Abstract
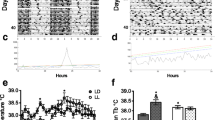
The nocturnal melatonin (MLT) surge is a relevant oncostatic signal for a variety of experimental malignancies. Population studies support the hypothesis that exposure to light at night may represent a new risk factor for breast cancer possibly through the suppression of pineal MLT production and/or circadian disruption. We tested the ability of constant light exposure to suppress MLT production in female nude rats and stimulate the growth of tissue-isolated MCF-7 human breast cancer xenografts via increased tumor linoleic acid (LA) metabolism. Rats maintained on an alternating light/dark cycle (L:D group) exhibited a robust circadian MLT rhythm that was abolished following constant light exposure. During the exposure of animals bearing tissue-isolated human MCF-7 breast cancer xenografts to constant light, the rate of tumor growth markedly increased relative to the L:D group. Tumor LA uptake and its metabolism to the mitogen 13-hydroxyoctadecadienoic acid (13-HODE) were also substantially higher under constant light conditions. This is the first biological evidence for a potential link between constant light exposure and increased human breast oncogenesis involving MLT suppression and stimulation of tumor LA metabolism.
Similar content being viewed by others
References
Reiter RJ: Melatonin: a multifaceted messenger to the masses. Lab Med 25: 438-441, 1994.
Cassone VM: The pineal gland influences rat circadian activity rhythms in constant light. J Biol Rhythms 7: 27-40, 1992
Brainard GC, Richardson BA, King TS, Reiter RJ: The in-fluence of different light spectra on the suppression of pineal melatonin content in the Syrian hamster. Brain Res 294: 333-339, 1984
Brainard GC, Richardson BA, King TS, Matthews SA, Reiter RJ: The suppression of pineal melatonin content and Nacetyltransferase activity by different light irradiances in the Syrian hamster: a dose-response relationship. Endocrinology 113: 293-296, 1983
Minneman KP, Lynch H, Wurtman: Relationship between environmental light intensity and retina-mediated suppression of rat pineal serotonin-N-acetyltransferase. Life Sci 15: 1791-1796, 1974
Reiter RJ, Hurlbut EC, Brainard GC, Steinlechner S, Richardson BA: Influence of light irradiance on hydroxyindole-O-methyltransferase activity, serotonin-N-acetyltransferase activity, and radioimmunoassayable melatonin levels in the pineal gland of the diurnally active Richardson's Ground Squirrel. Brain Res 288: 151-157, 1983
Blask DE: An overview of the neuroendocrine regulation of experimental tumor growth by melatonin and its analogues and the therapeutic use of melatonin in oncology. In: Bartsch C, Bartsch H, Blask DE, Cardinali DP, Hrushesky WJM, Mecke D (eds) The Pineal Gland and Cancer. Springer, Berlin, 2001, pp 309-342
Blask DE, Sauer LA, Dauchy RT: Melatonin as a chronobiotic/ anticancer agent: cellular, biochemical, and molecular mechanisms of action and their implications for circadianbased cancer therapy. Curr Top Med Chem 2:113-132, 2002
Hill SM, Keifer T, Teplitzky S, Spriggs LL, Ram P: Modulation of the estrogen response pathway in human breast cancer cells by melatonin. In: Bartsch C, Bartsch H, Blask DE, Cardinali DP, Hrushesky WJM, Mecke D (eds) The Pineal Gland and Cancer. Springer, Berlin, 2001, pp 343-358
Jöchle W: Trends in photophysiologic concepts. Ann NY Acad Sci 117: 88-104, 1964
Kothari L, Shah PN, Mhatre MC: Pineal ablation in varying photoperiods and the incidence of 9,10-dimethyl-1, 2-benzanthracene-induced mammary cancer in rats. Cancer Lett 22: 99-102, 1984
Kothari L: Influence of chronic melatonin on 9,10-dimethyl-1, 2-benzanthracene-induced mammary tumors in female Holtzman rats exposed to continuous light. Oncology 44: 64-66, 1987
Anderson LE, Morris JE, Sasser LB, Stevens RG: Effect of constant light on DMBA mammary tumorigenesis in rats. Cancer Lett 148: 121-126, 2000
Stevens RG: Electric power use and breast cancer: a hypothesis. Am J Epidemiol 125: 556-561, 1987
Hansen J: Increased breast cancer risk among women who work predominantly at night. Epidemiology 12: 74-77, 2001
Davis S, Mirick DK, Stevens RG: Night shift work, light at night, and risk of breast cancer. J Natl Cancer Inst 93: 1557-1562, 2001
Schernhammer ES, Laden F, Speizer FE, Willett WC, Hunter DJ, Kawachi I, Colditz GA: Rotating night shifts and risk of breast cancer in women participating in the nurse's health study. J Natl Cancer Inst 93: 1563-1568, 2001
