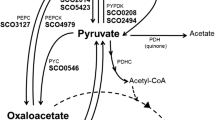
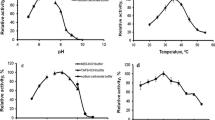
Abstract
The osmotolerant Zymomonas mobilis strain suc40, (containing plasmid pDS3154-inaZ), which is capable of producingsimultaneously ethanol and ice nuclei protein, was cultivated in a chemically defined complete sucrosemedium, as well as in a sugar beet molasses medium in continuous culture. The strain exhibited the normalMonod's relationship between biomass and dilution rate, and between growth substrate concentration and dilutionrate. Specific activities of a number of enzymes that appear to control important steps in the metabolic flux ofthe Entner-Doudoroff and pyruvate decarboxylation pathways were investigated over a range of growth ratesin steady state cultures. With the exception of glucose-6-phosphate dehydrogenase and gluconate kinase, all ofthe enzymes exhibited a very similar pattern for the wild type Z. mobilis CP4 and for the osmotolerant mutants,independent of the media used; the enzyme patterns remained relatively constant over the studied growth range.The specific activity of glucose-6-phosphate dehydrogenase was increased 2-fold by decreasing dilution rate insugar beet molasses. The specific activity of gluconate kinase was 2-fold lower at medium growth rates comparedwith that at either low or high growth rates. Pyruvate kinase, pyruvate decarboxylase and alcohol dehydrogenaseactivities were significantly higher compared with those of the enzymes governing the early steps of the Entner-Doudoroff pathway. The present study, which was designed to determine the behaviour of important enzymes insucrosemetabolismof Z. mobilis suc40/pDS3154-inaZ grown in continuous culture showed that the microorganismrequired regulation of specific enzyme activities at the transcriptional level when sugar beet molasses were used asthe growth medium.
Similar content being viewed by others
References
Algar EM & Scopes R (1985) Studies on cell-free metabolism: ethanol production by extracts of Zymomonas mobilis.J.Bio-technol. 2: 275–287.
Anderson AJ & Dawes EA (1984) Regulation of glucose-6-phosphate dehydrogenase in Zymomonas mobilisCP4. FEMS Microbiol. Lett. 27: 23–27
Barnell WO, Cheol Yi K & Conway T (1990) Sequence and genetic organization of a Zymomonas mobilisgene cluster that encodes several enzymes of glucose metabolism. J. Bacteriol. 172: 7227–7240
Barrow KD, Collins JG, Norton RS, Rogers PL & Smith GM(1984) 31P nuclear magnetic resonance studies of the fermentation of glucose to ethanol by Zymomonas mobilis. J. Biol. Chem. 259: 5711–5716
Bringer-Meyer S & Sahm H (1988) Metabolic shifts in Zymomonas mobilisin response to growth conditions. FEMS Microbiol. Rev. 54: 131–142
Buchholz SE, Dooley MM & Eveleigh DE (1987) Zymomonas-an alcoholic enigma. Trends Biotechnol. 5: 199–204
Dawes EA, Ribbons DW & Large PJ (1966) The route of ethanol formation by Zymomonas mobilis. Biochem. J. 98: 795–803
Dean ACR (1972) Influence of environment on the control of enzyme synthesis. J. Appl. Chem. Biotechnol. 22: 245–259
Doelle HW (1982) The existence of two separate constitutive en-zymes for glucose and fructose in Zymomonas mobilis.Eur.J. Appl. Microbiol. Biotechnol. 15: 20–24
Doelle HW & Greenfield PF (1985) Fermentation pattern of Zy-momonas mobilisat high sucrose concentrations. Appl. Micro-biol. Biotechnol. 22: 411–415
Doelle WH, Kirk L, Crittenden R, Toh H & Doelle BM (1993) Zymomonas mobilis-science and industrial application. Crit. Rev. Biotechnol. 13: 57–98
Fein JE, Barber DL, Charley RC, Beveridge TJ & Lawford HG (1984) Effect of commercial feedstocks on growth and morpho-logy of Zymomonas mobilis.Biotechnol. Lett. 6: 123–128
Fry JC (1989) Analysis of variance and regression in aquatic bacteriology. Binary 1: 83–88
Gibbs M & DeMoss RD (1954) Anaerobic disimilation of 14 C-labelled glucose and fructose by Pseudomonas lindneri. J. Biol. Chem. 207: 689–694
