of methods for determination of microbial biomass in waste water. Water Res. 35: 1649–1658.
Watanabe K, Miyashita M, Harayama S (2000) Starvation improves survival of bacteria introduced into activated sludge. Appl. Environ. Microbiol. 66: 3905–3910.
Ziglio G, Andreottola G, Barbesti S, Boschetti G, Bruni L, Foladori P, Villa R (2002) Assessment of activated sludge viability with flow cytometry. Water Res. 36: 460–468.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pacheco, C., Alves, C., Barreiros, L. et al. Epifluorescence microscope methods for bacterial enumeration in a 4-chlorophenol degrading consortium. Biotechnology Letters 25, 2089–2092 (2003). https://doi.org/10.1023/B:BILE.0000007070.12514.64
Issue Date:
DOI: https://doi.org/10.1023/B:BILE.0000007070.12514.64