Hahn RA: Profound bilateral blindness and the incidence of breast cancer. Epidemiology 2: 208-210, 1991
Feychting M, Ústerland B, Ahlbom A: Reduced cancer incidence among the blind. Epidemiology 9: 490-494, 1998
Verkasalo P, Pukkala E, Stevens RG, Ojama, Rudanko S-L: Visual impairment and cancer: a population-based cohort study in Finland. Cancer Causes and Control 10: 13-20, 1999
Lockley SW, Skene DJ, Arendt J, Tabandeh H, Bird AC, Defrance R: Relationship between melatonin rhythms and visual loss in the blind. J Clin Endocrinol Metabiol 82: 3763-3770, 1997
Stevens RG, Rea MS: Light in the built environment: potential role of circadian disruption in endocrine disruption and breast cancer. Cancer Causes Control 12: 279-287, 2001
Blask DE, Sauer LA, Dauchy RT, Holowachuk EW, Ruhoff MS: Melatonin inhibition of cancer growth in vivo involves suppression of tumor fatty acid metabolism via melatonin receptor-mediated signal transduction events. Cancer Res 59: 4693-470, 1999
Dauchy RT, Blask DE, Sauer LA, Vaughan GM: Light contamination during the dark phase in ‘photoperiodicallycontrolled’ animal rooms: effect on tumor growth and metabolism in rats. Lab Anim Sci 47: 511-518, 1997
Dauchy RT, Blask DE, Sauer LA, Brainard GC, Krause JA: Dim light during darkness stimulates tumor progression by enhancing tumor fatty acid uptake and metabolism. Cancer Lett 144: 131-136, 1999
Folch JM, Lees M, Sloane-Stanley GH: A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226: 497-507, 1957
McDonald-Gibson RG: Quantitative measurements of arachidonic acid in tissues or fluids. In: Benedetto C, McDonald-Gibson RG, Nigam S, Slater TF (eds) Prostaglandins and Related Substances. IRL Press, Washington, pp 259-268, 1987
Tamarkin L, Danforth D, Lichter E, DeMoss M, Cohen M, Chabner B, Lippman M: Decreased nocturnal plasma melatonin peak in patients with estrogen receptor positive breast cancer. Science 216: 1003-1005, 1983
Hayashi K, Okatani Y: Mechanisms underlying the effects of estrogen on nocturnal melatonin synthesis in peripubertal female rats: relation to norepinephrine and adenylate cyclase. J Pineal Res 26: 178-183, 1999
Osborne CK, Hobbs K, Clark GM: Effect of estrogens and antiestrogens on growth of human breast cancer cells in athymic nude mice. Cancer Res 45: 584-590, 1985
Ram PT, Yuan L, Dai J, Kiefer T, Klotz DM, Spriggs LL, Hill SM: Differential responsiveness of MCF-7 human breast cancer cell line stocks to the pineal hormone, melatonin. J Pineal Res 28: 210-218, 2000
Liu X-H, Connolly JM, Rose DP: The 12-lipoxygenase genetransfected MCF-7 human breast cancer cell line exhibits estrogen-independent, but estrogen and omega-6 fatty acidstimulated proliferation in vitro, and enhanced growth in athymic nude mice. Cancer Lett 109: 223-230, 1996
Reiter RJ: The pineal gland, melatonin and reproduction. In: Bittar EE, Bittar N (eds) Principles of Medical Biology. Vol 12, JAI Press, Greenwich, 1998, pp 141-154
Mhatre MC, Shah PN, Juneja HS: Effect of varying photoperiods on mammary morphology, DNA synthesis, and hormone profile in female rats. J Natl Cancer Inst 72: 141-1415, 1984
Shah PN, Mhatre MC, Kothari LS: Effect of melatonin on mammary carcinogenesis in intact and pinealectomized rats in varying photoperiods. Cancer Res 44: 3403-3407, 1984
Seshadri R, Subramanian A, Kothari LS: Effect of neonatal pineal ablation on estradiol receptors in mammary glands of rats housed under varying photoperiods. Indian J Exp Biol 30: 162-164, 1992
Graham GC, Cook MR, Gerkovich MM, Sastre A: Examination of the melatonin hypothesis in women exposed at night to EMF or bright light. Environ Health Perspect 109: 501-507, 2001
Depres-Bummer P, Levi F, Metzger G, Touitou Y: Lightinduced suppression of the rat circadian system. Am J Physiol 268: R1111-R1116, 1995
Dillon DC, Easley SE, Asch BB, Cheney RT, Brydon L, Jockers R, Winston JS, Brooks JS, Hurd T, Asch HL: Differential expression of high-affinity melatonin receptors (MT1) in normal and malignant human breast tissue. Am J Clin Pathol 118: 451-458, 2002
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Blask, D.E., Dauchy, R.T., Sauer, L.A. et al. Growth and Fatty Acid Metabolism of Human Breast Cancer (MCF-7) Xenografts in Nude Rats: Impact of Constant Light-Induced Nocturnal Melatonin Suppression. Breast Cancer Res Treat 79, 313–320 (2003). https://doi.org/10.1023/A:1024030518065
Issue Date:
DOI: https://doi.org/10.1023/A:1024030518065