Hoppner TC & Doelle HW (1983) Purification and kinetic charac-teristics of pyruvate decarboxylase and ethanol dehydrogenase from Zymomonas mobilisin relation to ethanol production.Eur. J. Appl. Microbiol. Biotechnol. 17: 152–157
Hunsley JR & Suelter CH (1969) Yeast pyruvate kinase I. Puri-fication and some chemical properties. J. Biol. Chem. 244: 4815–4818
Jones CW & Doelle WH (1991) Kinetic control of ethanol produc-tion by Zymomonas mobilis.Appl. Microbiol. Biotechnol. 35: 4–9
Karagouni AD & Slater HJ (1979) Enzymes of the Calvin cycle and intermediary metabolism in the cyanobacterium Anacystis nidulansgrown in chemostat culture. J. Gen. Microbiol. 115: 369–376
Kuby SA & Noltmann EA (1966) Glucose-6-phosphate dehydro-genase from brewer's yeast. Methods Enzymol. 9: 116–125
Lowry OH, Rosebrough NJ, Farr LA & Randall RJ (1951) Protein measurement with the folin phenol reagent. J. Biol. Chem. 193: 265–275
Maitra PK & Lobo Z (1971) A kinetic study of glycolytic enzyme synthesis in yeast. J. Biol. Chem. 25: 475–488
Osman YA, Conway T, Bonetti SJ & Ingram LO (1987) Glycolytic flux in Zymomonas mobilis: enzyme and metabolite levels during batch fermentation. J. Bacteriol. 169: 3726–3736
Park SC & Baratti J (1991) Batch fermentation kinetics of sugar beet molasses by Zymomonas mobilis. Biotechnol. Bioeng. 38: 304–313
Park SC & Baratti J (1993) Effects of potassium chloride on ethanol production by an osmotolerant mutant of Zymomonas mobilis.J. Biotechnol. Bioeng. 38: 542–549
Pawluk A, Scopes RK & Griffiths-Smith K (1986) Isolation and properties of the glycolytic enzymes from Zymomonas mobilis. Biochem. J. 238: 275–281
Petersen RG (1985) Separation of means. In: Design and Analysis of Experiments:Statistics: Textbooks and Monographs Series, Vol. 6 6 (pp7 2–111). Marcel-Dekker, New York
Rhee SK, Pagan RJ, Lefevre MF, Wong L & Rogers PL (1984) Ethanol production from desalted molasses using S. uvarumand Zymomonas mobilis. J. Ferment. Technol. 62: 297–300
Rogers PL, Lee KJ & Tribe DE (1980) High productivity eth-anol fermentations with Zymomonas mobilis. Proc. Biochem. Aug./Sept., 7–11
Savvides AL, Kallimanis A, Varsaki A, Koukkou AI, Drainas C, Typas MA & Karagouni AD (2000) Simultaneous ethanol and bacterial ice nuclei production from sugar beet molasses by a Zymomonas mobilisCP4 mutant expressing the inaZgene of Pseudomonas syringaein continuous cultures. J. Appl. Micro-biol. 89: 1002–1008
Scopes RK (1997) Allosteric control of Zymomonas mobilisglucose-6-phosphate dehydrogenase by phosphoenolpyruvate. Biochem. J. 326: 731–735
Scopes RK, Testolin V, Stoter A, Griffiths-Smith K & Algar EM (1985) Simultaneous purification and characterization of gluc-okinase, fructokinase and glucose-6-phosphate dehydrogenase from Zymomonas mobilis.Biochem. J. 228: 627–634
Toran-Diaz I, Jain VK & Baratti JC (1984) Ethanol production from fructose in continuous culture by free and flocculent cells of Zymomonas mobilis. Biotechnol. Lett. 6: 389–394
Tsai CS, Shi JL, Beehler BW & Beck B (1992) Enzyme activities of D-glucose metabolism in the fission yeast Schizosaccharomyces pombe. Can. J. Microbiol. 38: 1313–1319
Viikari L (1984) Formation of levan and sorbitol from sucrose by Zymomonas mobilis. Appl. Microbiol. Biotechnol. 19: 252–255
Zachariou M & Scopes RK (1985) Gluconate kinase from Zymomo-nas mobilis: isolation and characterization. Biochem. Int. 10: 367–371
Rights and permissions
About this article
Cite this article
Sawides, A.L., Kalliopi, K.I., Typas, m.A. et al. Enzymes of the Entner-Doudoroff and pyruvate decarboxylation pathways in Zymomonas mobilis wild-type CP4 and mutant strains grown in continuous culture. Antonie Van Leeuwenhoek 80, 287–295 (2001). https://doi.org/10.1023/B:ANTO.0000044214.58537.6b
Issue Date:
DOI: https://doi.org/10.1023/B:ANTO.0000044214.58537.6b